Introduction
Entomophagy, the consumption or application of insects as food by humans, is increasing due to the need for protein combined with increasing animal protein costs, food and feed insecurity, environmental issues, and population growth (Looy et al., 2014). More than 1,900 species, including beetles, caterpillars (Lepidoptera), bees/wasps/ants (Hymenoptera), grasshoppers/locusts/crickets (Orthoptera), cicadas/leafhoppers/planthoppers/scale insects/true bugs (Hemiptera), termites (Isoptera), dragonflies (Odonata), and flies (Diptera), have traditionally been used as food sources in some parts of the world (van Huis et al., 2013). Insects are a valuable sustainable food with ample energy content and high levels of protein, beneficial amino acids (leucine, isoleucine, and lysine), and unsaturated fatty acids (oleic acid, linoleic acid, and palmitic acid), minerals (copper, iron, magnesium, manganese, phosphorous, selenium, and zinc), and essential vitamins (vitamins A, B complex, and C) (Bukkens, 1997; Finke, 2002; Murefu et al., 2019; Tang et al., 2018). It was recently estimated that at least 2 billion people consume insects on a regular basis (van Huis et al., 2013) because of their nutritive value and their characteristic flavor.
Tenebrio molitor, whose larvae are known as mealworms, is a species of darkling beetle popularly consumed in Asia (Pal and Roy, 2014; Zhang and Zhao, 2019). It was recently accepted as a food ingredient by the Ministry of Food and Drug Safety in Korea, following promulgation of the Act on Fosterage and Support of the Insect Industry in 2010 (Yun and Hwang, 2016). Numerous studies have examined the nutritional components of mealworms, and their safety, functionality, and utilization (Baek et al., 2019; Borremans et al., 2018; Francis et al., 2019; Murefu et al., 2019; Parodi et al., 2018). However, the use of mealworms, like most edible insects, as a food source remains somewhat limited because many people are averse to their physical appearance. Therefore, most insects have been used in crushed or powdered forms as food additives (Seo and Cho, 2018). They can also be used as the main ingredients of seasoning or flavoring materials after suitable reaction processes, because they are rich reservoirs with suitable aroma characteristics (Finke, 2002; Parodi et al., 2018). Therefore, basic information on their aroma profiles is necessary before their application. Recently, Kroncke et al. (2019) compared the nutrient qualities and volatiles of mealworms using different drying methods. However, there is still little information on their aroma profiles either raw or after cooking.
Therefore, this study profiled the aroma components and sensory attributes of mealworms, comparing the aroma characteristics of raw and cooked samples, and examined the differences in their aroma properties according to cooking methods (i.e., raw, steamed, roasted, and fried).
Materials and Methods
Live mealworms at the 9th larval stage (under non-fasting condition) were purchased from a local market (MG-Natural, Damyang, Korea), separated from wheat bran, and stored in a deep-freezer at −70°C until use. Raw and cooked mealworms were prepared (i.e., steamed in boiling water for 30 min, roasted in a convection oven (EDF 213 XPT, ESCO, Eskişehir, Turkey) at 180°C for 5 min, or deep-fried (DKR-113, Delki, Goyang, Korea) in soybean oil at 180°C for 3 min) and then placed in a mortar, frozen in liquid nitrogen, and ground to powder.
Ground mealworms (30 g) were mixed with 0.1 mL of benzyl acetate (200 ppm in diethyl ether) as an internal standard and extracted with 150 mL of re-distilled diethyl ether at 300 rpm for 30 min. They were separated using a centrifuge (Combi-514R, Hanil, Gimpo, Korea) at 1,000×g and 4°C for 10 min, and the supernatants were filtered under a vacuum. Aroma components were then separated from the non-volatiles by solvent-assisted flavor evaporation (SAFE) with an operating vacuum that was typically below 5×10−5 Torr. The extract was dehydrated over anhydrous sodium sulfate, evaporated on a Vigreux column in a water bath at 40±2°C, and then concentrated under a slow stream of nitrogen gas to obtain a final volume of 0.1 mL.
