Introduction
Research on metabolic disorders has been gaining attention over the past decade (Chien et al., 2013; Colussi et al., 2017; Das et al., 2003). Fatty acids (FAs) have an important role in regulating biological metabolism and maintaining homeostasis in animals (Calder, 2016; Palmquist, 2009; Pawlosky and Salem, 2004) and are categorized as either saturated or unsaturated. Ingested triacylglycerol (TG) is hydrolyzed into FAs and glycerol, either to yield energy or to be stored as lipid droplets depending on the metabolic status of the animal (Carlier et al., 1991).
The main function of adipose tissue is to provide energy storage, thermal insulation, and cushioning to protect vital organs from mechanical shock (Lone et al., 2016). Additionally, adipose tissue also functions as connective tissue as well as an endocrine organ, secreting cytokines and hormones which regulate the metabolic activities of the whole animal system (Sun et al., 2011). The 3T3-L1 cells are widely used in biological studies on adipose tissue-particularly the lipid metabolism pathway. There are different stages of adipocytes-spanning from precursor cells to specialized mature adipocytes-which is regulated by adipogenic gene expression. The CCAAT/enhancer-binding protein family (C/EBPα), nuclear receptor peroxisome proliferator-activated receptor γ (PPARγ), fatty acid synthase (FASN), sterol regulatory element-binding protein 1 (SREBP-1), and carnitine palmitoyl transferase 1 (CPT-1) genes are associated with differentiate stages of adipocyte cells, and their expression levels are precisely regulated (Barber et al., 2013). Some in-vitro and in-vivo models demonstrate that PPARγ, C/EBPα and FASN gene regulation plays a significant role in white adipose tissue by impacting lipid formation and lipid storage, contributing to increased risk factors for obesity and its related metabolic disorder diseases (Ailhaud et al., 2008; Cooke et al., 2016; Simopoulos, 2016; Vecchione et al., 2016). Other metabolic pathways such as glucose and insulin regulation are also associated with the adipogenesis program of animal fat cells. Although studies of individual edible FAs have reported both benefits and disadvantages to health function, other forms obtained by plants and animals such as FA esters-the main composition in edible waxes-have not yet been defined with their precise modes of action.
Wax is an organic material which is composed of FA esters and primary and/or secondary long chain alcohol groups (Limpimwong et al., 2017). Natural waxes obtained from various plants and animals are being formulated as oleogels (a complex of wax and plant oils) and used to substitute saturated solid fat with unsaturated solid fat in food products (Fayaz et al., 2017). Lastly, oleogels became a food additive primarily due to its advantageous physical properties (e.g., as a gelling agent, thickening agent, fat replacer, etc.). The EFSA (2012) recommended a maximum permitted level of natural waxes (e.g., carnauba wax) for coating foodstuffs from 200 to 1,500 mg/kg of food to maintain an adequate safety level and no genotoxicity effect. Meanwhile, beeswax can also be used in formulating oleogels and is frequently used to maintain moisture and improve the texture of baked goods. The EFSA Journal (EFSA, 2007) established a no observed adverse effect level (NOAEL) for short-term BW application of 500–10,000 mg/kg using the rats feeding model. Several bakery products (Patel et al., 2014), chocolate (Doan et al., 2016), ice cream (Zulim et al., 2013), margarine (Hwang et al., 2013; Limpimwong et al., 2017), and instant fried noodles (Lim et al., 2017) have been innovatively developed as functional food products utilizing oleogels. Moreover, oleogels can also be applied in meat products because of its beneficial roles in physical and sensory quality, as well as in improving marbling and healthiness of meat products. In 2018, Wolfer et al. (2018) reported that oleogels mixed with soybean oil and rice bran wax can substitute pork fat as an ingredients added in frankfurter-sausages. Oleogel containing sausage product showed healthier FA profiles by reducing the amount of saturated FAs, which can reduce the risk of metabolic diseases such as obesity, diabetes or atherosclerosis. Health functionality of oleogels in combination with monoglycerides and phytosterols or with BW and ethyl cellulose has been described in bologna-type sausages and in healthier lipid pâtés (de Silva et al., 2019; Gomez-Estaca et al., 2019; Kouzounis et al., 2017). Although oleogels have been used as an ingredient in the production of various meat products, their therapeutic effect in preventing metabolic disorders, toxicity and side effects have not yet been evaluated.
Furthermore, additional evidence is needed to support the health effects of FAs, as there have been no studies to identify the effect of molecular regulation of adipogenic activity by individual natural waxes. Therefore, the purpose of this study was to investigate the effects of different types of FAs [palmitic acid (PA), stearic acid (SA), oleic acid (OA), linoleic acid (LA; ω-6) and alpha-linolenic acid (ALA; ω-3)] and two edible waxes [beeswax (BW) and carnauba wax (CW)] on adipogenesis, and their capacity of lipid accumulation combined with related gene expression profiles in in-vitro using the 3T3-L1 cell line model.
