ARTICLE
Mathematical Model for Predicting the Growth Probability of Staphylococcus aureus in Combinations of NaCl and NaNO2 under Aerobic or Evacuated Storage Conditions
Jeeyeon Lee1,2, Eunji Gwak1, Jimyeong Ha1,2, Sejeong Kim1,2, Soomin Lee1,2, Heeyoung Lee1,2, Mi-Hwa Oh3, Beom-Young Park3, Nam Su Oh4, Kyoung-Hee Choi5, Yohan Yoon1,2,*
Author Information & Copyright ▼
1Department of Food and Nutrition, Sookmyung Women's University, Seoul 04310, Korea
2Risk Analysis Research Center, Sookmyung Women's University, Seoul 04310, Korea
3National Institute of Animal Science, RDA, Wanju 55365, Korea
4R&D Center, Seoul Dairy Cooperative, Ansan 15407, Korea
5Department of Oral Microbiology, College of Dentistry, Wonkwang University, Iksan 54538, Korea
*Corresponding author: Yohan Yoon, Department of Food and Nutrition, Sookmyung Women’s University, Seoul 04310, Korea. Tel: +82-2-2077-7585, Fax: +82-2-710-9479, E-mail:
yyoon@sookmyung.ac.kr
Copyright © 2016, Korean Society for Food Science of Animal Resources. This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: Aug 09, 2016 ; Revised: Oct 28, 2016 ; Accepted: Nov 05, 2016
Published Online: Dec 31, 2016
Abstract
The objective of this study was to describe the growth patterns of Staphylococcus aureus in combinations of NaCl and NaNO2, using a probabilistic model. A mixture of S. aureus strains (NCCP10826, ATCC13565, ATCC14458, ATCC23235, and ATCC27664) was inoculated into nutrient broth plus NaCl (0, 0.25, 0.5, 0.75, 1, 1.25, 1.5, and 1.75%) and NaNO2 (0, 15, 30, 45, 60, 75, 90, 105, and 120 ppm). The samples were then incubated at 4, 7, 10, 12 and 15℃ for up to 60 d under aerobic or vacuum conditions. Growth responses [growth (1) or no growth (0)] were then determined every 24 h by turbidity, and analyzed to select significant parameters (p<0.05) by a stepwise selection method, resulting in a probabilistic model. The developed models were then validated with observed growth responses. S. aureus growth was observed only under aerobic storage at 10-15℃. At 10-15℃, NaCl and NaNO2 did not inhibit S. aureus growth at less than 1.25% NaCl. Concentration dependency was observed for NaCl at more than 1.25%, but not for NaNO2. The concordance percentage between observed and predicted growth data was approximately 93.86%. This result indicates that S. aureus growth can be inhibited in vacuum packaging and even aerobic storage below 10℃. Furthermore, NaNO2 does not effectively inhibit S. aureus growth.
Keywords: predictive model; S. aureus; NaCl; NaNO2; processed meat products
Introduction
Consumers consider many factors when purchasing certain foods, and their awareness of food safety is becoming more important because volumes of imported and processed foods are increasing (Choe et al., 2005; Kim and Kim, 2003). Some foods contain additives, which are included to improve the quality of the product. Such additives include NaCl and NaNO2, which play a role in preservation and food safety, especially in processed meat products (Pereira et al., 2015; Shapiro et al., 2016). However, recently, consumers have started to express a preference for processed meat products formulated with low concentrations of NaCl and NaNO2, because of the health issues involved (Bedale et al.,2016; Guàrdia et al., 2006; Kim et al., 2012).
