Introduction
The smoked and sliced duck products are particularly convenient for the consumers as they could be consumed in general without any additional heating, and thus their consumption has increased in Korea (Lee et al., 2015). However, Listeria monocytogenes has been isolated from sliced smoked ducks in Korea. Hence, there is a possibility for listeriosis through smoked ducks. A previous study also suggested that smoked duck products are not microbiologically safe enough for consumption, if they are consumed without additional heating, although smoked, sliced duck could be consumed with additional heating (Kim et al., 2016). Thus, smoked duck-related listeriosis is a valid possibility.
L. monocytogenes is a gram-positive zoonotic pathogen causing listeriosis (Leclercq et al., 2019). According to Leclercq et al. (2019), the 19 species consist Listeria genus, among which L. ivanovii and L. monocytogenes are considered pathogenic and only L. monocytogenes is considered as a foodborne pathogen, threatening human public health, especially in pregnant women, neonates, immunocompromised patients, and elderly. Although 13 L. monocytogenes strain serotypes have been identified, the human listeriosis cases occurred serotype 1/2a, 1/2b, and 4b (Kathariou et al., 2002). The pathogen could survive in foods, including high-salt-containing products at low temperatures. L. monocytogenes has been isolated from ready-to-eat (RTE) foods such as frankfurters and deli meat, which contain high fat approximately 30% (Burall et al., 2012). L. monocytogenes was previously considered to most antibiotics to be susceptible (Wieczorek et al., 2012) but Oh et al. (2018) suggested that recent studies have reported its resistance against amoxicillin, ampicillin, chloramphenicol, erythromycin, gentamicin, vancomycin, and, tetracycline. A previous study described the analysis of 400 dairy products, meat products, seafood, RTE foods, and fresh vegetables in Taipei identified Listeria isolates in these products resistant to chloramphenicol (3.70%), tetracycline (1.96%), and penicillin (7.58%) (Wang et al., 2012). However, studies on antibiotic-resistant L. monocytogenes are still limited in Korea (Oh et al., 2018).
L. monocytogenes reportedly possesses low thermotolerance, Linton et al. (1992) reported that L. monocytogenes with increased heat resistance. Recently, the heat resistance of Listeria spp. has increased compared to other bacteria. Gutler et al. (2011) suggested that inactivation of Salmonellaenteritidis in 10% salted egg yolk requires a minimum heating temperature of 63.3°C for a minimum duration of 3.5 min, according to the current USDA’s regulatory requirement. In 2014, the average pasteurization temperature (time) were 64.4°C (4.4 min) for egg yolks for the thermal inactivation of Listeria species in the egg products, according to the survey of the egg industry.
Therefore, the objective of this study was to isolate L. monocytogenes from smoked ducks in Korea and to determine the serotypes, antibiotic susceptibility profiles, as well as the heat resistance of the isolates.
Materials and Methods
Twelve smoked duck samples were collected from local supermarkets in Seoul, Korea. For qualitative analysis, smoked duck slices were aseptically removed from the packages, and 25 g slices were transferred aseptically into a sample bag (3M, St. Paul, MN, USA), with 50 mL Listeria enrichment broth (Becton Dickinson and Company, Sparks, MD, USA). The samples were homogenized for 1 min at high speed in a stomaker (BagMixer® 400, Interscience, Saint Nom, France), followed by incubation at 30°C for 24 h. After, 1 mL of primary enrichment was cultured in Fraser broth (Becton Dickinson and Company) containing Fraser broth supplement (Becton Dickinson and Company) at 37°C for 24 to 48 h. The tubes that turned dark black were streaked on Palcam agar (Oxoid, Basingstoke, UK) and inoculated at 30°C for 24 to 48 h in order to isolate colonies with black circles.