Materials and Methods
Determination of the wax concentration was based on the previous in vivo studies by Limpimwong et al. (2017), which were performed using rice bran wax at 1.5% (w/w) in the animal diet. So, the appropriate concentration (in ppm) of BW and CW was then applied to the 3T3-L1 cells in our study.
The dietary FAs including PA (C16:0), SA (C18:0), OA (C18:1), LA (C18:2), ALA (C18:3); dexamethasone (DEX); 3-isobutyl-1-methylxanthine (IBMX); insulin; low endotoxin FA-free bovine serum albumin (BSA); Oil Red O powder; and 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) powder were purchased from Sigma Chemical (St. Louis, MO, USA). Other reagents such as fetal bovine serum (FBS) (Gendepot, Barker,TX, USA), Dulbecco's modified eagle's medium (DMEM) (Gibco-BRL, Grand Island, NY, USA), antibiotic-antimycotic (Gibco-BRL, Grand Island, NY, USA), Dulbecco’s Phosphate Buffered Saline (DPBS) (Wel gene Fresh MediaTM Co., Gyeongsan, Korea), and formaldehyde (Junsei Chemical, Tokyo, Japan) were purchased through the agency company in Korea as mentioned above. BW (Hooper pharm GmbH Co., Hamburg, Germany) and CW (Starlight Co., Fortaleza, Brazil) were purchased from the commercial sources.
FAs complexed with BSA were prepared following the method of Wu et al. (2014). All the FA powder was dissolved in ethanol at 70°C to a concentration of 10 mM and used as the stock solution. Subsequently, the FA solution was diluted with 10% BSA and adjusted to the following concentrations: 25, 50, 100, 150, and 200 μM to form FA-BSA complexes. After incubation at 37°C for 1 h with constant shaking, the FA-BSA complexes were cooled at room temperature, filtrated through a membrane filter (0.45 μm), and stored at –20°C for further use.
The BW and CW were prepared and modified following the method of Lim et al. (2017). In brief, both waxes were dissolved in ethanol to make a 0.03% (w/w) wax stock solution. They were then heated to 80°C–90°C under magnetic stirring and kept in a water bath at 70°C for 20 min. The solution was filtered using 0.45 μm syringe filters (tubes and syringes warmed in the water bath at 70°C). From the stock solution, the working concentrations were prepared at 0.5, 1.0, 1.5, 2.0, 2.5, and 3.0 ppm and kept in a warm condition (about 40°C) before use, to prevent the coagulation of the wax and liquid phase. The concentration of both waxes was converted from % (w/w) from the in-vivo model study of Limpimwong et al. (2017) to ppm for our in-vitro model determination.
Cell culture and differentiation of the adipocyte cell line followed the method used by Kim and Jang (2014) with some modifications. In brief, the 3T3-L1 mouse embryo fibroblasts were obtained from the Korean Cell Line Bank (Seoul, Korea). Growth media for the cell culture was composed of 89% DMEM, 10% FBS and 1% Antibiotic-antimycotic (%, v/v). Cells were cultured in the incubator (ThermoFisher Scientific, FORMA STERICYCLE i160, Langenselbold, Germany) under 5% CO2 at 37°C. After the 3T3-L1 culture reached >90% confluence (Day 2), growth media supplemented with 0.5 mM IBMX, 1 μM DEX, and 1.7 μM insulin solution was applied. From Day 5 to Day 7, differentiation media (containing 1 μM DEX and 1.7 μM Insulin in growth media) was added to the 3T3-L1 cells. Finally, on Day 8 and Day 9, only 1.7 μM insulin in growth media was added to the cell culture until the analysis.
For the FA and wax treatments, stock solutions at different concentrations ranging from 25 to 200 μM and 0.5–3.0 ppm, respectively, were warmed in a water bath and added to the culture medium. The differentiation media was replaced with adipocyte growth media, which was refreshed every 2 days. After 24 h incubation (finished adipogenic differentiation between Day 7 and Day 9), the medium was collected and cells were harvested for the required assays. A FA-free vehicle served as a control (the experiment platform was modified from Kim et al., 2006; Moravcova et al., 2015; Wu et al., 2014).
The toxicity of the 3T3-L1 adipocyte supplemented with FAs and waxes was determined by the MTT assay. MTT stock solution (100 mL; 5 mg/mL in PBS) was diluted 10 times with growth media. The 3T3-L1 (pre-adipocytes) were seeded in a 96 well plate, 104 cells/plate at 37°C for 48 h, and treated with 100 μL of FAs or waxes in each well at different concentrations as prepared. The plates were kept in the incubator for an additional 4 h. After incubation, the solutions were removed, 100 μL DMSO was added to each well, and they were stored for 5 min in the incubator. The absorbance was measured by a spectrophotometer at 540 nm to determine the cell toxicity (Varioskan Flash, ThermoFisher Scientific, San Jose, CA) (Kim et al., 2006).
