Introduction
Various studies have demonstrated that staying active is vital for maintaining overall health and an extended lifespan (Nourhashémi et al., 2002; Srikanthan and Karlamangla, 2011). Therefore, concentrated efforts to maintain and increase skeletal muscle mass are significant for human health. Skeletal muscle is responsible for daily psycho-motor activities and strength-related physical performance (Brook et al., 2016). With increasing age, there is a natural loss in muscle mass and strength, termed sarcopenia. This is associated with an increased risk for falling, disability, losing independence due to the inability to carry out daily activities, thus impacting the quality of life (da Silva Alexandre et al., 2014; Landi et al., 2012). Many studies have suggested that enhancing the dietary protein quantity and quality along with exercising are the key strategies to maintain skeletal muscle health, especially during old age (Burd et al., 2019).
It is known that protein supplementation increases muscle strength (Andersen et al., 2005), prevents the decrease of total muscle mass (Bauer et al., 2013), and contributes to muscle hypertrophy (Stark et al., 2012) and exercise performance. Pre- or post-exercise ingestion of protein or essential amino acids can contribute to an increase in skeletal muscle protein synthesis (Devries and Phillips, 2015). Studies on the effects of protein consumption on resistance training exercise have also revealed that consistent protein/amino acid supplementation results in skeletal muscle hypertrophy with chronic resistance training (Stark et al., 2012). Additionally, these effects were most prominent when supplementation occurred around the time of resistance exercise, rather than at a time distal to exercise (Stark et al., 2012).
Whey protein comprises 20% of the total milk proteins and is rich in branched and essential amino acids and functional peptides, providing multiple health benefits such as anti-oxidative and -inflammatory effects, antihypertensive action, anti-obesity potential, and noticeable lean body mass increment (Patel, 2015). Other than nutritional and functional attributes, whey proteins can also potentially impact functioning of the food market, as there is an increasing demand for lean body mass protein supplements (Ten Haaf Dominique et al., 2018). Direct and indirect evidence has suggested that whey protein is chiefly suitable to be used in conjunction with resistance exercise for stimulating muscle hypertrophy (Devries and Phillips, 2015). In particular, the intake of whey protein or branched chain amino acid supplements in the form of beverages or powders was found to be effective in increasing skeletal muscles in young and elderly people (Hulmi et al., 2010). Due to the hydrophobic nature of the whey protein, their actual bioavailability remains unclear, since they are resistant to enzymatic hydrolysis in the gut (Aich et al., 2015; Dalgalarrondo et al., 1995; Reddy et al., 1988). Most bioactive peptides in whey protein, such as glycomacropeptide, β-lactoglobulin, α-lactoalbumin, and lactoferrin, are found in the parent protein sequence in inactive forms (Morr and Ha, 1993). However, through bacterial fermentation, the culturing of microorganisms (bacteria or yeast) in a medium containing protein substrates, facilitates the production of proteolytic enzymes, which catalyze protein breakdown, ultimately resulting in the liberation of bioactive peptides (Kieliszek et al., 2021). Particularly, during protein fermentation, microorganisms produce protease to hydrolyze proteins that have higher nutritional value (Christensen et al., 2022). Microbial proteases are proteolytic enzymes that release peptides with 2–20 amino acids; however, their activities are largely dependent on protein chain length, amino acid composition, and the sequence of amino acids (Sanjukta and Rai, 2016). Fermentation is one of the earliest biotechnological methods of food preservation and processing to be extensively applied in the world [1]: Foods (fermented food, food additives, functional materials and live probiotics); intestines (aids digestion and promotes absorption, synthetic bioactive substances, inhibits harmful bacteria, diabetes, cardiopathy and allergy); and industry (energy, soil transformation and sewage treatment; Sun et al., 2022).
The bioconversion of proteins through fermentation may have beneficial effects on various physiological parameters. The effects of Lactobacillus casei fermented milk products have been examined in ovariectomized rats; findings indicated that bone weight, bone mineral density, and bone breaking force all significantly increased (Kim et al., 2009). Moreover, the enzymatic hydrolysis of milk proteins has induces greater muscle protein synthesis, presumably due to the significant absorption of degraded proteins compared to that of non-fermented intact proteins (Koopman et al., 2009). With regard to casein hydrolyzed with enzymes, the faster rate of amino acid appearance and the greater peak of amino acid concentration after hydrolysis indicate their potential beneficial effects on muscle hypertrophy (Pennings et al., 2011). Growth promotion effects in rats were also observed with ingestion of fermented milk compared to that of non-fermented milk (Hargrove and Alford, 1978). Supplementation of diet with fermented milk was accompanied by the enhancement of skeletal muscle strength and hypertrophy in response to resistance training (Bridge et al., 2019; Thomas et al., 2011). Non-fermented whey protein supplementation was tested on muscle hypertrophy in various resistance training studies, where findings indicated that specific peptides may provide muscle recovery capabilities, while their antioxidant capacities may also contribute towards recovery physiological responses to exercise (Devries and Phillips, 2015; Ha and Zemel, 2003; Stark et al., 2012). There have been several studies on the improvement of muscle parameters with whey protein supplementation (Andersen et al., 2005), fermented whey proteins (FWP) on protein digestion and absorption, increment of branched chain amino acids levels, and effects of fermentation on calcium absorption and bone density improvement (Matsuda et al., 2022). However, the effects of fermented whey supplementation on muscle strength and physical performance have not been investigated. Therefore, this study aimed to investigate the free amino acid changes caused by fermenting whey proteins with indigenous lactic acid bacteria (LAB) from kimchi. We also investigated the effects of diet supplementation with fermented and non-FWPs on the muscle strengthening factors and exercise performance of healthy middle-aged males for 8-weeks.
