Introduction
Resveratrol, 3,5,4-trihydroxystilbene, is a polyphenolic hydrophobic bioactive compound found in various plants including grapes, peanuts, and mulberries (Liang et al., 2008). It has been reported that resveratrol has numerous health benefits, such as antioxidant, anti-inflammatory, and cardio-protective activities (Das and Ng, 2010). However, the dairy food application of resveratrol can be limited since hydrophobic bioactive compounds including resveratrol have extremely low water solubility and poor stability during food processing, storage, and digestive processes (Ha et al., 2013).
This food application problems regarding resveratrol can be solved by the use of nano delivery systems for hydrophobic bioactive compounds (Sessa et al., 2014). Nano delivery systems are carriers to encapsulate and protect bioactive compounds and have subcellular size between 1 and ~200 nm, which may increase the bioavailability of hydrophobic bioactive compounds (Fathi et al., 2012; Ron et al., 2010). For dairy food application, nano delivery systems need to be manufactured with inexpensive and food grade materials, such as whey protein isolate (Ron et al., 2010). Whey protein isolate (WPI) contains above 90% of whey proteins that are β-lactoglobulin and α-lactalbumin (Fioramonti et al., 2014; He and Ruan, 2009). Whey protein has high nutritional values, biodegradability, and GRAS statues (Livney, 2010). Moreover, whey protein has been known to bind various hydrophobic bioactive compounds, such as retinol, vitamin D, and resveratrol, via hydrophobic interactions, van der Waals attraction, and hydrogen bonds (Liang et al., 2008; Livney, 2010).
In this study, WPI can form a complex with food grade polysaccharide that is inulin to produce nano delivery system. Inulin, a non-digestible polysaccharide, has prebiotic activity. Prebiotics are indigestible carbohydrate substances, which can selectively enhance the growth and activity of probiotic bacteria during food processing and in the gastro-intestinal tract providing benefits to host (Donkor et al., 2007). Inulin can enhance the activity of beneficial live active cultures, such as Bifidobacetrium ssp. and Lactobacillus ssp. (e.g., Lactobacillus acidophilus) (Bruno et al., 2002; Donkor et al., 2007; Tobin et al., 2010). Moreover, inulin also inhibits the growth of harmful bacteria in gastrointestinal tract, helps to remove toxins from bodies, and enhances the body’s natural defenses (Aryana et al., 2007). The use of whey protein/polysaccharide complex can provide additional protections on the encapsulated hydrophobic bioactive compounds (Lee et al., 2013; Zimet and Livney, 2009). Therefore, the successful manufacture of WPI/inulin nano delivery system may provide benefits to dairy industry due to its additional protection and potential prebiotic effect. However, there have been no studies on the formation of WPI/inulin nano delivery system and its physicochemical and potential prebiotic properties.
The formation of those barriers may provide protective effects, which can reduce the diffusion of bioactive compounds from WPI/inulin nano complexes, and thereby could improve the protection of encapsulated bioactive compounds (Livney, 2010; Ron et al., 2010). In this study, we hypothesized that whey protein and inulin concentration levels, which are important factors affecting the complex formation, can be key factors the physicochemical properties of whey protein/inulin nano complexes containing resveratrol and nano complexes may have the potential prebiotic effects. The objectives of this research were to produce WPI/inulin nano complexes for the delivery of resveratrol, to study how WPI and inulin concentration levels affected the physicochemical properties of nano complexes, and to investigate the potential prebiotic effects of nano complexes.
Materials and Methods
Whey protein isolate (WPI) was provided by Davisco Food International Inc. (USA). CaCl2, resveratrol, was purchased from Sigma-Aldrich (USA). Inulin (orafti® HP) was purchased from BENEO-Orafti (Belgium).
WPI/inulin nano complexes were manufactured by the modified ionic gelation method with CaCl2 (Leclerc et al., 2005; Lee et al., 2008). Five milliliters of WPI solution (2, 4, and 6 mg/mL) were mixed with 5 mL of inulin solution (2, 4, and 6 mg/mL). The final concentration level of WPI and Inulin was 1, 2, and 3% (w/v). Forty microliters of 1 mM CaCl2 solution were added to 10 mL of WPI/inulin mixtures followed by adjusting to pH 9.5 with 0.1 M NaOH. Final concentration of CaCl2 was 4 mM. WPI/inulin mixtures were then heated at 70℃ for 10 min and cooled to room temperature (~20℃) in ice water. To encapsulate resveratrol, 200 μL of resveratrol (5 mg/mL) in ethanol was added to 4.8 mL of WPI solution (2, 4, and 6 mg/mL) and then mixed with 5 mL of inulin solution (2, 4, and 6 mg/mL) followed by the addition of 4 mM CaCl2 to WPI/inulin mixtures. WPI/inulin mixtures containing resveratrol were adjusted to pH 9.5, heated at 70℃ for 10 min, and then cooled to room temperature.