The lipid accumulation was determined by following the method of Kim et al. (2006) with some modifications. In brief, after nine days of adipogenic differentiation, the cultured cells were washed with DPBS and fixed with 10% formaldehyde for 5 min at room temperature. After the fixing step, cells were washed again, dried at room temperature, and stained with Oil Red O solution (prepared at a 3:2 ratio with distilled water) stored for 1 h without light exposure. For lipid accumulation and droplet morphological observation, the stained cells were washed three to four times with distilled water. The obtained Oil Red O stain was collected and eluted with isopropanol, transferred to a 96 well plate, and quantified by measuring the optical density at 490 nm. The obtained data from three replications were recorded. Finally, an optical microscope (CXX53SF, OLYMPUS, Tokyo, Japan) was used to capture the lipid droplet morphology for further analysis.
For lipid droplet size and area distribution, the Oil Red O stained cells were observed under a microscope at 40X magnification (Life Technologies, Carlsbad, CA, USA). From the center of the plates for every treatment, at least six images were captured which were then adjusted to grayscale. The lipid droplet size was assessed using an ICY software package (Version 1.9.4.1) which included the ImageJ program; the manual guidelines of the software were followed. The grayscale threshold images were set with size index range (the scale ranged from 20 to 220 μm) with the filter. Determination of the lipid droplet size and area distribution was performed and recorded with three replications, and the data were expressed as mean±SD.
Total RNA was isolated from differentiated 3T3-L1 cells using the MagListoTM 5M Tissue Total RNA Extraction Kit (Bioneer Corporation, Seoul, Korea) according to the manufacturer's protocol. The concentration and purity of RNA were determined by Nanodrop ONEc equipment (Thermo Fisher Scientific, Wilmington, DE, USA). Total RNA was converted into cDNA using the reverse transcription kit (AccuPower® CycleScript RT PreMix, Bioneer Corporation, Seoul, Korea) according to the manufacturer's guidelines. Quantitative real-time polymerase chain reaction (PCR) was performed with AccuPower® 2X Greenstar qPCR Master Mix (Bioneer Corporation, Seoul, Korea) by StepOnePlus Real-Time PCR system (Applied Biosystems, Marsiling, Singapore). The reactions were performed in triplicate, in 96-well plates, using the following thermo-cycling condition: initial denaturation at 95°C for 5 min; this was followed by 30 cycles, each consisting of denaturation at 95°C for 15 s and annealing at 57°C to 61°C for 45 s, and a final extension step at 72°C for 45 s. The presence of PCR product was detected above a fixed threshold (the cycle threshold: Ct) and determined for each sample. The primer sequences for real-time PCR were based on previously published sequences as listed in Table 1. Data were analyzed by StepOne Software v2.3 using the ΔΔCT method. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a housekeeping gene (modified from the methods used by Li et al., 2016; Yu et al., 2013).
All determinations were conducted in triplicate and expressed as mean±SD. The collected results were subjected to analysis of variance (ANOVA) with Duncan's multiple range test, and the significant difference was determined as p<0.05 by using SPSS software (SPSS 16.0 for Windows, SPSS Inc., Chicago, IL, USA).
Results and Discussion
The toxicity levels of 3T3-L1 cells treated with FAs and two natural waxes are shown in Fig. 1. For FA supplementation (Fig. 1A), a significant difference in all treatments was observed when compared to the control group (p<0.05). The highest cell viability was seen in the 50 μM ALA treatment, whereas the 200 μM SA and PA treatments presented the lowest cell viability when compared to the control group (p<0.05). The increased dose of FA concentration dramatically reduced the adipocyte cell activity (Fig. 1A). Previous studies were consistent with our findings that an increase of SA concentration reduced cell viability and disturbed the intracellular metabolism pathway of adipocyte cells (Rabkin and Lodha, 2009). Kokta et al. (2008), who studied the lipid accumulation in 3T3-L1 cells by FA treatment, found that a 0.3 mM treatment of linoleic and OA yielded lower cell numbers as compared to other treatments. Moreover, Li et al. (2017) also reported that stearidonic acid (a plant-based omega-3 fatty acid) treatment at 400 μM yielded a significant reduction in adipocyte cell numbers while treatment at 200 μM had no effect. A study by Wu et al. (2015) on the effect of omega-3 free FAs [eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and ALA] on tamoxifen-induced cell apoptosis demonstrated that individual ALA-treated cells at a concentration of 200 μM significantly reduced MCF-7 cell proliferation as compared to DHA and EPA treatments at the same concentration. However, these differences may be caused by the effect of the above omega-3 FAs on various cytokines, which influence mechanisms such as lipogenesis, lipolysis, and proteolysis, consequently leading to an unusual proliferation of cells as well as cell apoptosis (Wu et al., 2015). Hence, it is possible to conclude that the quantity of 3T3-L1 cell proliferation and migration was FA dose-dependent. Our results strongly suggest that cells treated at a higher concentration led to higher cell apoptosis. According to the results (Fig. 1A), the 200 μM FA treatments in our study had a significant impact on the reduction of cell proliferation, especially within the treatment groups of saturated FAs, when compared with other treatment groups. Consequently, this concentration was used to determine the lipid accumulation, along with lipid droplet size and area distribution, of adipose tissue cells for further analysis.