The processed meat industry has tried to use substitutes for additives, especially NaNO2, and consumers are satisfied with the appearance of the products (Lee et al., 2015a). In processed meats, NaNO2 plays a role in color fixing and inhibiting pathogenic bacteria, such as Listeria monocytogenes, Clostridium botulinum, and Staphylococcus aureus (Hospital et al., 2016; Karina et al., 2011; Latham et al., 2016; Tompkin et al., 1973). Although NaNO2 substitutes may fix the color in processed meat products, most have no antimicrobial activity. Thus, using a NaNO2 substitute or a low concentration of NaNO2 may result in greater pathogenic bacterial growth than in conventional meat products.
S. aureus is a gram-positive enterotoxigenic bacterium (CDC, 2014). Twenty to thirty percent of people are carriers of S. aureus (Normanno et al., 2007), and it can contaminate food during processing; the pathogen may produce enterotoxins at 105-106 CFU/g of S. aureus (Chiefari et al., 2015; Park et al., 1992). Ham can be contaminated with S. aureus during slaughter, processing, or handling (Borch et al., 1996; Ingham et al., 2004). Park et al. (2012) reported that they had isolated S. aureus from 0.6% of the ham samples they examined, and Atanassova et al. (2001) isolated the pathogen from 35.6% of smoked ham.
Therefore, we developed mathematical models to predict the growth probability of S. aureus in a combination of NaCl and low-concentration NaNO2 under both aerobic and vacuum storage conditions.
Materials and Methods
Inoculum preparation
Five S. aureus strains (NCCP10826, ATCC13565, ATCC14458, ATCC23235 and ATCC27664) were cultured in 10 mL nutrient broth (NB; Becton, Dickinson and Company, USA) at 35℃ for 24 h. The one-tenth milliliter aliquots of the cultures were subcultured in 10 mL fresh NB at 35℃ for 24 h. The subcultures were then centrifuged at 1,912 g for15 min at 4℃, and washed twice with phosphate-buffered saline (PBS, pH 7.4; 0.2 g of KH2- PO4, 1.5 g of Na2HPO4·7H2O, 8.0 g of NaCl, and 0.2 g of KCl in 1 L of distilled water). Each cell suspension of the S. aureus strains was mixed, and the mixture was serially diluted with PBS to obtain 4 Log CFU/mL.
Sample preparation and inoculation
NB was formulated with NaCl (0, 0.25, 0.5, 0.75, 1, 1.25, 1.5, and 1.75%) and NaNO2 (0, 15, 30, 45, 60, 75, 90, 105, and 120 ppm). Two hundred five microliter of the samples were placed into each well of a 96-well microtiter plate (SPL Life Sciences Co., Ltd., Korea), and 25-μL portions of S. aureus inoculum were inoculated into the samples. The microtiter plates were sealed with paraffin film (Parafilm M®; Bemis Company Inc., USA) for aerobic storage, or placed in airtight containers with AnaeroGen packs (Oxoid Ltd., UK) for vacuum storage. The AnaeroGen packs were replaced every 24 h. The microtiter plates were stored at 4-15℃ for up to 60 d, depending on storage temperature, under aerobic or vacuum conditions. We used plain NB and NB plus S. aureus cells for the negative and positive controls, respectively.