Transmission electron microscopy (TEM, Netherlands) with accelerating voltage of 120 kV was used to investigate the formation and morphological characteristics of WPI/inulin nano complexs. Nano complexes were placed on a 200 mesh copper grid treated with carbon (TED PELLA Inc., USA) and negatively stained with 0.4% uranyl acetate for 15 s. Nano complexes on a grid were airdried at room temperature followed by observation with TEM.
The physicochemical properties, such as particle size, polydispersity index, and zeta-potential value, of WPI/inulin nano complexes were determined by the use of a particle size analyzer (Zeta sizer Nano-ZS, UK). Prior to the measurement, nano complexes were 10-fold diluted with deionized water. Measurement was performed at room temperature with scattering angles of 173°.
The encapsulation efficiency of resveratrol was investigated by the modified method of Ha et al. (2013) using HPLC system (Agilent 1260 Series, USA). To separate unencapsulated resveratrol (free resveratrol) from resveratrol- loaded nano complex suspensions, those suspensions were centrifuged at 10,000 g for 30 min. After centrifugation, clear supernatants (free resveratrol) was collected by the use of 0.45-μm syringe filter. Twenty microliters of standard and supernatant (free resveratrol) were injected through a reversed-phase C18 column (3.5 μm, 4.6 × 150 mm, Waters Company, USA) and then the amounts of free resveratrol were analyzed using HPLC system. HPLC system was composed of a quaternary pump (Agilent 1260 Series, G1311B) and a diode array detector (Agilent 1260 Series, G1315C). The mobile phase consists of 52% of 0.5% acetic acid in methanol and 48% of water. The flow rate was set at 0.7 mL/min while detection wavelength was set at 303 nm. A calibration curve was constructed by plotting peak area versus the amount of various concentration levels of resveratrol from 0 to 0.05 mg/mL and was used to determine the amount of unencapsulated resveratrol (free resveratrol). Encapsulation efficiency was calculated by the following equation.
Probiotic culture, L. acidophilus ATCC 43121, was obtained from the Dairy Food Microbiology Laboratory at Korea University (Korea). Probiotic culture was grown in de Man, Rogosa, and Sharpe (MRS) media (Difco Laboratories, USA) at 37℃ for 18 h prior to use. Stock cultures were maintained at −80℃ using 20% glycerol as a cryoprotectant.
The prebiotic effect of WPI/inulin nano complex was assessed by determining the viability of L. acidophilus ATCC 43121 in minimum media using standard plate method (Koh et al., 2013). Minimum media contain 5.0 g of peptone, 2.5 g of sodium acetate, 0.5 mL of 1 M magnesium sulfate heptahydrate, 0.5 mL of 1 M manganese (II) sulfate tetrahydrate, 5.0 mL of Tween 80, 1.0 g of diammonium citrate, and 1.0 g of dipotassium phosphate per liter. To determine the potential prebiotic effect of WPI/inulin nano complex, L. acidophilus ATCC 43121 was inoculated at 106 cfu/mL in minimum media without inulin (negative control), with 1% (w/v) inulin (positive control), with 1% (w/v) WPI, and with WPI/inulin nano complex (the final concentration levels of WPI and inulin were 0.5%, w/v, respectively). Probiotic culture was then incubated at 37℃ for 0, 6, 12, 24, and 48 h. The number of viable cells were investigated by using standard plate method on MRS agar.
One-way analysis of variance (ANOVA) with Fisher’s significant differences (LSD) test was used to determine the effects of manufacturing variables, such as WPI and inulin concentration levels, on the physicochemical properties of WPI/inulin nano complexes. Statistical significance was set at p<0.05. The impacts of each treatments (negative control, positive control, WPI, and WPI/inulin nano complex), incubation time, and their interactions on the viability of L. acidophilus ATCC 43121 during 48 h incubation were assessed by two-way ANOVA. All data were expressed as mean±standard errors of three replicates. SAS software package was used for the statistical analyses (Version 9.1, SAS Institute Inc., USA).