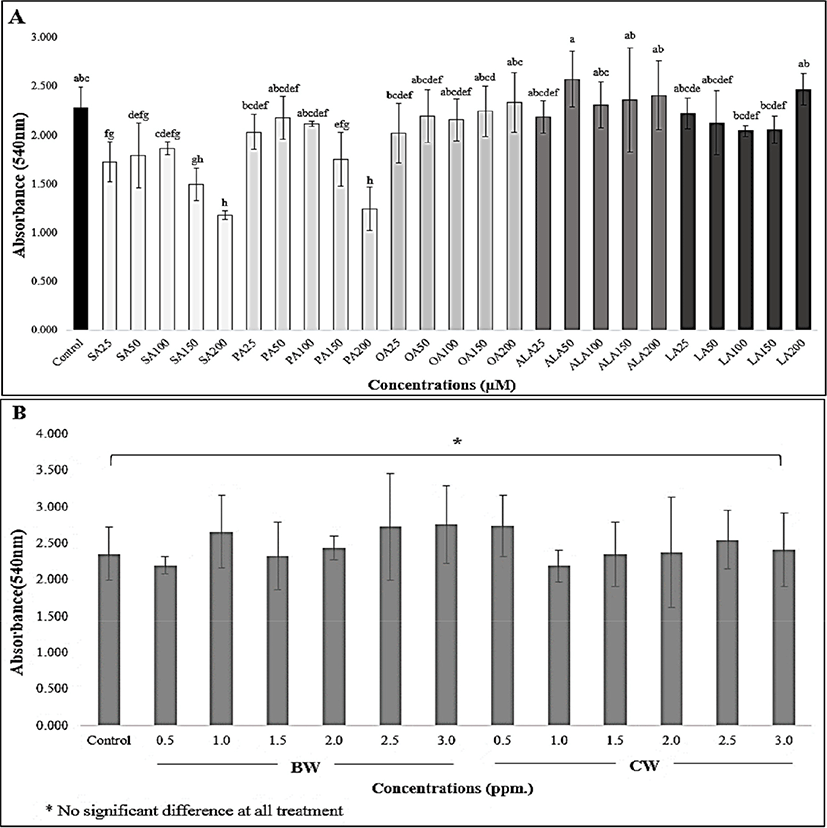
To identify the toxicity effect of BW and CW on 3T3-L1 preadipocytes, the prepared concentrations of these waxes were supplemented for 48 h, and the cell toxicity was quantified. Our study shows that there was no toxicity observed in the BW and CW treatments at all dose levels when compared to the control group (p>0.05), which is presented in Fig. 1B. Consequently, for further quantification of lipid assembly in 3T3-L1 adipocytes, the working concentrations of both waxes were performed at 1.0, 2.0, and 3.0 ppm.
In general, preadipocytes differentiate into mature adipocytes by the process of lipid accumulation together with other mechanisms (e.g., FA metabolism pathway or gene expression in the cell). Thus, the lipid droplet size and area distribution are important parameters that have often been studied after FA treatment. The control and FA-treated 3T3-L1 cells (both at 50 μM and 200 μM concentrations) were collected and stained with Oil Red O staining solution for 1 h. The effect of FAs on lipid accumulation is shown in Fig. 2. A significant increase in lipid accumulation was observed in all treatments when compared to control (p<0.05). The SA group showed a high capacity of lipid hoarding in 3T3-L1 cells compared to the remaining groups of saturated FAs, while ALA was the lowest (p<0.05). The mono- and polyunsaturated FAs (OA, LA, and ALA) reduced the lipid accumulation in adipocytes. Our study found consistent results with Barber et al. (2013) who reported that polyunsaturated FA (PUFA) treatments (including EPA, DPA, and DHA) significantly reduced the lipid accumulation capacity as well as lipid droplet size. Additionally, Madsen et al. (2005) and Kokta et al. (2008) found a similar trend on adipogenesis of 3T3-L1 cells treated with omega-3, -6 and -9 FAs. The high lipid accumulation in preadipocytes may cause obesity and its related metabolic disorders (Kang et al., 2016; Kong et al., 2017; Park and Sung, 2015). However, these findings did not clearly explain the effect of FAs on adipogenesis activity. According to previous study, which compared OA and PAs on lipid accumulation of hepatocytes (Ricchi et al., 2009), OA is more steatogenic (but less apoptotic) than PA in the cultured liver cell. Furthermore, rates of net uptake of FAs into adipose tissue were not significantly different in serum biochemical analysis from healthy men who had intake the diet containing either OA, PA, or SA (Summers et al., 2000). Therefore, OA supplement in our cell culture model may affect the lipid holding capacity or adipocyte remodeling program of 3T3-L1 cells. Consequently, our study aims to identify this phenomenon through the underlying molecular mechanisms, such as the expression of gene-related adipogenesis, which is presented in the next part.