Probabilistic model development
During storage, the growth responses for each combination (n=4) were determined by turbidity every 24 h. If a combination was turbid, it was designated as “growth (score=1)”, otherwise it was designated as “no growth (score=0)”. The growth response data were analyzed by logistic regression as follows:
where P is the probability of growth, ai are estimates, NaCl is the NaCl concentration, NaNO2 is the NaNO2 concentration, Time is the storage time and Temp is the storage temperature. Among the parameters, NaNO2 and storage Time were transformed for proper application to the model. In the equation, the significance parameters (p<0.05) were selected by a stepwise selection method using SAS® (Version 9.3; SAS Institute Inc., USA). In addition, the estimates of selected parameters were used to produce growth/no growth interfaces at 0.1, 0.5 and 0.9 probability.
Minimum bactericidal concentration (MBC) test
To determine the MBC of NaCl and NaNO2 for S. aureus, the aqueous portions of the microtiter plate wells that were clear were streaked on mannitol sorbitol agar (MSA; Becton, Dickinson and Company), and the plates were incubated at 37℃ for 24 h to observe S. aureus survival by colony.
Validation of model performance with emulsion type sausage
To evaluate the performance of the developed probabilistic model, the predicted growth response from the model was compared with the observed growth response from real food. To prepare observed growth response data, emulsion-type sausages were manufactured according to the formulation given in Table 1. Each batch of the formula was mixed for 6 min using a cutter (MSK 760-II; Mado, Germany), and stored at 4℃ for 1 h. The mixtures were then filled into collagen casings (30 g per casing) using a Konti A50 automatic sausage can filler (Frey, Germany). The resulting sausages were then smoked at 75℃ for 40 min in a smokehouse (MAXI 3501; Kerres, Germany) and chilled. The vacuum-packaged smoked sausages were heated at 80℃ for 15 min and stored at 4℃ until required. The sausages (25 g) were placed in a sterilized plastic container containing S. aureus inoculum at 3 Log CFU/mL, and gently stirred for 2 min to complete inoculation. The samples were air-dried for 15 min to allow S. aureus cell attachment, and transferred to sample bags. The bags were sealed for the aerobic or vacuum-packaged experiments and stored at 10℃ for 65-70 d and 15℃ for 35-43 d, respectively. During storage, S. aureus cell counts were enumerated on MSA (Becton, Dickinson and Company). If the S. aureus cell count increased by more than 1 Log CFU/g compared with that on day 0, the result was considered to be “growth”, otherwise “no growth” was recorded (Gwak et al., 2015; Koutsoumanis et al., 2004).
Table 1.
Formulation of emulsion-type sausages
| Ingredients (%) |
No NaNO2 |
10 ppm NaNO2 |
|
| 1.00% NaCl |
1.25% NaCl |
1.50% NaCl |
1.00% NaCl |
1.25% NaCl |