Materials and Methods
LAB were isolated from homemade kimchi that had been collected from different regions of Korea. Screening of selected strains was performed by observing colony shape on the plates, gram staining, and strain morphology using a microscope. Additionally, the identity of selected strains were confirmed by 16S-rRNA gene sequencing. The 16S-rRNA gene sequences were analyzed using the Gene Bank database (Macrogen, Seoul, Korea), with identification being performed based on the 16S-rRNA gene sequence homology.
Proteolytic activity was assessed using an agar diffusion method according to the method outlined by Zhang et al. (2013), with certain modifications. Milk agar plates containing 1.6% skimmed milk and 1.5% agar were prepared. Then, 100 μL of each strain at a final concentration of approximately 108 CFU/mL was inoculated onto plates and incubated at 37°C for 48 h. The absence of an inhibitory zone was subsequently observed.
The DH of WPC (Hilmar 8000 whey protein concentrate, Hilmar Ingredients, Hilmar, CA, USA) solution [7% (w/v) whey protein, 3% (w/v) glucose] was measured in samples of fermented WPC (every 4 h for 20 h) using the 2,4,6-trinitrobenzenesulfonic acid (TNBS) method (Adler-Nissen, 1979). The TNBS reagent consisted of 0.1% (w/v) TNBS in distilled water. All samples and standard solutions were prepared in 1% (w/v) sodium dodecyl sulphate solution. Subsequently, 2 mL samples were dissolved in distilled water to 10% (v/v) in a test tube and 4 mL of 0.72 N trichloroacetic acid was added. They were then incubated at 25°C for 20 min. Next, 0.2 mL of sample was taken after filtration through a PVDF 0.45 μm sterile syringe membrane filter (Sigl, Altötting, Germany) and added to a test tube containing 2 mL of 0.2125 M sodium phosphate buffer (pH 8.2). The 2 mL TNBS reagent was added to each test tube, and then incubated in the dark at 50°C for 60 min. After incubation, 4 mL of 0.1 N HCl was added to stop the reaction and mixed into each tube. Samples were then allowed to cool at room temperature for 30 min, before absorbance values were measured at 420 nm using a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA). The DH was defined as the proportion of the total number of peptide bonds that were cleaved during hydrolysis (Adler-Nissen, 1986) and calculated as follows:
Where h is the number of hydrolyzed peptide bonds and htot is the total number of peptide bonds present (Nielsen et al., 2001). Leucine was used as a standard (at concentrations ranging from 0 to 2 mM) to allow for the determination of free amino group contents of the samples.
Stock LAB culture was thawed, and strains were activated in MRS (BD Difco, Franklin Lakes, NJ, USA). The WPC solution was heated in a water bath at 37°C. Activated strains were used as inoculums at 1% (v/v). Hydrolysis was then performed in a shaking water bath at 37°C to 1.886×g for 20 h. Samples were aseptically withdrawn every 4 h throughout the 20 h incubation. Cell viability was determined by plating appropriate dilutions of the cultures in BCP agar. Plates were incubated at 37°C for 48 h and colony-forming units CFU/mL were determined and expressed as Log CFU/mL.
L. casei DK211 was selected as the fermentation powder depending on the whey protein’s proteolytic activity and tolerance to bile salts and acids. The whey protein fluid was fermented using L. casei DK211 with 7% whey protein concentrate (WPC-80) and 3% glucose as substrates. The substrates were dissolved in 90% water and pasteurized at 60°C for 30 min. This sterilized liquid was inoculated with the strain, DK211 (5×1010 CFU/mL) 0.01% (v/v) for fermentation at 38°C–40°C for 30 h. Fermentation was terminated when the pH and acidity reached 4.3 and 1.0, respectively. The finished product, that is cultured whey protein fluid was dried with dextrin and rice powder in a freeze dryer (FVD-1) under the following conditions: Degree of vacuum, 0.16 torr; and ending at 50°C of food temperature. Finally, the FWP supplement powder was obtained for clinical trials (Fig. 1).