Single isolated colony on Palcam agar was aseptically transferred into 10 mL tryptic soy broth containing 0.6% yeast extract (TSBYE), and cultured at 30°C for 24 h. One-milliliter of the cultures were transferred into microtubes and centrifuged at 8,000×g for 3 min, and the resulting supernatants were discarded. The pellet was suspended with 0.1 mL of dH2O and then heated at 100°C for 10 min to be used as the DNA template. After adding 0.5 μM primer hlyA (F: 5’CCTAACATATCCAGGTGCTCTC 3’ R: 5’CTGATTGCGCCGAAGTTTAC 3’, described in Table 1, following Burall et al., 2011) and 2 μL of DNA template to 20 μL of a phage hot start II DNA polymerase kit (Thermo Fisher, Waltham, MA, USA), withho Taq DNA polymerase (pH 7.4 at 25°C; 50% glycerol; 1 mM DTT; 20 mM Tris-HCl; 0.1 mM EDTA; 100 mM KCl; and 200 μg/mL BSA), 1× reaction buffer, and 200 μM dNTPs (dNTP; Promega, Madison, USA), the PCR reaction was performed. For the hlyA amplification, after an initial stage of heating at 98°C for 30 s, the amplification conditions were performed that denaturation at 98°C for 5 s, annealing at 60°C for 5 s, and extension at 72°C for 10 s (35 cycles). A final extension was performed at 72°C for 1 min. The amplified products were followed by electrophoresis using a 1.5% agarose gel. To identify L. monocytogenes, by 16S rRNA sequencing the hlyA-positive samples were further analyzed. The 16S rRNA sequencing were performed using the primers 27F (5’AGAGTTTGATCMTGGCTCAG3’) and 1492R (5’TACGGYTACCTT GTTACGACTT 3’) (Lane, 1991). The PCR reaction was conducted with 20 ng genomic DNA template in a 30-μL reaction mixture using EF-Taq (Solgent, Daejeon, Korea). The Taq polymerase was activated at 95°C for 2 min, performed by 35 cycles of 95°C for 1 min, 55°C for 1 min, and 72°C for 1 min, and a final stage of 72°C for 10 min. Using a multiscreen filter plate (Millipore, Bedford, MA, USA) the amplified products were purified and then added to Hi-Di formamide (Applied Biosystems, Foster City, CA, USA). The mixed products were stored at 95°C for 5 min and on ice for 5 min, and then analyzed using an ABI Prism 3730XL DNA analyzer (Applied Biosystems).
| Primer | Size (bp) | Sequence (5′ to 3′) | Reference |
|---|---|---|---|
| hlyA | 350 | F: CCTAACATATCCAGGTGCTCTC R: CTGATTGCGCCGAAGTTTAC |
Burall et al., 2011 |
Five virulence genes (plcB, inlA, inlB, hlyA, and actA) were detected with the primers using PCR analysis (Table 2). Among the isolated colonies from the Palcam agar plates. The isolated colonies on the plates were suspended in 50 μL of 0.05N NaOH (Samchun, Gwangmyeong, Korea) added 0.25% sodium dodecyl sulfate (SDS). The suspension was then suspended with 100 μL of sterile dH2O and inoculated at 99°C for 15 min. For the PCR amplification, using Phire Hot Start II DNA Polymerase Kit (Thermo Fisher, Waltham). This mixture (2 μL) was added 0.5 μM each primer, Taq DNA polymerase, and 1× reaction buffer (200 μM dNTP, 1.5 mM MgCl2). The PCR reaction was performed with initial denaturation at 98°C for 30 s, followed by 98°C for 5 s, 60°C for 5 s, and 72°C for 10 s (35 cycles), and a final extension at 72°C for 1 min using Rotor-Gene Q (Qiagen). For the detection of the products, 20 μL of the PCR reactions were added 4 μL of loading star (Dyne Bio, Seongnam, Korea), followed by electrophoresis using a 1.5% agarose gel.