1.50% NaCl |
| Pork meat |
60 |
60 |
60 |
60 |
60 |
60 |
| Pork fat |
20 |
20 |
20 |
20 |
20 |
20 |
| Ice |
20 |
20 |
20 |
20 |
20 |
20 |
| Total |
100 |
100 |
100 |
100 |
100 |
100 |
| NaCl |
1.00 |
1.25 |
1.50 |
1.00 |
1.25 |
1.50 |
| NaNO2 |
- |
- |
- |
0.0029 |
0.00303 |
0.00305 |
| Phosphate |
0.03 |
0.03 |
0.03 |
0.03 |
0.03 |
0.03 |
| Isolated soy protein |
1.00 |
1.00 |
1.00 |
1.00 |
1.00 |
1.00 |
| Mixed spice |
0.50 |
0.50 |
0.50 |
0.50 |
0.50 |
0.50 |
| Sugar |
0.50 |
0.50 |
0.50 |
0.50 |
0.50 |
0.50 |
| Potassium sorbate |
0.20 |
0.20 |
0.20 |
0.20 |
0.20 |
0.20 |
Download Excel Table
Results and Discussion
In vacuum condition, S. aureus growth was not observed at any growth temperature up to 60 d, regardless of the NaCl and NaNO2 concentrations, indicating that S. aureus can be inhibited effectively in vacuum packaging, even at low concentrations of NaCl and NaNO2, and therefore, no probabilistic model was developed (data not shown). Under aerobic conditions, S. aureus growth was not observed below 10℃ up to 60 d, regardless of the NaCl and NaNO2 concentrations, but the MBC test showed that S. aureus cells were not completely destroyed. Their cell counts were just reduced or retained at all concentrations of NaCl and NaNO2 examined in this study. This result indicates that NaCl concentrations up to 1.75% and NaNO2 concentrations up to 120 ppm, and their combinations, may not be sufficient to destroy S. aureus at low temperatures, and S. aureus cells that survive below 10℃ may grow above 10℃, allowing S. aureus to produce enterotoxins. In agreement with this result, Lee et al. (2015b) reported that the Tmin (theoretical minimum growth temperature) value for S. aureus was 10.2℃ in cheese. However, Lee et al. (2013) and Le Marc et al. (2009) reported lower Tmin values for carbonara sauce (5.2℃) and milk (5.8℃). These results indicate that the Tmin values for S. aureus depend on the food matrix. The low temperature adaptation of S. aureus is related to the lipoamide dehydrogenase gene (lpd) in the bkd gene cluster, which causes the production of branched-chain fatty acids in phospholipids, resulting in improved membrane fluidity (Singh et al., 2008; Yoon et al., 2015).
S. aureus growth was observed at 10, 12 and 15℃, and the probability model was developed to describe the growth pattern using logistic regression. Significant parameters affecting S. aureus growth are presented in Table 2, and the parameters with estimates were used to produce the growth/no growth interfaces at 0.1, 0.5 and 0.9 probabilities in Figs. 1 and 2. The results in Table 2 show that S. aureus growth was affected (p<0.0001) by storage temperature, storage time, and the concentrations of NaCl and NaNO2, but no interaction effects, including for NaCl×NaNO2, were observed.
Table 2.
Estimates of the parameters selected from the logistic regression analysis by a stepwise selection method to produce the interfaces between growth and no growth of Staphylococcus aureus at desired probabilities under aerobic conditions
| Variables |
Estimate |
SE |
p-value |