Free amino acid quantification of the FWP was performed with an HPLC (Agilent LC system, Agilent, Santa Clara, CA, USA) fitted with a PDA detector. To quantify the free amino acids in the FWP sample, it was mixed with water and directly derived using O-phthaldialdehyde. Sample separations were then determined using 5 μm Capcellpak UG120 C18 column (4.6×250 mm). The derivative samples were eluted using a 40 mM NaH2PO4 (pH 7.8) as solvent A and acetonitrile: methanol: water (45:45:10, v/v/v) as solvent B with a total time of 38 min, at 40°C, and a flow rate maintained at 1.5 mL/min. Each standard amino acid solution (Agilent LC system, Agilent) was used as an external standard (Lee et al., 2018).
The molecular weight distribution profile was determined by gel permeation chromatography on a TSK gel G2000 SWXL 7.8×300 mm column (Tosoh, Tokyo, Japan) using an Agilent 1100 HPLC system (Agilent LC system, Agilent) in accordance with the method by Zhuang et al. (2009). HPLC was subsequently carried out in the mobile phase (acetonitrile: water: trifluoroacetic acid=20:80:0.1, v/v) at a flow rate of 0.5 mL/min and monitored at 220 nm at 30°C. A molecular weight calibration curve was then prepared using the average elution volume of the following standards: Tripeptide GGG (Mr 189), tetrapeptide GGYR (Mr 451), bacitracin (Mr 1450), aprotinin (Mr 6500), and cytochrome C (Mr 12500; Sigma-Aldrich, St. Louis, MO, USA; Liu et al., 2017).
From July 2022 to December 2022, healthy adult men aged 30–60 years were recruited for this study being conducted at Dankook University Hospital, Chungcheongnam-do, Korea. All subjects provided their informed consent for inclusion before participating in the study. The study was conducted in accordance with the declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Institutional Review Boards (IRBs) of Dankook University Hospital in August 2022 (No. DKUH 2022-07-016-002).
The inclusion criteria for this study were an age over 30 and under 60 years, understanding of the purpose of this study, voluntary agreement to participate in the study, and a body mass index (BMI) of 18.5 to 30 kg/m2 for the men as measured by bioelectrical impedance analysis (BIA; Inbody, Seoul, Korea). Those who took functional foods or protein supplements, were involved in regular exercise, took subscribed medicines, or had orthopedic problems and medical illnesses were excluded.
This was a double-blind randomized controlled trial performed over 8-weeks with two parallel groups. Participants were asked to maintain their usual diet and physical activity other than the designated exercise program conducted the during 8-week intervention. All participants were provided a total of 63 packs (37 g/pack) containing fermented whey protein supplementation [FWPS; protein 54.06% (FWP powder 97.3%, flavor 2.70%)] or whey protein concentration supplementation [WPCS; protein 54.06% (WPC 67.57%, dextrin 29.73%, flavor powder 2.70%); SFC Bio, Seoul, Korea; Table 1]. The two protein supplements were similar in taste and appearance and participants were instructed to consume the supplementation twice a day for 8-weeks.
The exercise program for this study was conducted three times a week for a total of 8-weeks using a three-division exercise method. Exercise intensity was performed using 70%–80% repetition maximum. During the 4th week of the exercise program, exercise intensity was reset according to individual performance. There were 4–6 sports events, 4–5 sets per event, 60–90 s break between sets, with 25 sets in total (Fig. 2).