Results and Discussion
WPI/inulin nano complexes were prepared by using the modified ionic gelation method with CaCl2. In this study, various concentrations of WPI and inulin mixtures at pH 9.5 were heated at 70℃ for 10 min in the presence of 4 mM CaCl2 followed by cooling to room temperature. The heat treatment of WPI solutions above 70℃ leads to partial unfolding of whey proteins, which can result in the exposure of hydrophobic residues and free sulfhydryl groups to aqueous environment. It can contribute to an increased possibility of hydrophobic attractions and formation of disulfide bonds between whey proteins (Ha et al., 2013), which may result in increased aggregation of whey proteins molecules. Since isoelectric point of whey protein is about 5.3 (Chen and Subirade, 2005), whey proteins are negatively charged at pH 9.5 and hence there can be electrostatic repulsions between whey protein molecules. In the presence of positive ions, such as calcium, may improve the aggregation between whey protein molecules (Bryant and McClements, 2000; Koewmaneechai and McClements, 2002). Inulin also had gel-forming property and can form gels with whey protein (Tobin et al., 2010). Whey proteins, mainly β-lactoglobulin, can interact with inulin via hydrogen bondings and hydrophobic interactions (Gilbowski and Gilbowska, 2009; Schaller-Povolny and Smith 2002). Gilbowski (2009) reported that heating of whey protein solutions at 80℃ for 30 min in the presence of inulin resulted in the formation of whey protein/inulin complexes by interacting between hydrophobic groups of whey protein and non-polar groups of inulin. The morphological characteristics of WPI/inulin nano complexes with various WPI concentration levels were shown in Fig. 1. In TEM micrographs, globular particles with size raining from ~10 to 100 nm were observed indicating that WPI/inulin nano complexes were successfully formed (Fig. 1). It seems that an increase in WPI concentration level resulted in the formation of larger WPI/inulin nano complexes.

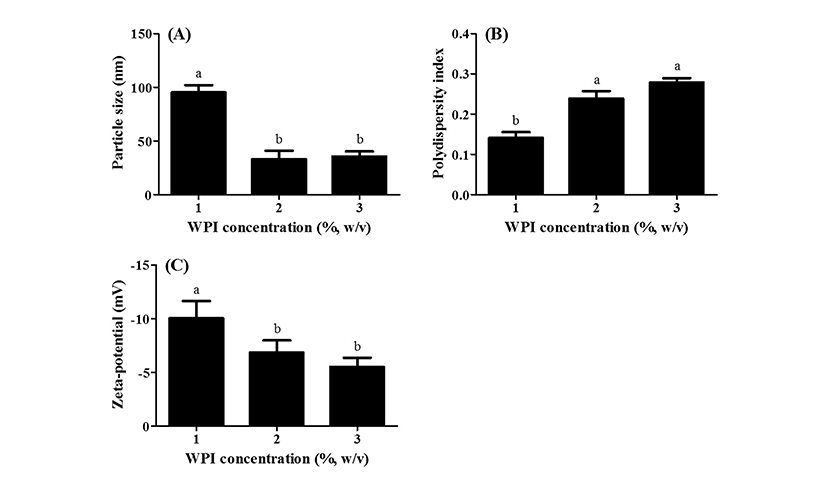
Effects of WPI and inulin concentration levels on the physicochemical properties, such as particle size, polydispersity index, and zeta-potential value, of WPI/inulin nano complexes were shown in Figs. 2 and 3, respectively. There was a significant (p<0.05) decrease in the size of nano complexes from 95 to 36 nm when WPI concentration level was increased from 1 to 3% (w/v) (Fig. 2A). In this study, WPI/inulin nano complexes were prepared with 4mM CaCl2 at pH 9.5, where whey proteins are negatively charged. The presence of calcium ions could shield negatively charged whey proteins resulting in a decrease in electrostatic repulsions among whey protein molecules (Bryant and McClements, 2000). It may enhance intramolecular associations within whey protein molecules contributing to the formation of more compact whey protein structures. This may result in a reduction in the size of WPI/inulin nano complexes at higher WPI concentration level. All the polydispersity index values of WPI/inulin nano complexes manufactured with 1, 2, and 3% (w/v) WPI were below 0.3 (Fig. 2B) indicating that there were adequate uniform particle distributions observed in WPI/inulin nano complexes (Hu et al., 2008). In Fig. 2C, WPI/inulin nano complexes prepared with 1, 2, and 3% (w/v) WPI concentration levels exhibited zeta-potential values ranging from −5.5 to −10.0 mV. As WPI concentration level was increased, zeta-potential values were significantly (p<0.05) increased from −10 to −5.5 mV indicating that the negative charges of nano complexes were reduced with increased WPI concentration level. Because the isoelectic pH of WPI is about pH 5.0 (Demetriades et al., 1997), WPI/inulin nano complexes manufactured at pH 9.5 had negative surface charges. At higher WPI concentration level, there may be more calcium ions shielding negatively charged whey proteins, which can lead to a reduction in negative surface charges (Bryant and McClements, 2000).