| Interception |
−38.2620 |
0.2078 |
<0.0001 |
| Temperature |
0.7171 |
0.0038 |
<0.0001 |
| NaNO2 concentration/10 |
0.0951 |
0.0021 |
<0.0001 |
| NaCl concentration |
−0.9255 |
0.0148 |
<0.0001 |
| Log (Time) |
10.2063 |
0.0599 |
<0.0001 |
Download Excel Table
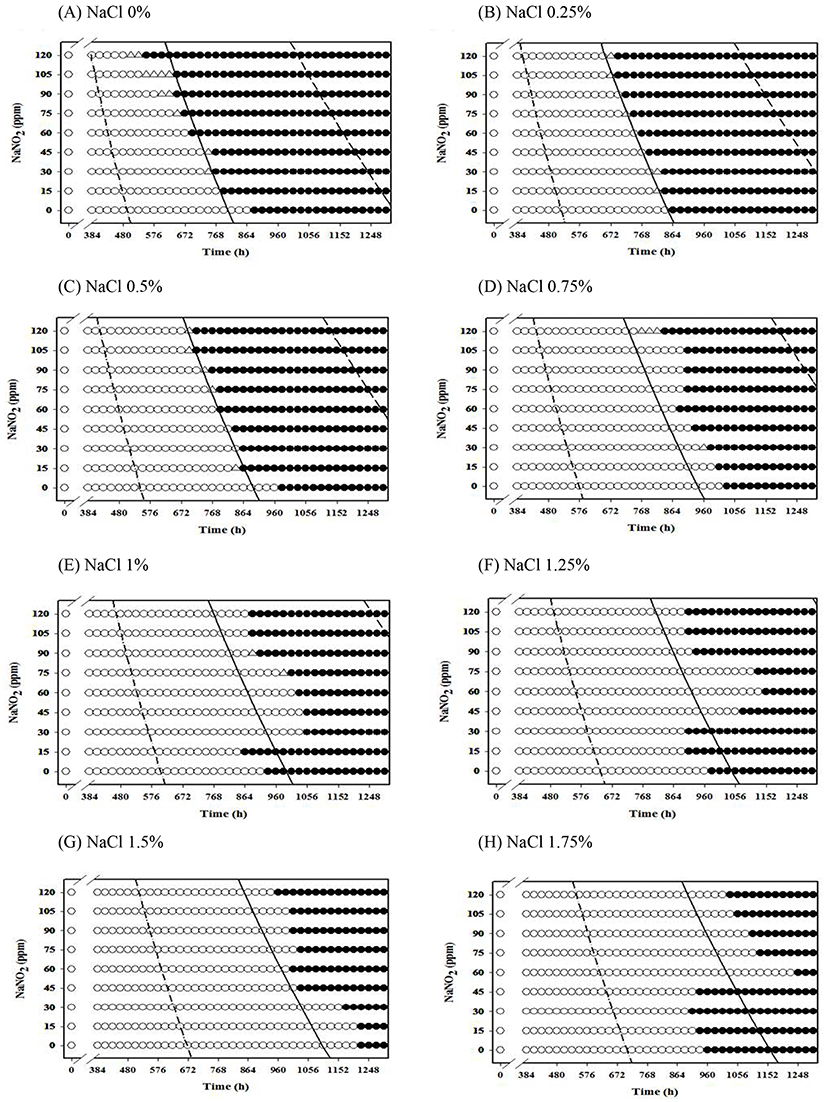
Fig. 1.
Growth/no growth interfaces of Staphylococcus aureus in nutrient broth at 10℃ with respect to NaNO2 concentration and storage time for various NaCl concentrations under aerobic conditions at growth probabilities of 0.1 (left line), 0.5 (middle line) and 0.9 (right line); no growth: ○, growth: ●, 50% growth: △.
Download Original Figure
Fig. 2.
Growth/no growth interfaces of Staphylococcus aureus in nutrient broth at 15℃ with respect to NaNO2 concentration and storage time for various NaCl concentrations under aerobic conditions at growth probabilities of 0.1 (left line), 0.5 (middle line) and 0.9 (right line); no growth: ○, growth: ●, 50% growth: △.
Download Original Figure
At 10℃, NaCl and NaNO2 did not inhibit the growth of S. aureus, as well as combination of NaCl and NaNO2 at less than 1.25% NaCl. However, interestingly, the initiation time for S. aureus growth decreased as NaNO2 concentration increased at less than 1.25% NaCl (Fig. 1). Schlag et al. (2008) reported that the nreABC gene is involved in nitrate reduction. Therefore, the antibacterial effect of NaNO2 on S. aureus was not detected.
A NaCl concentration of more than 1.25% inhibited S. aureus growth (Fig. 1). In addition, no difference in initiation time for S. aureus growth was observed among the various NaNO2 concentrations, and the initiation times were longer than those at less than 1.25% NaCl. Even at more than 1.25% NaCl, the combination effect was not observed (Fig. 1), as shown in Table 2, and the S. aureus growth response at 0 ppm NaNO2 was similar to that at 120 ppm (Fig. 1). This result indicates that a NaCl concentration of more than 1.25% is needed to inhibit S. aureus growth, but NaNO2 is not effective in inhibiting S. aureus growth. However, Lee et al. (2015c) reported a NaCl and NaNO2 combination effect on Lactobacillus in frankfurters, and Jo et al. (2014) reported a combination effect on Pseudomonas spp. in processed meats. These results suggest that the NaCl and NaNO2 combination effect depends on the type of foodborne bacteria. S. aureus grew better at 12℃ than at 10℃, and demonstrated a NaCl concentration-dependent growth response (Fig. 2). In addition, no obvious effect of NaNO2 on the inhibition S. aureus growth was observed (Fig. 2), which was similar to the result at 15℃ (data not shown). In agreement with these observations, a study by Bang et al. (2008) also showed that nitrite had no effect on inhibiting S. aureus growth.