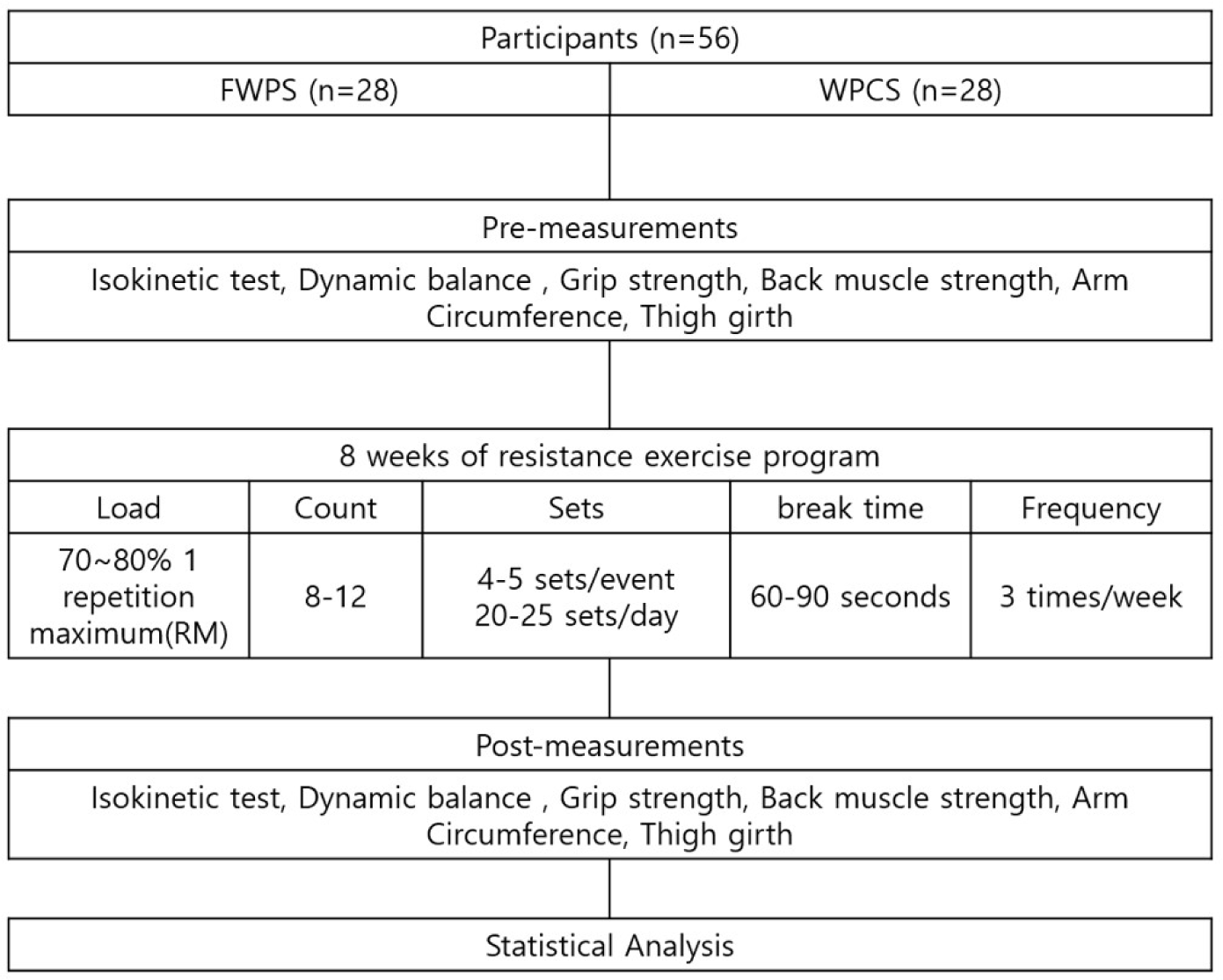
Skeletal muscle mass (SMM) was calculated as the sum of muscle mass in the arms and legs. There were two types of skeletal muscle mass index (SMI): Appendicular skeletal muscle mass (ASM) adjusted for height [ASM (kg)/Height (m2)], BMI (LBM/BMI)=LBM (kg)/BMI (kg/m2) was assessed using dual-energy X-ray absorptiometry (DXA) (horizon-wi, Hologic, Marlborough, MA, USA) after at least 8 h of fasting. SSM adjusted for height [SSM (kg)/Height (m2)] was assessed using BIA.
Next, the secondary outcome measurements were taken including isokinetic measurements which were performed on the left and right knees, five times at 90°/sec and 20 times at 180°/sec using Biodex pro 4 (Biodex, Shirley, NY, USA). Grip strength was measured twice each on the left and right sides at 3 min intervals, using a digital grip strength meter (TKK-5401 grip strength meter, TAKEI, Yokohama, Japan). Back muscle strength was measured twice in total using a digital back muscle strength measuring instrument (TKK-5402 back muscle strength measuring instrument, TAKEI). The left and right sides were measured twice for the dynamic balance ability test using the Y-balance test kit (Functional Movement System: FMS, Dallas, TX, USA). Vertical jump was measured twice at 3-minute intervals, using a digital jump meter (TKK-5046 vertical jump meter, TAKEI).
Each participant’s blood was collected after at least 8 h of fasting, both before and after the intervention. Blood samples were drawn by a registered phlebotomist with complete blood cell count (CBC), albumin, blood sugar, alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, total bilirubin, and blood urea nitrogen (BUN) being measured in the hospital laboratory.
Data were analyzed using the R software (version 4.2.1, 2017; R Core Team). To check if there was a difference in baseline between the two groups, an independent t-test was performed for the continuous variable and a chi-square test for the categorical variable. Changes between baseline and after 8-weeks were analyzed using a paired t-test. General characteristics of the subjects were analyzed using descriptive statistics and were expressed as n (%). Additionally, only data from participants who completed all follow-up studies (n=56) were analyzed (per protocol analysis, PP). The p-value<0.05 were considered statistically significant.
Results
After isolating LAB from kimchi, strain identification was performed using 16s rRNA sequencing (Table 2). DK211 was identified as L. casei (99% similarity to GeneBank sequence), DK301 was identified as Lactobacillus sakei, and DK310 was identified as Lactobacillus paracasei (100% similarity with GeneBank sequence).