| Primer | Size (bp) | Sequence (5′ to 3′) | Reference |
|---|---|---|---|
| inlA | 255 | F: CCTAGCAGGTCTAACCGCAC R: TCGCTAATTTGGTTATGCCC |
Mathakiya et al., 2009 |
| inlB | 146 | F: AAAGCACGATTTCATGGGAG R: ACATAGCCTTGTTTGGTCGG |
Corantin et al., 2005 |
| actA | 268 (385) |
F: GACGAAAATCCCGAAGTGAA R: CTAGCGAAGGTGCTGTTTCC |
Jaradat et al., 2002 |
| plcB | 261 | F: GGGAAATTTGACACAGCGTT R: ATTTTCGAAGGTAGTCCGCTTT |
Corantin et al., 2005 |
| hlyA | 174 | F: GCATCTGCATTCAATAAAGA R: TGTCACTGCATCTCCGTGGT |
Wesley et al., 2002 |
In order to identify the serotypes, both antigen-antibody agglutination assay (Denka Seiken, Tokyo, Japan) and multiplex PCR with five primers (prs, lmo0737, lmo1118, ORF2819, ORF2110) were performed. The isolates were cultured in 10 mL TSBYE and inoculated at 30°C for 24 h. To increase the activity of the strain, 0.1 mL of the culture was inoculated into 10 mL fresh TSBYE and inoculated at 30°C for 24 h. The cultures were streaked onto brain heart infusion agar (BHI agar; Becton Dickinson and Company, Sparks) and inoculated at 30°C for 24 h. One isolated colony was suspended in 0.3 mL of 0.2% sodium chloride solution. The mixed suspension was boiled at 100°C for 30 min and then centrifuged at 1,912×g for 20 min. The pellet that supernatant was removed, was added 0.1 mL of 0.2% sodium chloride. One drop of OI/II, OV/VI antisera (Denka seiken, Tokyo, Japan) and saline solution were added to the slide glass, mixed with 5 μL of the antigen, and the agglutination was judged to occur within 1 min. When the OI/II antisera were positive, the OI and OIV antisera were used for the agglutination reaction. When the OV/VI serotype was positive, OVI, OVII, OVIII, and OIX antisera were used for the agglutination. To test the agglutination assay for the H antigen, the isolates were cultured in 10 mL TSBYE and inoculated at 30°C for 24 h. One tenth milliliter of the culture was inoculated into 10 mL of fresh TSBYE and inoculated at 30°C for 24 h. The cultures were plated on BHI broth semi-fluid medium with 0.2% agar in triplicates. The cultures were mixed with the same volume of physiological saline with 1% formalin and used as an antigen. The antigen (0.3 mL) was dispensed in a test tube, and 2 drops of H antisera (Denka Seiken, Tokyo, Japan) were dropped and placed in a water bath (Wisebath; Wisd Laboratory Instruments, Wertheim, Germany) at 50°C for 1 h. To perform the serotyping multiplex PCR test, 1 colony of the isolates from the Palcam agar was suspended in 50 μL of 0.05N NaOH containing 0.25% SDS, heated at 100°C for 15 min, then cooled at room temperature for 2 min. After cooling, the mixture was centrifuged at 14,000×g for 3 min. The supernatant was used as template DNA for PCR. The PCR was performed using the Qiagen multiplex PCR kit. Four microliters of primer set, and 19 μL of H2O, and 2 μL of template DNA were added into 25 μL of PCR premix with MgCl2, KCl, dNTPs, and Taq polymerase, and homogenization was initiated. For the L. monocytogenes gene of serovar amplification, the first stage was initiated at 94°C for 15 sec, performed by the amplification conditions using denaturation at 94°C for 30 s, annealing at 57°C for 90 s, and extension at 72°C for 60 sec (35 cycles), and then final 30 sec extension was performed at 60°C. The amplified products were subjected to electrophoresis on 2% agarose.
To evaluate whether L. monocytogenes isolates have antibiotic resistance, an antibiotic susceptibility examination was performed according to the Clinical Laboratory Standards Institute procedure to evaluate antibiotic resistance (CLSI, 2014). A single isolated colony of L. monocytogenes on Palcam agar was transferred into 10 mL of TSBYE and inoculated at 30°C for 24 h. After incubation, using 0.5 McFarland opacity standard (Biomerieux, Marcy l’Etotile, France) the cultures were adjusted in phosphate-buffered saline (PBS; pH 7.4, 8.0 g of NaCl, 0.2 g of KCl, 1.5 g of Na2HPO4, 0.2 g of KH2PO4, 1 of distilled water). The sterilized cotton swab containing the suspension was swabbed on the surface of a Muller-Hinton agar (Becton Dickinson and Company) plate, which was dried. Antibiotic discs (Oxoid) were then placed on a MHA plate using disc dispenser and inoculated at 30°C for 24 h. Nine antibiotic discs [ciprofloxacin (5 μg), chloramphenicol (30 μg), clindamycin (10 μg), doxycycline (30 μg), erythromycin (15 μg), minocycline (30 μg), penicillin G (10 units), rifampicin (5 μg), and tetracycline (30 μg)] were examined in this study. After the inoculation, the diameters of the inhibition zones were measured. Antibiotic susceptibility profiles of the isolates were classified as either resisant, intermediate, or sensitive, according to the M45 guidelines of the Clinical and Laboratory Standards Institute (CLSI, 2014).