GC-MS analysis was performed using an Agilent 7980B gas chromatography-5977B mass selective detector (Agilent Technologies, Palo Alto, CA, USA) equipped with an HP-5MS column (30 m length×0.25 mm i.d.×0.25 mm film thickness). The carrier gas was helium at a constant flow rate of 0.8 mL/min. One microliter of the extract was injected in the split ratio (10:1) mode. The oven was started at 40°C for 5 min, increased to 200°C at a rate of 4°C /min, and held at 200°C for 10 min. The injector and detector temperatures were 250°C and 230°C, respectively. The mass detector was operated in electron ionization mode with an ionization energy of 70 eV and a scan range of 50 and 550 a.m.u.
Aroma components were identified based on comparison of their mass spectra with those of the NIST 17 (ver. 2.2) and Wiley 7.0 databases or by manual interpretation. Retention index (RI) values were compared with those reported previously (Adams, 2007; Babushok et al., 2011; Bianchi et al., 2007; Kang et al., 2016). The RIs of volatiles were calculated using n-alkanes (C7–C22) as external references. Semiquantitative analysis of aroma components was performed by comparing their peak areas to that of the internal standard compound (0.1 mL of 200 ppm benzyl acetate in diethyl ether, v/v) on the GC-MS total ion chromatogram.
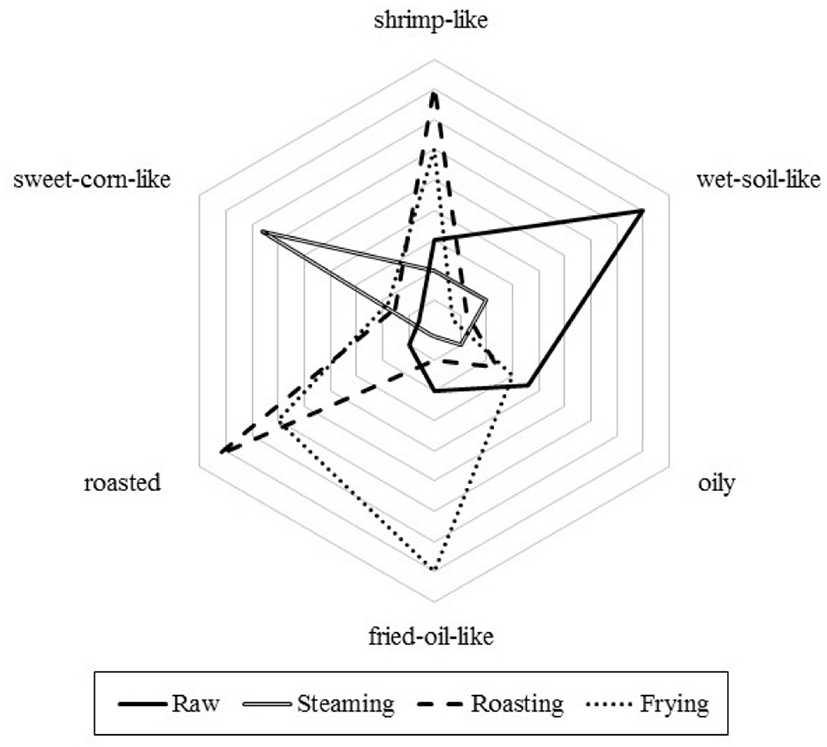
To evaluate sensory aroma attributes perceived in raw and cooked mealworms, sensory aroma profiling modified from flavor profiling (Stampanoni, 1994) was performed (IRB No. WKIRB-201903-HR-014). Ten subjects (female, 21–25 years of age) who previously participated in descriptive analyses in the Department of Food Science and Biotechnology at Wonkwang University were selected and trained until they could reliably discriminate among aroma attributes: shrimp-like (aroma associated with shrimp snacks), wet-soil-like (aroma associated with damp soil), oily (aroma associated with fresh olive oil), roasted (aroma associated with baked cookies), sweet-corn-like (aroma associated with sweet corn), and fried-oil-like (aroma associated with oil fried once or twice). Each subject was given 5 g of each sample in a porcelain container (8 cm i.d.×4.5 cm height). All samples were coded with random three-digit numbers. The intensities of attributes perceived in each sample were evaluated on a 9-point intensity scale, ranging from “weak” (score of 1) to “strong” (score of 9).