The proteolytic activity of LAB was evaluated according to the degree of formation of clear zones generated in skim milk agar (Hayes et al., 2006; Reddi et al., 2016). The diameter of the transparent ring observed around the well is a relative indicator of proteolytic activity. Among the three strains used in the experiment, L. casei DK211 showed higher proteolytic activity compared to that of the commercial strain (LA-5, Lactobacillus acidophilus; NU-TRISH®, Chr. Hansen, Hørsholm, Denmark; Table 3).
LAB was inoculated into a protein solution for hydrolysis. The DH is indicated in Fig. 3. Hydrolysis of proteins by LAB increased the number of peptides and total amount of free amino groups. All three LABs showed a rapid increase in the DH from 0–4 h.
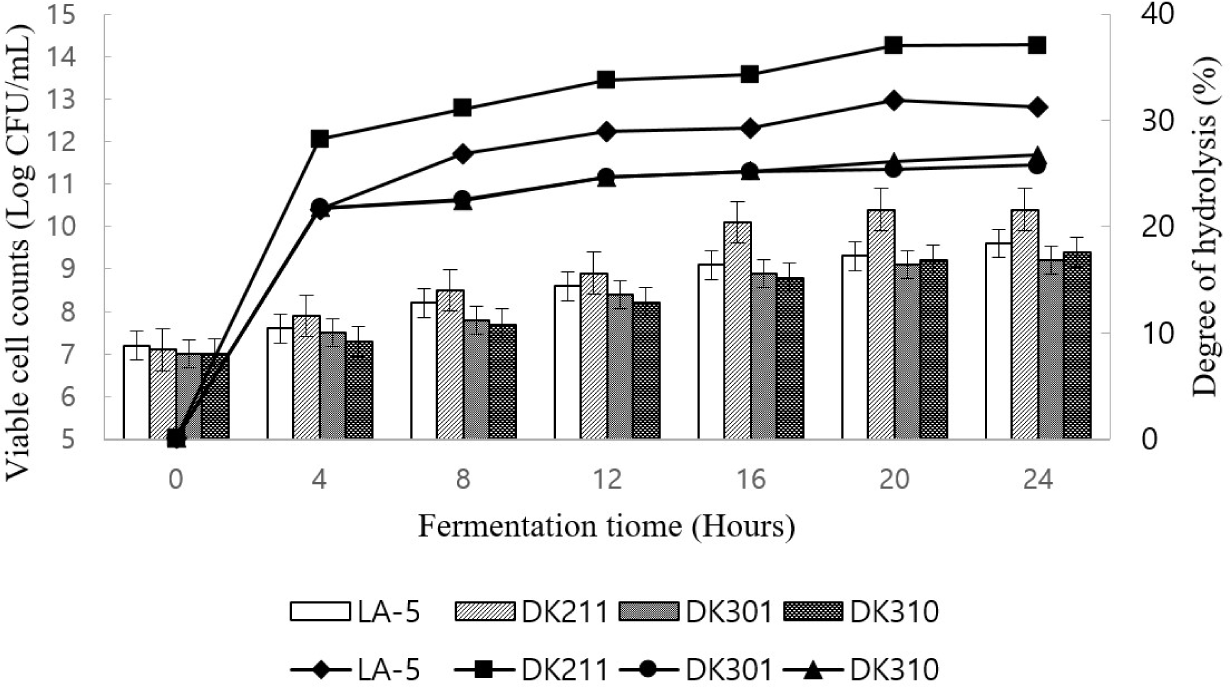
The highest DH occurred after 20 h. The highest DH was confirmed in the WPC solution fermented with the DK211 strain compared to that of the commercial strain (LA-5).
The degree of protein degradation was represented as a curve over time. The results indicated that the LAB strain exhibited proteolytic activity by breaking down proteins.
Viable cell counts are shown in Fig. 3. There were significant differences in the viable counts among the samples after fermentation. The initial viable cell counts ranged from 7.12 Log CFU/mL and after 20 h of incubation, that of DK211 were the highest (10.4 Log CFU/mL) compared to other cultures.