To test the heat resistance, the isolates on Palcam agar were inoculated onto TSBYE and cultured at 30°C for 24 h. One tenth milliliter of the cultures were inoculated into 10 mL fresh TSBYE at 30°C for 24 h. After the incubation, seven cultures were centrifuged at 1,912×g and 4°C for 15 min, and then the pellets were resuspended and diluted with PBS in order to obtain an inoculum concentration of 6.5 to 7.5 Log CFU/mL. One tenth milliliter of the L. monocytogenes suspensions were transferred into 10 mL of fresh TSBYE and heated in a water bath at 60°C for 10 min. In order to quantify the survival, 1 mL of the samples were spread-plated on TSA containing 0.6% yeast extract (TSAYE) at 0, 2, 5, 8, and 10 min. The TSAYE were inoculated at 30°C for 48 h, and then the colonies were counted manually. D-values (in minutes) were calculated from survivor curves as described in the Laboratory Manual for Food Canners and Processors, volume 1 (National Canners Association, 1968).
Results
Five samples among the total of twelve (41.7%) showed positive results in the qualitative analysis. These isolates harbored the hlyA gene (Fig. 1) and were eventually identified as L. monocytogenes through our 16s rRNA analysis (data not shown).
Five virulence genes (inlA, inlB, actA, plcB, and hlyA) were observed in the L. monocytogenes isolates. All strains contained these aforementioned five genes. Interestingly, all seven isolates of this study harbored the 385 bp amplicon of actA (Fig. 2A–C), a gene which is essential for F-actin assembly and cellular movements, with a length reportedly related to pathogenicity (Wiedmann et al., 1997). A previous study described that the invasion efficiency of L. monocytogenes carrying the 385 bp actA amplicon was approximately 1 to 2.5 times higher than that of L. monocytogenes carrying the 268 bp version of actA in healthy cells (Ha et al., 2018). Thus, these results indicate that the isolates from smoked duck products exhibited high pathogenicity.

The L. monocytogenes serotypes were determined using both agglutination assay and multiplex PCR (Oh et al., 2018), and a common serotype from both assays were used as the serotype in Table 3. The serotypes of the isolates were 1/2a (3 cases out of 7, 42.9%), 1/2b (3 cases out of 7, 42.9%), and 3b (1 case out of 7, 13.3%), except for one isolates, which are generally regarded as the most common serotypes of foodborne diseases (Table 3). A previous study suggested that serotype 1/2a has an increased persistence in food and seems to transfer more plasmids, which confer resistance to toxic metals and other compounds (Wu et al., 2015). The serotype 1/2b (42.9%) was present more often in non-pregnant individuals with insignificant occurrence of severe underlying diseases (McLauchlin. 1990). Finally, serotype 3b, which is not a highly prevalent serotype, exhibited a very high adhesion and invasion efficiency in Caco-2 cells (Jaradat and Bhunia, 2003).
L. monocytogenes is sensitive to many of antibiotics. However, the emergence of resistant strains has been recently reported (Wu et al., 2015). All seven isolates of this study were resistant to penicillin G, and three of them were also resistant to clindamycin (Table 4). Several previous studies reported the low prevalence of penicillin G-resistant L. monocytogenes. Duck and goose-derived, beef and broiler meat-derived, and beef, pork, or poultry-derived penicillin G-resistant L. monocytogenes isolates have been reported in 31.6%, 0% to 5%, and 0% of the cases, respectively, in different studies (Jamali et al., 2014). Therefore, the result from our study suggests that the previously reported L. monocytogenes penicillin G-resistance has obviously increased. A previous study suggested that the resistance of gram-positive bacteria, including L. monocytogenes, to β-lactam antibiotics (e.g., penicillin) might be associated with a reduced antibiotic affinity of the penicillin-binding proteins (Malouin et al., 1986). In addition, Thedieck et al. (2006) suggested that L. monocytogenes activates the antimicrobial peptide sensor system when it detects a treat to antibiotics and that active efflux systems might contribute to the adaptation of Listeria to these environmental challenges. The use of β-lactam antibiotics (e.g., penicillin) shows a global increase. Penicillin, a β-lactam antibiotic, has been reported as the first-line listeriosis treatment in immunocompromised patients (Hof, 2003). These results suggest that the resistance against β-lactam antibiotics has increased, and thus, the use of these antibiotics should be reduced. Table 4 shows that certain L. monocytogenes isolates were resistant to clindamycin (3 cases out of 7, 42.9%). The high prevalence of the clindamycin-resistant L. monocytogenes isolates is consistent with a previous report (Wieczorek et al., 2012). Another study also showed that 35% of the L. monocytogenes isolates from poultry, pork, and beef were resistant to clindamycin (Gómez et al., 2014). Clindamycin binds to the ribosome (50S subunit) of bacteria and inhibits synthesis of protein (Depardieu et al., 2007). The results of this study suggest the emergence of multi-resistant strains in smoked ducks, representing a potential threat to human health.