Analysis of variance (ANOVA) using SPSS software (ver. 24.0; IBM, Armonk, NY, USA) was performed for statistical evaluation of the differences in the aroma compositions of raw mealworms and those treated with different cooking methods. The results of Duncan’s multi-range test were evaluated at p<0.05 significance level. The values of aroma components are presented as the mean±SD of three replicates. Partial least squares regression (PLSR) was used to determine the relationship between instrumental and sensory data sets, performed with SIMCA-P (ver. 11.0; Umetrics, Umeå, Sweden).
Results and Discussion
The aroma components of raw and cooked mealworms were extracted using SAFE and then analyzed by GC-MS. Table 1 lists the aroma components identified in mealworms, their relative peak areas, and their RIs on the HP-5ms column. In total, 46 aroma components were found in the mealworms, including four pyrazines, three pyrrolidines, three aldehydes, six ketones, five alcohols, three acids and esters, and 22 hydrocarbons. The aromas of raw mealworms were primarily composed of hydrocarbons (50.11%) and aldehydes (37.14%). In particular, 4-methylbenzaldehyde (no. 10) was the most prevalent individual component in raw mealworms and increased in steamed and roasted mealworms. Pyrazines, pyrrolidines, and carbonyls were increased or newly appeared in cooked mealworms (especially with roasting and frying), although there were some quantitative and qualitative differences among cooking methods. These components can be generated through a complex series of thermal reactions, including Maillard or non-enzymatic browning reactions between reducing sugars and amino acids, thermal degradation of lipids, and decomposition of sugars, and their interactions, which then produce the characteristic flavors differentiated from those of raw mealworms (Amrani-Hemaimi et al., 1995; Schenker et al., 2002).
3) There are significant differences (p<0.05) among samples according to heating methods by using Duncan’s multiple comparison test between the samples having different letter in low.
In this study, ten trained panelists described the sensory aroma attributes of raw and cooked mealworms. Six sensory aroma attributes were perceived in raw and cooked mealworms: oily, shrimp-like, wet-soil-like, roasted, sweet-corn-like, and fried-oil-like aroma attributes; the mean intensities of each attribute are shown in Fig. 1. The intensities of the attributes differed significantly among mealworms (p<0.05). Raw mealworms had a strong wet-soil-like odor and less-intense oily, shrimp-like, and sweet-corn-like aroma attributes. In comparison, steamed mealworms had a strong sweet-corn-like aroma attribute, and weak shrimp-like and wet-soil-like aroma attributes. The intensities of two sensory attributes (e.g., roasted and shrimp-like) were noticeably increased in roasted mealworms, whereas strong fried-oil-like and roasted aroma attributes were major in fried mealworms.
Multivariate statistical techniques can be used to extract, organize, and visualize statistically interpretable and reliable data information. In particular, PLSR can be used to establish the relationship between two datasets by predicting one (X) from the other (Y) (Dijksterhuis, 1994). In this study, the PLSR model calculated the cross-validation based on 46 aroma components (x variable) and six sensory aroma attributes (y variable) analyzed in raw and cooked mealworms and then visualized the differences among raw and cooked mealworms (Fig. 2). It is also possible to understand which variables carry class-separating information and which variables are mainly associated with samples (Ledauphin et al., 2010; Zhang et al., 2013). Fig. 2 shows the distributions of raw and cooked mealworms based on the first and second PLS components of the PLSR plot. Raw and steamed mealworms were located on the negative PC 1 axis, whereas roasted and fried ones were on the positive PC 1 axis. Fried vs. roasted and raw vs. steamed mealworms were further separated along the PC 2 axis. Fried and raw mealworms were located on the positive PC 2, whereas steamed and roasted ones were on the negative PC 2 axis. The first PLS component was mainly defined by the aroma