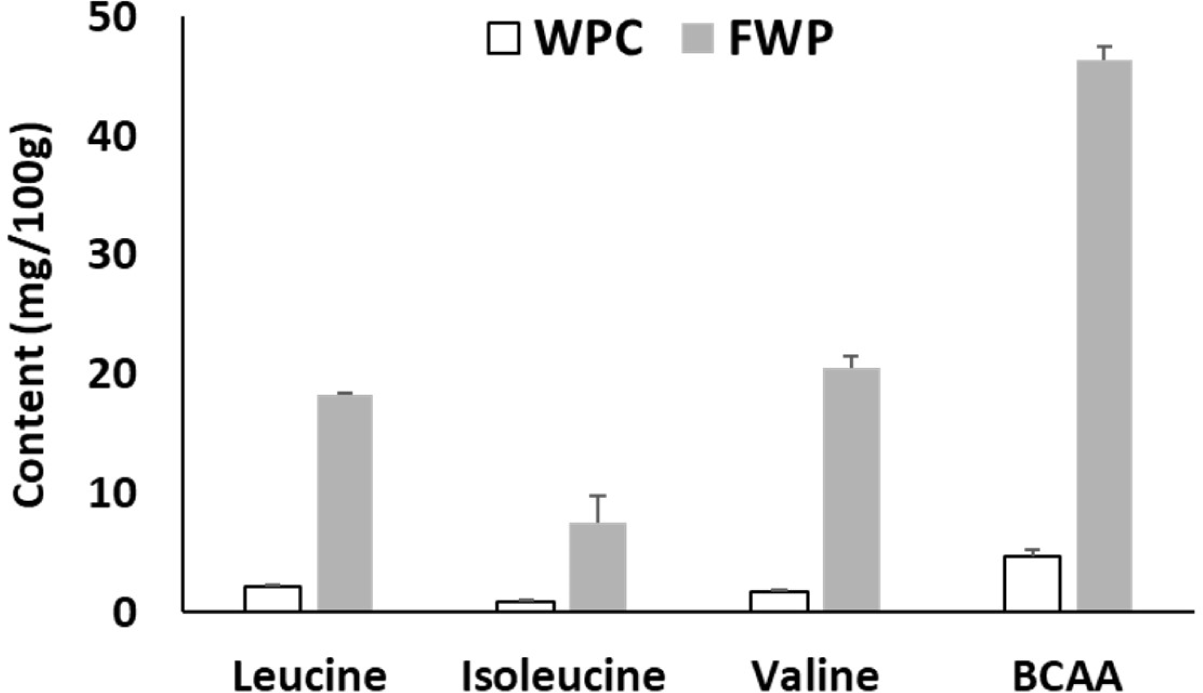
Changes in free amino acid content are shown in Table 4. Alanine, proline, histidine, leucine, methionine, valine, glutamic acid, and asparagine all showed increases of more than 500%, with the total free amino acid content increasing from 21.9 mg/100 g to 182.69 mg/100 g.
Leucine, valine, and isoleucine are branched-chain amino acids (BCAAs). FWP showed a significant increase in BCAA content compared to that in WPC (Fig. 4).
Protein contents of WPC and FWP were 94% and 70.41%, respectively, in the molecular weight range of 5,000 or higher (Table 5).
Data for body composition, muscle mass, blood tests, and CBC are shown in Table 6. No significant differences in baseline characteristics between the FWP and WPC groups were observed (Table 7).
The FWPS and WPCS supplementation resulted in a substantial increase in muscle strength for each group after 8-weeks of supplementation, although there was no significant difference between the two groups (Table 8; Fig. 5).
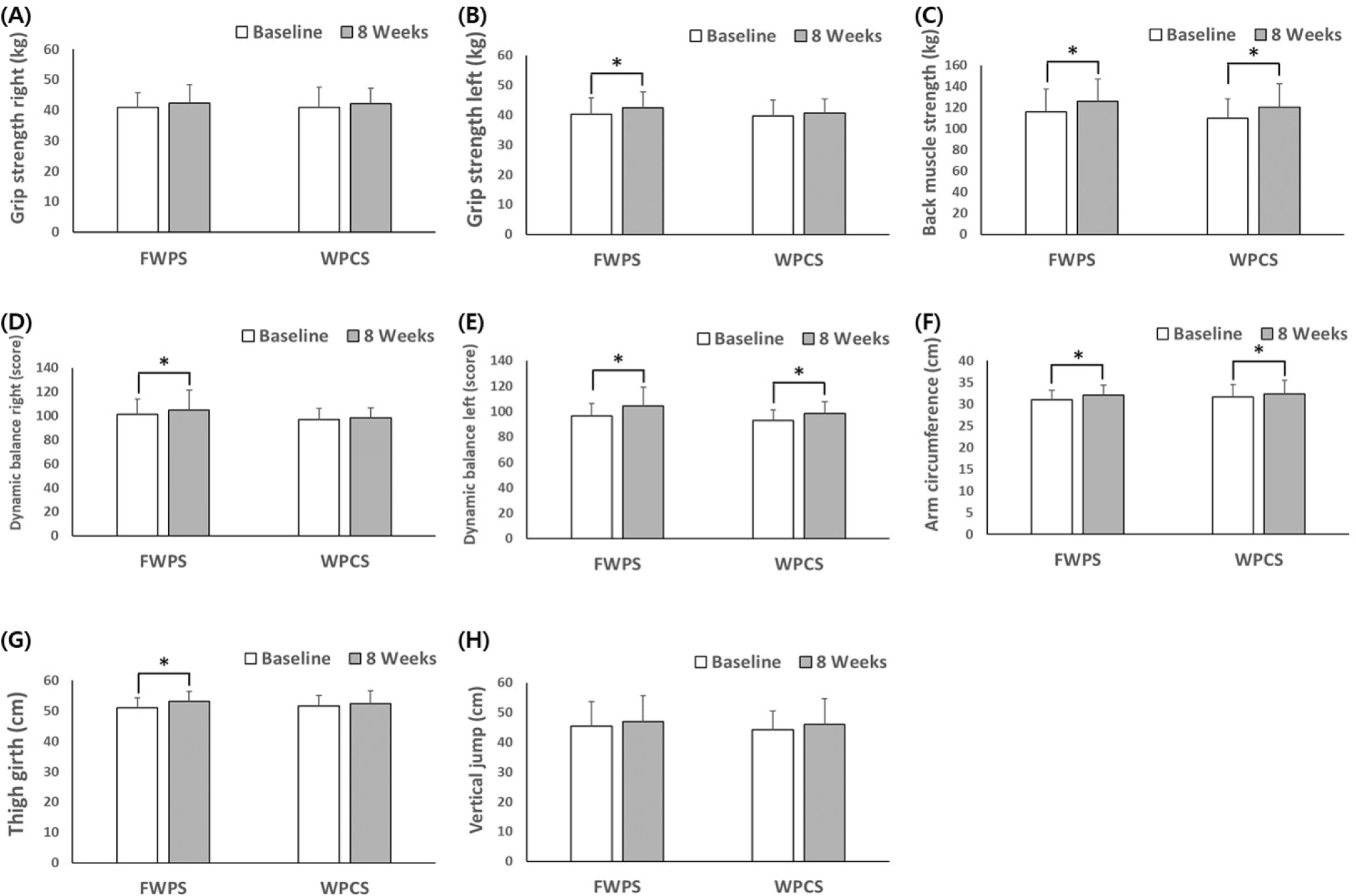
The significant effect of fermented whey supplementation was observed in the dynamic balance of both the right and left sides (p=0.032 respectively). In addition, statistical significance was observed in grip strength (left), upper arm circumference, and flat leg circumference before and after 8-weeks of FWPS intake. Conversely, no statistically significant differences for these parameters were observed in the WPCS group.
Discussion
In this study, several LAB isolated form homemade kimchi were identified based on their 16S-rRNA sequence.
The comparative proteolytic activities and DH of commercial strains (LA-5) and LAB were measured and the best performing strain [L. casei (DK211)] was selected.