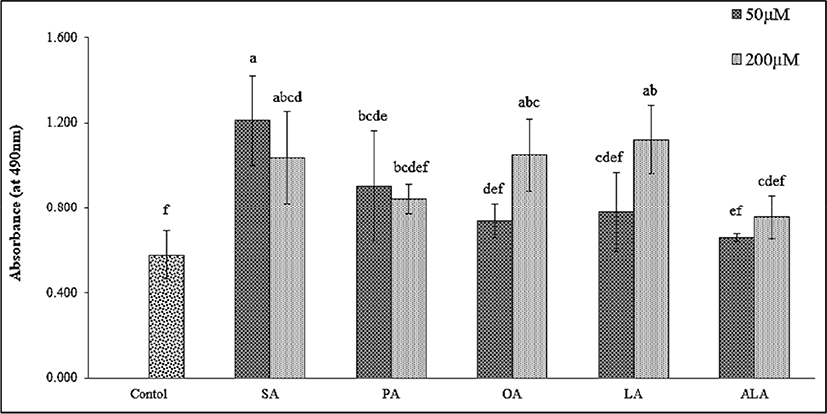
Fig. 3 illustrates the effect of FAs on lipid droplet size and area distribution in adipocyte cells. The two-dimensional images obtained from the microscopic evaluation of lipid morphology were collected and analyzed. The group of PA-treated cells had the highest average lipid droplet size (182.68±10.09 to 187.55±2.71 μm) and percentage of area distribution (11.93±1.69 to 16.19±0.75%). Interestingly, the measurements of the 200 μM LA treatment group (181.84±5.87 μm, 14.15±0.71%) and 50 μM OA treatment group (176.85±2.43 μm, 10.90±1.29%) were higher than the SA treatment group (102.67±2.54 to 126.24±5.79 μm, 12.41±0.99 to 12.96±0.81%) which was in contrast to triglyceride accumulation. It is possible that this finding is associated with lipid droplet size regulation and other related pathways such as neutral lipid synthesis and atypical lipid droplet fusion, which control both lipid droplet growth and storage (Camp et al., 2002; Prostek et al., 2016; Sampath and Ntambi, 2004). On the other hand, a higher amount of ω-6 PUFA supplementation might induce lipid hoarding in adipocyte cells, contributing to obesity (Madsen et al., 2005). Sears and Ghosh, (2016) and Ortega et al. (2016) also mentioned that an excessive amount of ω-6 PUFA intake increases the risk factor of cardiovascular disease in in-vivo studies via the inflammatory effect of endothelial cells. Meanwhile, Kim et al. (2006) noted that DHA treatment (25–200 μmol/L) in 3T3-L1 cells decreased the average lipid droplet size and percentage of lipid area in a dose-dependent manner. Previous studies have reported that natural plant products, or metabolites suppress lipid droplet accumulation by regulating adipogenesis-related transcription factors such as PPARγ and SREBP-1c (Prostek et al., 2016). Many similar studies on anti-lipid droplet formation and accumulation during adipogenesis have also been described (Hasegawa et al., 2017; Manickam et al., 2010; Park and Sung, 2015; Sears and Ghosh, 2016).
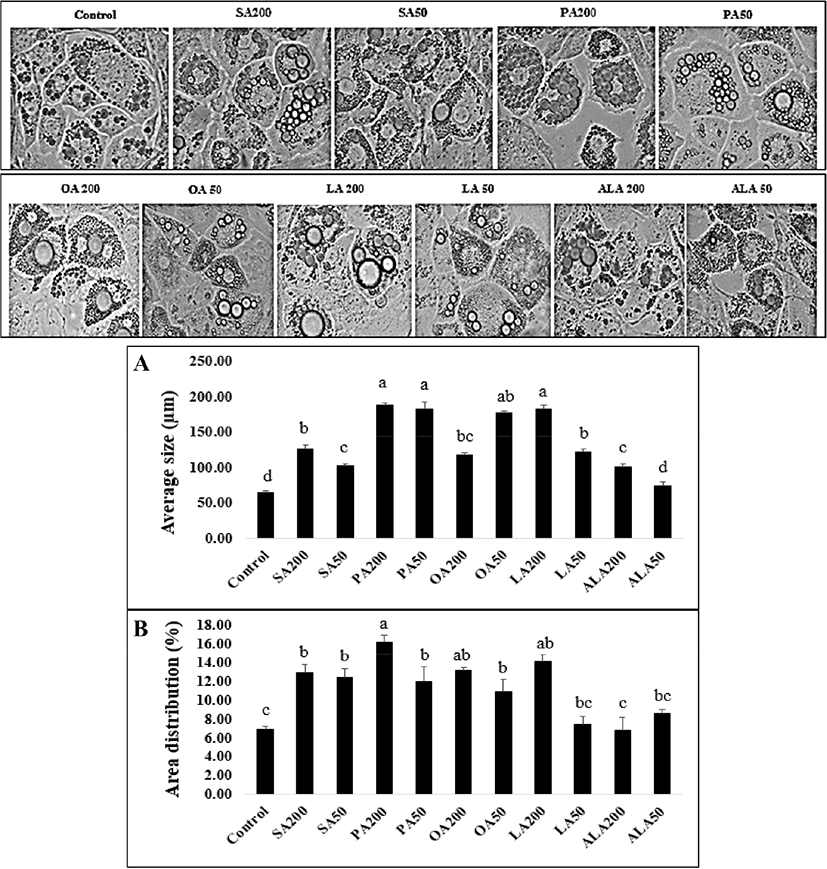
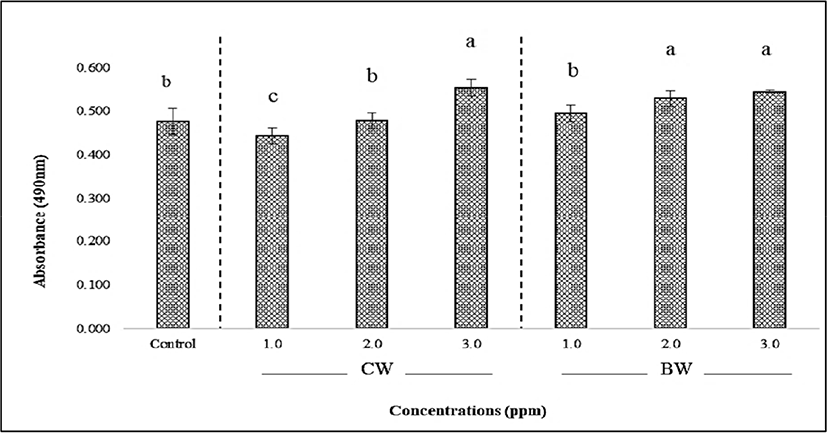
The effect of BW and CW on lipid accumulation in 3T3-L1 adipocytes is shown in Fig. 4. The CW- and BW-treated 3T3-L1 cells were tested at three concentrations as mentioned in the toxicity test method. An increased concentration resulted in higher lipid accumulation in the 3T3-L1 cells. The lowest absorbance was observed at 1.0 ppm of CW (0.443) while the highest (0.553) was at 3.0 ppm. There