To evaluate the performance of the developed probabilistic models in this study, observed growth responses were collected from real food (emulsion-type sausages) in an additional study, and the observed growth responses from the study were compared with the predicted growth responses from developed probabilistic models. Because the predictions from the developed probabilistic models were expressed as numbers, growth was determined at more than 0.5 of growth probability (Yoon et al., 2012). In addition, growth responses (growth or no growth) from the sausages were determined at 1 Log CFU/g of S. aureus growth (Gwak et al., 2015; Koutsoumanis et al., 2004). Comparisons between predicted and observed growth response are presented in Table 3; the observed growth responses mostly agreed with the predicted growth responses. The accordance percentage between the predicted and observed growth responses was 93.86%, indicating that the developed probabilistic model was capable of predicting the growth responses of S. aureus in emulsion-type sausages, formulated with various concentrations of NaCl and NaNO2.
Table 3.
Comparisons between observed and predicted growth responses of Staphylococcus aureus in emulsion-type sausage under aerobic conditions
| Temperature (℃) |
NaNO2 (ppm) |
NaCl (%) |
Time (h) |
Observed growth response |
Predicted growth response |
| 10 |
0 |
1.00 |
0-1,3201) |
NG |
NG |
| 1,440 |
NG |
G |
| 1,560 |
G |
G |
|
| 1.25 |
0-1,320 |
NG |
NG |
| 1,440 |
NG |
NG |
| 1,560 |
G |
G |
|
| 1.50 |
0-1,320 |
NG |
NG |
| 1,440 |
NG |
NG |
| 1,560 |
NG |
G |
|
| 10 |
1.00 |
0-1,320 |
NG |
NG |
| 1,440 |
NG |
G |
| 1,560 |
G |
G |
| 1,680 |
G |
G |
|
| 1.25 |
0-1,320 |
NG |
NG |
| 1,440 |
NG |
G |
| 1,560 |
G |
G |
| 1,680 |
G |
G |
|
| 1.50 |
0-1,320 |
NG |
NG |
| 1,440 |
NG |
NG |
| 1,560 |
G |
G |
| 1,680 |
G |
G |
|
| 15 |
0 |
1.00 |
0-5282) |
NG |
NG |
| 696 |
NG |
G |
| 864 |
G |
G |
| 1,032 |
G |
G |
|
| 1.25 |
0-528 |
NG |
NG |
| 696 |
NG |
G |
| 864 |
G |
G |
| 1,032 |
G |
G |
|
| 1.50 |
0-528 |
NG |
NG |
| 696 |
NG |
G |
| 864 |
G |
G |
| 1,032 |
G |
G |
|
| 10 |
1.00 |
0-4803) |
NG |
NG |
| 600 |
NG |
G |
| 720 |
G |
G |
| 840 |
G |
G |
|
| 1.25 |
0-480 |
NG |
NG |
| 600 |
NG |
NG |
| 720 |
G |
G |
| 840 |
G |
G |
|
| 1.50 |
0-480 |
NG |
NG |
| 600 |
NG |
NG |
| 720 |
G |
G |
| 840 |
G |
G |
Download Excel Table
In conclusion, the probabilistic models were appropriate for describing the growth responses of S. aureus at different concentrations of NaCl and NaNO2. Vacuum storage can inhibit S. aureus growth in emulsion-type sausages, and storage below 10℃ can inhibit S. aureus growth under aerobic storage conditions, even at low concentrations of NaCl and NaNO2. In storage above 10℃, a NaCl concentration of more than 1.25% is necessary to inhibit S. aureus growth effectively, but NaNO2 may not effectively inhibit S. aureus growth.
Acknowledgements
This work was carried out with support from the “Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ009237)” of the Rural Development Administration, Republic of Korea, and the Sookmyung Women’s University research grant for the Brain Korea 21 research team.
References
Atanassova V., Meindl A., Ring C. Prevalence of Staphylococcus aureus and staphylococcal enterotoxins in raw pork and uncooked smoked ham - a comparison of classical culturing detection and RFLP-PCR. Int. J. Food Microbiol. 2001; 68:105-113.