descriptors and contrasted shrimp-like, fried-oil-like, roasted, and oily aroma attributes on the positive dimension and wet soil-like and sweet-corn-like aroma attributes on the negative dimension. In particular, fried and roasted mealworms were evaluated as possessing strong shrimp-like, roasted, and fried-oily like attributes. By contrast, wet soil-like and sweet-corn-like attributes were strongly correlated with raw and steamed mealworms, respectively. In addition, 2,3-dimethyl-heptane (no. 28), bis(2-methylpropyl)-hexanedioate (no. 22), 2-butoxy-ethanol (no. 18), and ethyl-benzene (no. 29), were related to wet-soil-like aroma attributes, which were close to raw mealworms. In comparison, 2,4,6-trimethyl-heptane (no. 32), 2,4-dimethyl-dodecane (no. 42), and 3,5-dihydroxy-6-methyl-2,3-dihydropyran-4-one (no. 14) were associated with sweet-corn-like odor notes. 2,6-Dimethyl-pyrazine (no. 1), heptan-2-one (no. 11), 2,3,5-trimethyl-pyrazine (no. 4), and 1-butyl-pyrrolidine (no. 5) were correlated with the sensory attribute of shrimp-like odor, whereas 1-(1-pentenyl)-pyrrolidine (no. 7), pentan-1-ol (no. 17), oct-2-ene (no. 26), 1-(2-methyl-1-butenyl)-pyrrolidine (no. 6), 2,6,11-trimethyl-dodecane (no. 45) were responsible for fried-oil-like aroma characteristics. 2,5-Dimethyl-pyrazine (no. 2), oct-1-one-3-ol (no. 18), 2-ethyl-5-metyl-pyrazine (no. 3), and benzaldehyde (no. 8) were associated with roasted sensory attributes. In general, raw meat (or seafood) has little aroma and only blood-like (or fresh fish-like) taste. However, it has lots of nonvolatile precursors of muscle flavor including free amino acids, peptides, reducing sugars, vitamins, and nucleotides. The interaction of nonvolatile precursors with one another and/or their degradation products, via the Maillard reaction and lipid degradation during thermal processing, produces a large number of aroma components which contribute to the development of desirable aroma of cooked meat/or seafood (Shahidi, 1998). In particular, pyrazines, pyridines, thiophenes, thiazoles, thiazolines, and Strecker aldehydes which contributing to the roasted aroma characteristics derive from the Maillard reaction, whereas those responsible for species aroma characteristics are formed from lipid degradation (alcohol, aldehyde, ketones, and furans) (Amrani-Hemaimi et al., 1995; Schenker et al., 2002; Shahidi, 1998). In our study, pyrazines, pyrrolidines, and aldehydes were produced in cooked mealworms. They have been known as major savory-type (e.g., meaty, roasted, baked, popcorn-like, and so on) aroma components (Amrani-Hemaimi et al., 1995; Schenker et al., 2002; Shahidi, 1998). However, pyrazines were dominant only in roasted and fried mealworms, but not detected in boiled ones. According to previous study, the substituted pyrazines were main products from thermal reactions (especially Maillard reaction), and decreased as the water content increase (Eichner and Karel, 1972; Lu et al., 2005). In addition, Mottram (1994) reported that high temperatures for cooking lead to high formation rate of the rapid oxidation of unsaturated fatty acids. The major lipid oxidation components (e.g., pentan-1-ol and oct-1en-3-ol) in our study were also more found in roasted and deep-fried ones.

Conclusion
The sensory aroma attributes of raw mealworms had strong wet-soil-like notes and less intense oily, shrimp-like, and sweet-corn-like notes. Hydrocarbons with solvent-like aroma note and aldehydes described as sweet and roasted aroma characteristic were the major aroma components in raw mealworms. The cook-treatments (e.g., steaming, roasting, and frying) increased the intensities of roasted, fried-oil-like, and sweet-corn-like aroma attributes of mealworms. Specifically, nitrogen-containing heterocyclic components (e.g., pyrazines and pyrrolidines) and carbonyls contributed to those aroma attributes and were associated with their differences. Raw mealworms are rich in Maillard reaction precursors and their changes in the aroma compositions during thermal processing were similar to those of meat or seafood. Therefore, basic information on their aroma profiles should be valuable in the application of flavoring materials as main ingredients.