was no significant difference observed between the highest concentrations both BW and CW treatment groups (p>0.05), but a significant increase was observed when compared with the control group (p<0.05). The effects of BW and CW on lipid droplet size morphology and area distribution in 3T3-L1 adipocytes were illustrated in Fig. 5. The average lipid droplet size in both BW and CW treatment groups showed a tendency to decrease in an inverse dose-dependent manner. Additionally, BW showed a smaller size overall than CW. Lipid droplet size decreased when the concentration of waxes increased, contrastingly, while still exhibiting a high percentage of lipid area distribution (Fig. 5) or storage (Fig. 4). This may result from mechanisms of the lipid droplet size regulator, ppl-GAL4, which specifically expresses GAL4 in the adipose tissue. It may also be associated with MRT function which regulates the lipid droplet size through transcriptional repression of plin1 (Yao et al., 2018). Although the effects of individual BW and CW on lipid droplet size and area distribution in adipocytes have not been documented yet, based on our results, it is possible to suggest that the dose-dependent effects of both CW and BW treatments influence these parameters in 3T3-L1 cells through the adipogenic differentiation process. The shrinkage of lipid droplet size is caused by the degradation of neutral lipids under energy demanding conditions and is regulated by neutral cytosolic lipases and lysosomal acid lipases. In adipose tissue and hepatic cells, the cytoplasmic lipid droplets-phospholipids and proteins-are related to lipid droplet size morphology (Yu and Li, 2017). Moreover, TG-synthesis enzymes (GPAT, AGPAT, and DGAT) and lipases are suggested to control lipid droplet size by increasing or decreasing TG synthesis and hydrolysis, respectively (Cohen et al., 2015). In light of these findings, it is reasonable to assume that the supplementation of BW and CW plays a vital role in the regulation of lipid droplet size, but not area distribution or lipid storage in 3T3-L1 cells. Although a low concentration of waxes has been reported as safe by EFSA (2007 and 2012), the effect of these edible waxes on the endogenous molecular mechanism in 3T3-L1 adipocytes cells has not been reported. Therefore, to better understand these effects, we determined the expression (mRNA level) of adipogenic/lipogenic transcriptional factors related to adipogenesis in 3T3-L1 cells supplemented with both waxes.