Bang W., Hanson D. J., Drake M. A. Effect of salt and sodium nitrite on growth and enterotoxin production of Staphylococcus aureus during the production of air-dried fresh pork sausage. J. Food Prot. 2008; 71:191-195.


Bedale W., Sindelar J. J., Milkowski A. L. Dietary nitrate and nitrite: Benefits, risks, and evolving perceptions. Meat Sci. 2016; 120:85-92.


Borch E., Nesbakken T., Christensen H. Hazard identification in swine slaughter with respect to foodborne bacteria. Int. J. Food Microbiol. 1996; 30:9-25.


Chiefari A. K., Perry M. J., Kelly-Cirino C., Egan C. T. Detection of Staphylococcus aureus enterotoxin production genes from patient samples using an automated extraction platform and multiplex real-time PCR. Mol. Cell. Prob. 2015; 29:461-467.


Choe J.-S., Chun H.-K., Hwang D.-Y., Nam H.-J. Consumer perceptions of food-related hazards and correlates of degree of concerns about food. J. Korean Soc. Food Sci. Nutr. 2005; 34:66-74.


Guàrdia M. D., Guerrero L., Gelabert J., Gou P., Arnau J. Consumer attitude towards sodium reduction in meat products and acceptability of fermented sausages with reduced sodium content. Meat Sci. 2006; 73:484-490.


Gwak E., Oh M.-H., Park B.-Y., Lee H., Lee S., Ha J., Lee J., Kim S., Choi K.-H., Yoon Y. Probabilistic models to predict Listeria monocytogenes growth at low concentrations of NaNO2 and NaCl in frankfurters. Korean J. Food Sci. An. 2015; 35:815-823.


Hospital X. F., Hierro E., Stringer S., Fernández M. A study on the toxigenesis by Clostridium botulinum in nitrate and nitrite-reduced dry fermented sausages. Int. J. Food Microbiol. 2016; 218:66-70.


Ingham S. V., Losinski J. A., Dropp B. K., Vivio L. L., Buege D. R. Evaluation of Staphylococcus aureus growth potential in ham during a slow-cooking process: use of predictions derived from the U.S. Department of Agriculture Pathogen Modeling Program 6.1 predictive model and an inoculation study. J. Food Prot. 2004; 67:1512-1516.


Jo H., Park B., Oh M., Gwak E., Lee H., Lee S., Yoon Y. Probabilistic models to predict the growth initiation time for Pseudomonas spp. in processed meats formulated with NaCl and NaNO2. Korean J. Food Sci. An. 2014; 34:736-741.


Karina P., Julio C., Leda G., Noemi Z. Behavior of Listeria monocytogenes type1 355/98 (85) in meat emulsions as affected by temperature, pH, water activity, fat and microbial preservatives. Food Control. 2011; 22:1573-1581.


Kim H.-C., Kim M.-R. Consumers' awareness and information-seeking behaviors towards food hygiene (2): Focused on foodborne illness. J. Korean Home Economics Assoc. 2003; 41:117-128.

Kim M. K., Lopetcharat K., Gerard P. D., Drake M. A. Consumer awareness of salt and sodium reduction and sodium labeling. J. Food Sci. 2012; 77:S307-S313.


Koutsoumanis K. P., Kendall P. A., Sofos J. N. Modeling the boundaries of growth of Salmonella Typhimurium in broth as a function of temperature, water activity, and pH. J. Food Prot. 2004; 67:53-59.


Latham E. A., Anderson R. C., Pinchak W. E., Nisbet D. J. Insights on alterations to the rumen ecosystem by nitrate and nitrocompounds. Front. Microbiol. 2016; 7 Article 228.

Le Marc Y., Valík L., Medvedová A. Modelling the effect of the starter culture on the growth of Staphylococcus aureus in milk. Int. J. Food Microbiol. 2009; 129:306-311.


Lee H., Kim K., Lee S., Yoon Y. Kinetic behaviour of Staphylococcus aureus on cheese as a function of water activity and temperature. J. Dairy Res. 2015b; 82:64-69.