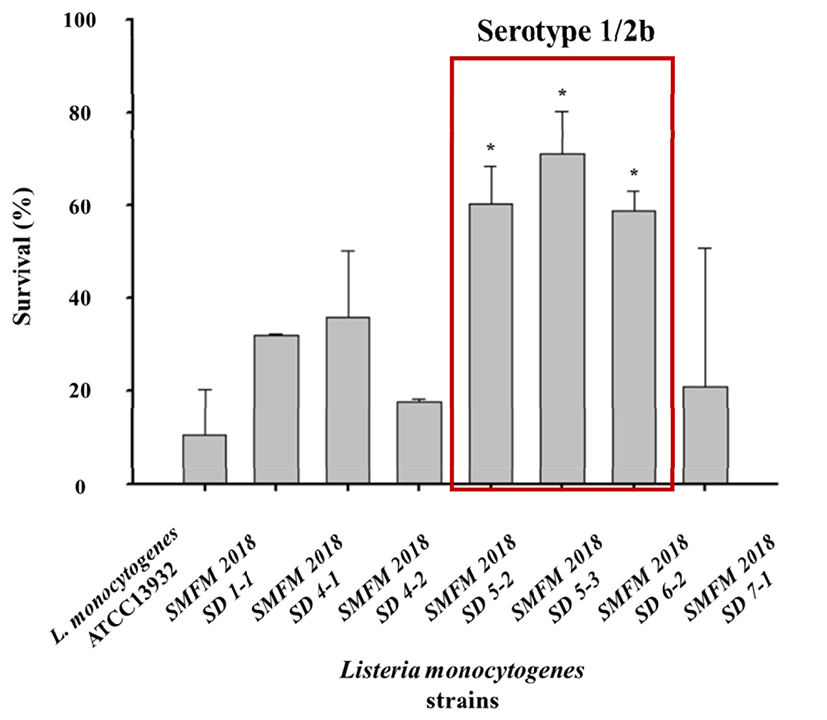
In this study, five isolates showed thermal resistance, compared to the standard strain L. monocytogenes ATCC13932, after 60°C of heat exposure for 10 min, and three of these isolates decreased to less than 1.7 Log CFU/mL (63.6% survival) (p<0.05), and they all represented serotype 1/2b (Fig. 3). This result suggests that the L. monocytogenes serotype 1/2b exhibits high heat resistance at 60°C, compared to other serotypes. Such thermal tolerance at 60°C has also been observed in the case of the serotype 1/2b among 13 L. monocytogenes serotypes (Shen et al., 2014). The L. monocytogenes D-value at 60°C was reportedly 3.10 min (Dogruyol et al., 2020). However, the D-values at 60°C of three isolates (SMFM2018 SD 5-3, SMFM2018 SD 6-2, and SMFM2018 SD 7-1) in our study were 5.41, 6.48, and 6.71, respectively (p<0.05), as summarized in Table 5. Therefore, compared to the other studies, these L. monocytogenes isolates exhibited higher heat resistance.
Discussion
In conclusion, this study showed a high smoked duck product-derived L. monocytogenes prevalence in Korea. In addition, all seven L. monocytogenes isolates, presented in this study, were resistant to penicillin G and certain strains were even resistant to clindamycin as well. These strains possessed the longer, 385-bp amplicon of the actA gene. We found that three of all the isolates were heat resistant and they identified as serotype 1/2b. Thus, 1/2b might exhibit better heat resistance than other serotypes. These results indicated that the risk of L. monocytogenes contamination in smoked ducks could be considered high due to antibiotic resistance, invasion efficiency, and the heat resistance of the 1/2b serotype.