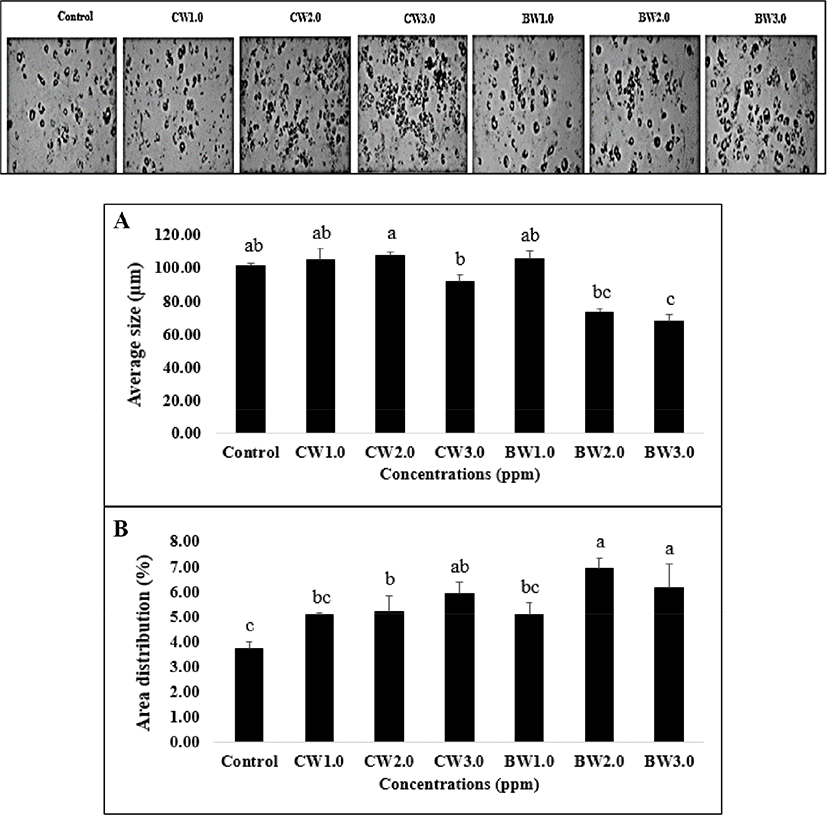
We found a significant reduction in lipid accumulation, lipid droplet size, and area distribution in the low dose FA-treated cells, and found a significant increase of lipid hoarding together with area distribution-but not droplet size-with a high dose treatment of waxes in 3T3-L1 cells. Hence, to understand the effect of FAs and natural waxes on adipogenesis, the regulation of mRNA expression was observed in 3T3-L1 cells. The effects of saturated and unsaturated FAs (SA and ALA) and both waxes, at concentrations of 50 μM and 3.0 ppm, respectively, were selected and assessed. The effect of SA, ALA, BW, and CW on adipogenic transcription factor gene expression was presented in Fig. 6. In the FA-treated cells, the SA group had a higher level of FASN, SREBP-1, C/EBPα, and PPARγ expression when compared to ALA, but not CPT-1. In fact, the CPT-1 gene plays a vital role in increasing mitochondrial activity as well as fatty acid beta-oxidation in adipocytes (Vankoningsloo et al., 2005; Wakil and Abu-Elheiga, 2009), and is considered the counterpart of the FASN pathway-which influences the lipid hydrolysis and lipid hoarding capacity in adipose tissue. Our findings demonstrated that the blocking of CPT-1 mRNA expression by SA treatment led to a significantly higher stimulation of FASN mRNA expression level, which induced lipogenesis. This finding supports the triglyceride accumulation in adipocytes (Fig. 2). According to Wojcik et al. (2014), the presence of SA in 3T3-L1 cells resulted in an increased level of FASN gene expression, while the ALA-stimulated cells showed a similar lower level compared to our present study (Fig. 6B). Guo et al. (2005) reported that beta-oxidation in 3T3-L1 adipocytes increased after treatment with EPA at 100 μM for 24 h. Moreover, EPA and DHA were also noted to activate 5' adenosine monophosphate-activated protein kinase (AMPK) in adipose tissue, which is obtained from FA oxidation mechanism effects (Lorente-Cebrian et al., 2009). A possible reason for EPA and DHA resulting in increased beta-oxidation in 3T3-L1 adipocytes may have to do with their interaction with thermogenesis-mediated mechanisms by uncoupling protein 1 (UCP-1). However, further investigation is needed on the mitochondrial UCP-1 pathway to confirm this mechanism. Moreover, these transcription factors should be investigated in a comprehensive study to confirm the regulation of the beta-oxidation process. Even though there is no report of any organic compound, such as FA esters or long-chain fatty alcohols like natural waxes, on fat cell activity, a comparison of the results between BW and CW in our experiment implies that BW treatment shows a higher CPT-1 mRNA expression level than CW treatment, but without a significant difference (p>0.05). The FASN mRNA level of both BW and CW also showed a strong correlation to the CPT-1 pattern. After interpreting these results, it can be suggested that ALA and natural waxes have anti-lipogenic as well as anti-adipogenic effects and induce the expression of the CPT-1 gene (Fig. 6A and 6B).