The physicochemical properties of WPI/inulin nano complexes manufactured with various inulin concentration levels were shown in Fig. 3. There were no significant (p<0.05) differences in the particle size of WPI/inulin nano complexes when inulin concentration level was increased from 1 to 3% (Fig. 3A). It was reported that the presence of whey proteins may disturb and weaken the inulin gels (Gilbowski, 2009; Gilbowski and Gilbowska, 2009). Inulin gel formation is done by crowding effect of inulin crystal seeds which interacts each other via hydrogen and van der Waals bonding. Since whey proteins can interact with inulin by hydrogen bonding and hydrophobic interactions, whey proteins could interrupt the aggregation and growing of inulin crystal seeds, which may result in the formation of nano complexes with similar particle size. (Gilbowski and Gilbowska, 2009; Schaller- Povolny and Smith 2002). An increase in inulin concentration level from 1 to 3% resulted in an increase in polydispersity index values from 0.141 to 0.285 (Fig. 3B). Although the different polydispersity index values of nano complexes were observed, all WPI/inulin nano complexes manufactured with 1-3% inulin concentration level exhibited polydispersity index value below 0.3, which was considered as the formation of nano complexes with a narrow (homogeneous) size distribution. There were no significant (p<0.05) differences in the zeta-potential values of nano complexes when inulin concentration level was increased from 1 to 3% (Fig. 3C).
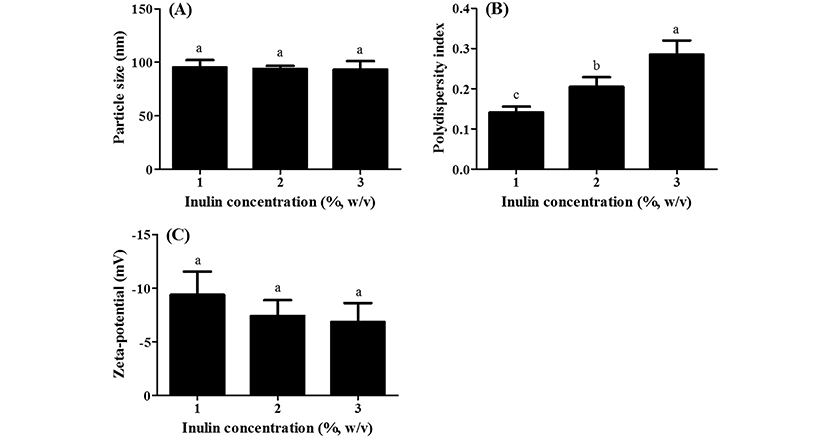
Fig. 4 exhibits the impacts of WPI and inulin concentration levels on the encapsulation efficiency of resveratrol in WPI/inulin nano complexes. A significant increase (p<0.05) in the encapsulation efficiency of resveratrol from 78 to 84% were observed as WPI concentration level was increased from 1 to 3% (Fig. 4A). Whey proteins can bind various hydrophobic bioactive compounds including resveratrol via hydrophobic interactions and hydrogen bondings (Liang et al., 2008; Livney, 2010). Liang et al. (2008) reported that β-lactoglobulin can bind resveratrol and form 1:1 complexes. As WPI concentration level was increased, there were more whey protein molecules participating in the formation of WPI/inulin nano complexes. It may offer more binding sites for resveratrol, which can result in an increase in the encapsulation efficiency of resveratrol.