Fermented whey products were manufactured using this L. casei DK211 strain adapting freeze drying. After fermentation with this strain, it was found that the total concentration of BCAAs, known as a group of amino acids, most beneficial for muscle biosynthesis, and other amino acids such as alanine, proline, histidine, methionine, glutamic acid, and asparagine were increased by up to 500% compared to that of the non-fermented control. The overall free amino acid contents increased from 21.9 mg/100 g to 182.69 mg/100 g, indicating the efficacy of L. casei DK211 fermentation. The BCAAs leucine, isoleucine, and valine belong to the nine essential amino acids in humans, accounting for upto 35% of the total essential amino acids in muscle proteins and 40% of the preformed amino acids in the whole body required by mammals (Harper et al., 1984).
Whey protein is a rich source of BCAAs and is considered to have beneficial effects on skeletal muscle turnover (McGregor and Poppitt, 2013). ter Borg et al. (2019) reported that lower blood levels of essential amino acids (EAA), particularly BCAAs were found to be associated with a lower skeletal muscle index. BCAA-rich supplementation significantly increased muscle mass, muscle strength, and physical performance in elderly individuals with sarcopenia compared with a control diet (Bai et al., 2021). In this study, fermenting whey protein with LAB resulted in further increase of BCAAs, thus indicating the expected positive effects on skeletal muscle with FWPs.
Fermented proteins are more easily digested and better utilized by the body from the changed protein composition that results in desirable proportions or smaller molecular weight components (Yarlina et al., 2020). According to several previous reports, LAB can be a strong candidate for an effective fermentation process, since they are capable of producing various enzymes (Pescuma et al., 2010). Whey fermentation using LAB can improve protein and lactose digestibility, simultaneously contributing to the flavor and texture of the end product (Pescuma et al., 2010).