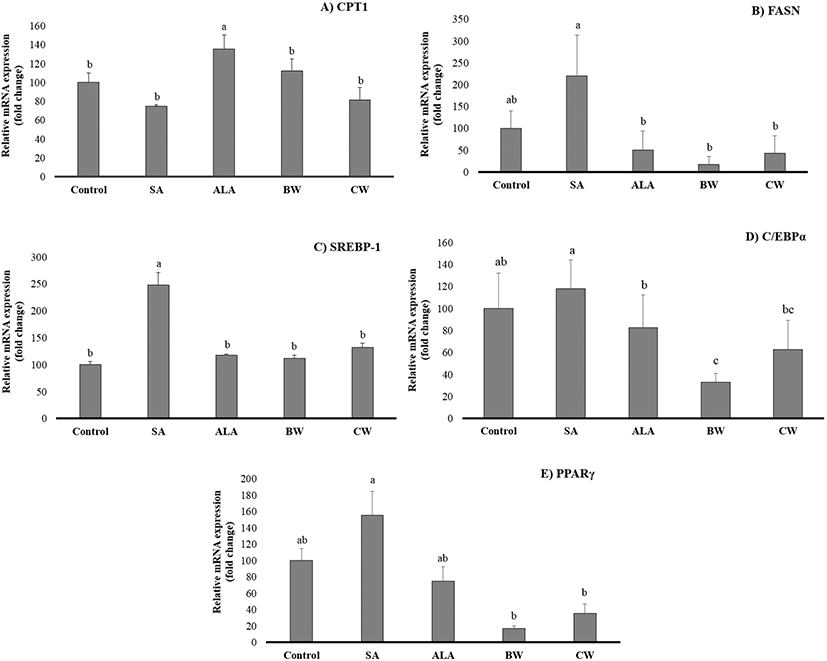
The SA-treated 3T3-L1 cells demonstrated the highest expression of SREBP-1, C/EBPα, and PPARγ when compared to ALA, CW, and BW treatments (Fig. 6C, 6D, and 6E). There was no significant difference observed between the expression levels of BW and CW treatment groups (p>0.05), except for C/EBPα, for which CW expression levels were higher than BW (p<0.05). SREBP-1 is associated with PPARγ activation, which produces the PPARγ ligand (Li et al., 2016). PUFAs are also known to inactivate the lipogenic gene by down-regulating the expression of the SREBPs (Madsen et al., 2005). According to our results, mRNA expression of PPARγ, SREBP-1, and C/EBPα were reduced by ALA treatment, suggesting that ALA, BW, and CW suppressed the lipid accumulation in 3T3-L1 adipocytes through regulation of these genes. A study by Park and Sung (2015) found a similar result with the same fat cell categories after carnosic acid treatment. ALA and natural waxes interfere with the production of PPARy agonist by activating adipose tissues in the liver through the stimulation of the epidermal growth factor receptors, lowering the potential of lipid hoarding in hepatocytes. A previous scientific report, using an in vivo model, has demonstrated that the overexpression of PPARγ in the liver leads to hepatic steatosis (fatty liver disease) as well as an abnormal regulation of lipogenic gene expression, particularly the FASN, ACC and SCD1 genes (Zuo et al., 2006). Additionally, the increased SREBP-1 mRNA level had an effect on synthetic PPARγ-ligands, inducing adipocyte proliferation of new fat cells generation and leading to an increase of fat accumulation in animal (Madsen et al., 2005). Therefore, in the case of FA treatments, it is possible to conclude that the SA upregulated FASN and SREBP-1 comparatively-which shows that long-chain FA synthesis is through FASN-regulated SREBP-1 utilizing the mitochondrial Acetyl-CoA and Malonyl-CoA pathways. The down regulation of CPT-1 suggests that reduced beta-oxidation of FAs supports the anabolic reactions of FAs, whereas the DNA binding transcription factors C/EBPα and PPARγ have shown moderate upregulation which helps in the cell cycle regulation and differentiation of adipocytes. When comparing BW and CW, the BW treatment showed a similar effect to that of ALA. Thus, ALA and BW treatments regulated the lipogenesis pathway, reducing the pro-obesogenic effect. Results from the current study show that 3T3-L1 cells supplemented with ALA, BW, and CW effectively decreased the lipogenesis pathway via regulating adipogenic gene expression showing pro-obesogenic effect. Although this conclusion came from our in vitro study, which is faster and easier to perform, further in vivo study will be necessary to validate these data.
Conclusion
According to the results of this study, ALA and BW are a better choice for adipose tissue function, particularly with regard to cell viability, as they reduce the lipid droplets size and area distribution along with triglyceride storage in 3T3-L1 adipocytes during differentiation. Furthermore, ALA and BW may contribute to the reduction of lipogenesis in 3T3-L1 adipocytes by regulating adipogenesis-related gene expression.