Lee J., Skandamis P., Park A., Yoon H., Hwang I.-G., Lee S.-H., Cho J.-I., Yoon Y. Development of mathematical models to predict Staphylococcus aureus growth in sauces under constant and dynamic temperatures. Food Sci. Technol. Res. 2013; 19:331-335.


Lee N., Kim C. S., Yu G. S., Park M. C., Jung W. O., Jung U. K., Joung J. Y., Kim K. H., Yook H. S. Effect of nitrite substitution of sausage with addition of purple sweet potato powder and purple sweet potato pigment. J. Korean Soc. Food Sci. Nutr. 2015a; 44:896-903.


Lee S., Lee H., Kim S., Lee J., Ha J., Gwak E., Oh M.-H., Park B.-Y., Kim J.-S., Choi K.-H., Yoon Y. Probabilistic models to describe the effect of NaNO
2 in combination with NaCl on the growth inhibition of Lactobacillus in frankfurters. Meat Sci. 2015c; 110:302-309.


Normanno G., La Salandra G., Dambrosio A., Quaglia N. C., Corrente M., Parisi A., Santagada G., Firinu A., Crisetti E., Celano G. V. Occurrence, characterization and antimicrobial resistance of enterotoxigenic Staphylococcus aureus isolated from meat and dairy products. Int. J. Food Microbiol. 2007; 115:290-296.


Park C. E., Akhtar M., Rayman M. K. Nonspecific reactions of a commercial enzyme-linked immunosorbent assay kit (TECRA) for detection of staphylococcal enterotoxins in foods. Appl. Environ. Microb. 1992; 58:2509-2512.

Park H. J., Go E. K., Wee S.-H., Yoon H.-C., Heo E.-J., Kim Y.-J., Lee H.-S., Moon J.-S. Analysis of foodborne pathogenic contamination of cooked hams and sausage in Korean processing facilities. Korean J. Food Sci. An. 2012; 32:103-111.


Pereira H. C., Souza V. R., Azevedo N. C., Rodrigues D. M., Nunes C. A., Pinheiro A. C. M. Optimization of low sodium salts mix for shoestring potatoes. J. Food Sci. 2015; 80:S1399-S1403.


Schlag S., Fuchs S., Nerz C., Gaupp R., Engelmann S., Liebeke M., Lalk M., Hecker M., Götz F. Characterization of the oxygen-responsive NreABC regulon of Staphylococcus aureus. J. Bacteriol. 2008; 190:7847-7858.


Shapiro L., Eason C., Bunt C., Hix S., Aylett P., Mac-Morran D. Encapsulated sodium nitrite as a new toxicant for possum control in New Zealand. New Zeal. J. Ecol. 2016; 40:1-5.


Singh V. K., Hattangady D. S., Giotis E. S., Singh A. K., Chamberlain N. R., Stuart M. K., Wilkinson B. J. Insertional inactivation of branched-chain alpha-keto acid dehydrogenase in Staphylococcus aureus leads to decreased branched-chain membrane fatty acid content and increased susceptibility to certain stresses. Appl. Environ. Microb. 2008; 74:5882-5890.


Tompkin R. B., Ambrosino J. M., Stozek S. K. Effect of pH, sodium chloride, and sodium nitrite on enterotoxin A production. Appl. Environ. Microb. 1973; 26:833-837.

Yoon H., Lee J-Y., Suk H-J., Lee S., Lee H., Lee S., Yoon Y. Modeling to predict growth/no growth boundaries and kinetic behavior of Salmonella on cutting board surface. J. Food Prot. 2012; 75:2116-2121.


Yoon Y., Lee H., Lee S., Kim S., Choi K.-H. Membrane fluidity-related adaptive response mechanisms of foodborne bacterial pathogens under environmental stresses. Food Res. Int. 2015; 72:25-36.