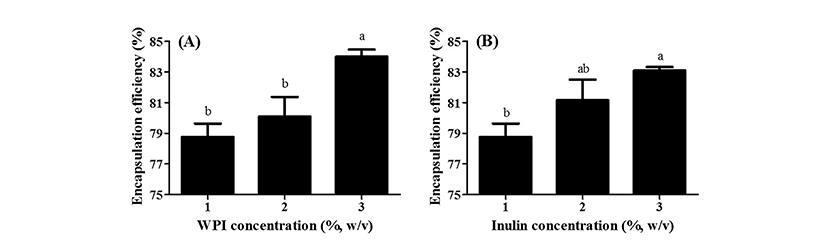
When inulin concentration level was increased from 1 to 3%, the encapsulation efficiency of resveratrol was significantly (p<0.05) enhanced from 79 to 83% (Fig. 4B). It has been reported that the use of whey protein and polysaccharide (e.g., pectin and alginate) complexes for delivery systems could provide additional protections to encapsulated bioactive compounds due to their barrier effects against the diffusion of bioactive compounds in the complex (Ha et al., 2013; Lee et al., 2013; Livney, 2010; Ron et al., 2010). It is expected that the diffusion of bioactive compounds in whey protein and polysaccharide complexes can be relatively reduced compared with those in free solution. In this study, resveratrol-loaded nano complexes were formed with whey protein and inulin. Since inulin can form complexes with whey proteins, there may be more barrier effects in WPI/inulin nano complexes prepared with higher inulin concentration level, which can reduce the diffusion of resveratrol in nano complex. It may result in an increase in the encapsulation efficiency of resveratrol.
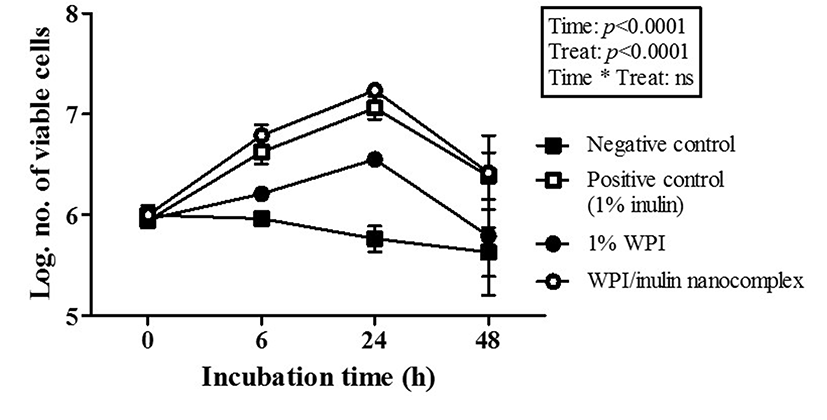
The potential prebiotic effect of WPI/inulin nano complex was studied by determining the number of viable cells during incubation at 37℃ for 48 h. Two-way ANOVA results exhibited that there were significant (p<0.0001) differences among treatments and incubation time while their interactions were not significantly different (Fig. 5). It indicates that treatments, which were the addition of various prebiotic substrates, such as 1% inulin, 1% WPI, and WPI/inulin nano complexes, to minimum media and incubation time had significant (p<0.0001) effects on the number of viable cells during 48-h incubation. Compared with negative control (minimum media only), increase in the number of viable cells during 48-h incubation was observed in positive control (minimum media containing 1% inulin) (Fig. 5). At 24-h incubation at 37℃, the number of viable cells of positive control was about 1.2 log cycle higher than those of negative control. Addition of 1% WPI led to higher number of viable cells than negative control, but lower than positive control (Fig. 5) indicating that inulin had more pronounced effects to enhance the growth of L. acidophilus ATCC 43121 than WPI. It was reported that inulin can promote the growth of L. acidophilus subsp. (Donkor et al., 2007). To investigate the potential prebiotic effect of nano complex form of inulin, WPI/inulin nano complexes were added to minimum media and incubated at 37℃ for 48 h. Compared with negative control and 1% WPI, the viability of cells grown in minimum media containing WPI/inulin nano complexes was higher than that of negative control and 1% WPI while similar probiotic viability was observed between WPI/inulin nano complexes and free inulin (positive control) (Fig. 5). These results indicate that both free and nano complex forms of inulin exhibited the potential prebiotic effect on L. acidophilus ATCC 43121.
Conclusions
The globular forms of WPI/inulin nano complexes, which had size between A and B were successfully manufactured. Manufacturing variables, such as WPI and inulin concentrations levels, were critical factors affecting the physicochemical properties of WPI/inulin nano complexes. WPI/inulin nano complexs were useful to entrap hydrophobic bioactive compound, which is resveratrol, and had potential prebiotic effect. This study exhibited that WPI/inulin nano complexe can be used as a delivery system for hydrophobic bioactive compounds as well as a potential prebiotic source for probiotics.