When analyzing amino acids and molecular weights, the changes in amino acid composition and protein molecular weight distribution confirmed that protein degradation occurred by fermentation. Given that fermentation increases BCAAs, it is not only considered as a more digestible method, but also an effective product manufacturing method to promote muscle health. In this study the subjects were not elderly, however, the average age was 44.8 years, representing the importance of maintaining muscle mass and performance later in life. Starting as early as the 4th decade of life, skeletal muscle mass undergoes a natural decline at a rate of approximately 0.8% per year, in a process termed sarcopenia (Paddon-Jones and Rasmussen, 2009). Many studies on protein supplementation interventions have focused on the elderly aged over 65 years, whilst research on pre-elderly individual remains rare despite its significance (Pennings et al., 2011; Visser et al., 2005). Certain intervention studies have reported that protein supplementation alone or in combination with resistant exercise training improved lean muscle mass and/or physical performance, whilst other studies have found no significant effects (Labata-Lezaun et al., 2020). Specifically, in cases of whey protein supplementation, most studies found a promotional effect on muscle building, simultaneously claiming that the underlying mechanisms could be attributed to the enrichment of BCAAs. BCAA supplementation has already shown beneficial effects for decreasing exercise-induced muscle damage and promotion of muscle protein synthesis (Shimomura et al., 2004).
In this study, BCAAs of FWPs were found to be higher compared to those of control non-FWPs. Therefore, it is reasonable to assume that a higher BCAAs content may have contributed to improved muscle performance, although the exact plasma leucine concentrations of the subjects were not determined. According to the present 8-week intervention, a significant effect of fermented whey supplementation was observed in the dynamic balance of both the right and left sides (p=0.032 respectively). In addition, statistical significance was observed in the grip strength (left), upper arm circumference, and flat leg circumference before and after 8-weeks of FWPS intake, although no statistical significance was found for these parameters in the control WPCS group. When FWP supplements were consumed twice daily for 8-weeks alongside resistance exercise training, dynamic balance improved, as measured by the Y-balance test. This YBT, which requires muscle strength, flexibility, and proprioception, is particularly related to lower extremity musculoskeletal injury (Butler et al., 2016). It has also been reported that dynamic balance and core strength are positively correlated, thus suggesting their significance in overall coordination of muscle performance (Almutairi et al., 2022). A decrease in YBT has been associated with a decrease in muscle function implicated in dynamic balance control, often resulting in an increased risk of falling due to aging (Lang et al., 2010). There have also been several other reports emphasizing the importance of YBT. For example, Cattaneo et al. (2007) and Sosnoff et al. (2014) showed that exercises improving dynamic balance would reduce the risk for falling among the elderly. Another preliminary study has found that a decrease in dynamic balance ability applying YBT distance was accompanied by a decline in muscle strength later in life. All of these reports suggested that YBT is a dependable indicator that can be used for reducing risk of falling and muscle strength improvement. Furthermore, in several studies, dynamic balance was reported to have a correlation with sarcopenia (Lang et al., 2010; Lin et al., 2022; Yu et al., 2020). The present study showed that FWPS effectively promoted muscle performance and improved dynamic balance. These data also imply that dietary intervention among the elderly could improve muscle strength, reduce risk of falling and dealing with sarcopenia. In this study the increment of muscle health as shown with grip strength improvement was observed in the fermented group (Lang et al., 2010; Lee et al., 2015; Lin et al., 2022; Yu et al., 2020). In several previous studies, the strong connection of daily protein intakes and grip strength have illustrated the background preventive role of proteins in maintaining muscle health. No significant differences observed in this study between FWPS and WPCS after 8-week trials in muscle health criteria such as upper arm circumference and flat leg circumference, including left side grip strength. However, it is noteworthy that grip strength (left side), upper arm circumference, and flat leg circumference of the fermented group increased significantly from the baseline in the FWPS, while this was not observed with control non-fermented group. The exact performance of lactic acid fermented products with resistance exercise requires further investigation and the specific features of enhancing recovery from heavy resistance exercise training should be examined in depth.
This study investigated the protein degradation, changes in amino acid composition, molecular weight distribution of fermented proteins, and increment of BCAAs important components of muscle building utilizing whey. It also investigated the effects of FWPS for 8-weeks on muscle parameters and physical performance in healthy middle-aged male adults consuming non-FWP products. It was concluded that FWPS was effective in improving muscle strength and performance considering the results of better outcomes for handgrip, and right and left dynamic balance. There have been no prior studies that investigated the effects of lactic acid FWPS on promoting health by affecting muscle performance. The development of lactic acid fermented products can be regarded as one of the most significant areas of food development based on their health benefits. Considering that the market for proteins has been growing in recent times, it is expected to attract significant attention as a new type of protein source.
To summarize, it was found that a specific strain of kimchi LAB, namely L. casei DK211, was most effective for fermenting whey proteins. After providing fermented whey products to healthy male subjects for 8-weeks, it was found that fermented whey was more effective in muscle mass building and performance in maintaining dynamic balance.
Conclusion
In this study, FWPS was shown to be effective in promoting physical performance in terms of dynamic balance measurement and muscle health, as indicated by the increment of grip strength (left), upper arm circumference, and flat leg circumference from the base line. These results suggested that the whey protein bioconverted by L. casei DK211 was the most effective protein supplement for enhancing muscle health in adults undergoing regular resistance exercise.













