Introduction
Antioxidants are often added to products containing pork meat to efficiently prevent lipid oxidation and color deterioration. Health and safety concerns suggest that natural antioxidants (from plant or fruit sources) be used. Antioxidants can act as reducing chemicals, free-radical terminators, and metal ion chelators (Mathew and Abraham, 2006). Natural antioxidants are increasingly used to enhance nutritional and health values, and to improve food quality (Mathew and Abraham, 2006; Sebranek et al., 2005; Yoo et al., 2008).
Oxidative processes trigger lipid degradation, contributing to deteriorations in the texture, color, and flavor of meat products (Min and Ahn, 2005), and causing loss of freshness (Sebranek et al., 2005). Inhibition of such oxidation maintains valuable nutritional properties and quality, enhances health benefits, and prolongs shelf life. Antioxidants delay lipid oxidation and improve human defenses against infection. Antimicrobial agents previously incorporated into edible films include organic acids; antimicrobial enzymes; and bacteriocins such as triclosan, grape seed extract (GSE), green tea extract (GTE), essential oils, and chitosan (Cha and Chinnan, 2004; Jongjareonrak et al., 2008).
Meat packaging is an important step in food processing, and is in fact the final step of several production lines. Packaging featuring natural additives is attracting increasing interest from researchers and industries. Recently, efforts have become focused on incorporation of natural antioxidants and/or antimicrobial agents into packaging materials (Chamanara et al., 2012; Siripatrawan and Noipha, 2012). Such films reduce changes in color and texture and inhibit the loss of useful nutrients (Gennadios et al., 1997). Edible films containing antioxidants have been used to inhibit microbial growth and extend food shelf life (Min et al., 2007), and may gradually replace synthetic films, maintaining food quality by protecting against moisture loss, oxygen uptake, and changes in aroma (Huber and Embuscado, 2009).
Procyanidin from GSEs is good sources of antioxidants, as they have high phenolic content (Perumalla and Hettiarachchy, 2011). Grape seeds are especially rich in the phenolics catechin, epicatechin, and procyanidins (Shi et al., 2003). Procyanidins are polyphenols that are found in some vegetables and fruits, and are used as dietary supplements and in pharmacology. The materials scavenge free radicals and bind readily to proteins. Natural polyphenols including procyanidins have been used to treat hide collagen under acidic conditions (He et al., 2011). Procyanidins also exert anti-inflammatory, anti-allergic, and anti-cancer effects; protect against heart disease and skin aging; and stabilize capillary walls, thus improving human health (Facino et al., 1996; Halpern et al., 1998, Nirmala and Narendhirakannan, 2011). Procyanidins are considered valuable non-toxic antioxidants appropriate for use as food preservatives. A previous study found a correlation between procyanidin level and antioxidant activity (Jerez et al., 2007).
Pigs are rich in polyunsaturated fatty acids (PUFA); therefore, it is necessary to delay lipid oxidation of pork meat products using appropriate packaging materials. An edible film impregnated with procyanidins might maintain meat quality, and as such, pork consumption is expected to rise in the future. Pork meat is popular and healthy, despite some negative perceptions. However, pork preservation has not been well studied by procyanidin. Thus, we investigated the effects of an edible film containing procyanidins on pork loin during chilled storage.
Materials and Methods
Procyanidin concentrations were determined using the 2,2’-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid; ABTS) assay. We prepared edible films with procyanidins at 0%, 0.1%, 0.3%, 1%, and 5% (w/w). In preliminary work, we found that procyanidins at 0.1% or 0.3% (w/w) reduced the levels of thiobarbituric acid-reactive substances (TBARS), and maintained the color of pork patties. Thus, we used these procyanidin concentrations in all subsequent experiments; the ABTS radical-scavenging activities of films with higher procyanidin levels were similar to those of the film with 0.3% (w/w) procyanidin.
Procyanidins were obtained from Konkuk University, Korea, as a light yellow powder of purity 99.3% (w/v). Procyanidins were extracted from grape seeds according to previously described (Carando et al., 1999). Films containing procyanidins were prepared as follows. Distilled water (1.5 L) was heated to 80℃, and κ-carrageenan (30 g) and plasticizer (4 g polyethylene glycol solution [PEG]: glycerol=1:1 solution) (Table 1) was added. After cooling at room temperature to 50℃, films were formed after addition of 0.1% and 0.3% (w/w) procyanidins to aliquots of the solution in petri dishes, and were dried for 24 h at 40℃.
| Material | Procyanidin concentration, % |
||
|---|---|---|---|
| 0 | 0.1 | 0.3 | |
| Distilled water (L) | 1.5 | 1.5 | 1.5 |
| κ-Carrageenan | 30 | 30 | 30 |
| Polyethylene glycol | 2 | 2 | 2 |
| Glycerol | 2 | 2 | 2 |
| Procyanidin | - | 0.03 | 0.09 |
We purchased pork loin from a local meat market. All of the meat samples were trimmed, cut into sections 1 cm in thickness, and wrapped anaerobically in films containing different procyanidin concentrations. We measured color values, pH, volatile basic nitrogen (VBN) level, the extent of lipid peroxidation, and changes in microbial load, during chilled storage for 14 d.
Three meat surface color parameters (lightness [L*], redness [a*], and yellowness [b*]) were assessed after unpacking the meat using a Minolta Color Reader (CR-400; Minolta, Japan) and calibrated (L*=86.3, a*=0.3165, and b*=0.3142) with the aid of a calibration plate. All of the color determinations were made in quadruplicate on different regions of the meat surface.
Pork loin (5 g) was homogenized (PT-MR2100, Kinematica, Switzerland) in 45 mL distilled water, and pH values were measured using a digital pH meter (Orion 3-Star Plus, Thermo Scientific, USA) on days 0, 1, 3, 7, 10, and 14. Triplicate readings were obtained.
Meat freshness (VBN level) was measured using the Conway microdiffusion method (Conway, 1950). Pork loin (5 g) was homogenized at 14,000 rpm in 45 mL distilled water for 1 min and filtered through No.1 filter paper (Whatman, UK). The filtrate (1 mL) was transferred to the outer chamber of a Conway microdiffusion unit, and 1 mL boric acid solution (0.01 N; H3BO3) and 50 μL Conway reagent (0.066% methyl red: 0.066% bromocresol green) were added to the inner chamber of the device. The Conway unit was sealed immediately after addition of 1 mL 50% (w/v) potassium carbonate (K2CO3) to the outer chamber. VBN levels were determined following the addition of sulfuric acid (0.02 N; H2SO4) to the inner chamber of the unit. All of the results are given as mg/%.
Lipid peroxidation was measured using a TBARS assay as previously described (Witte et al., 1970). Each sample (10 g) was placed in 35 mL 2 M phosphate buffer (with 20% (v/v) trichloroacetic acid [TCA]) and 50 mL 7.2% (w/v) butylated hydroxytoluene (BHT), and homogenized at 14,000 rpm for 1 min followed by filtration through Whatman No. 1 paper. Filtered samples (5 mL) were mixed with 5 mL amounts of 0.005 M 2-thiobarbituric acid (TBA), and incubated for 15 h in the dark. Absorbance at 530 nm (A530) was measured using a SpectraMax Plus 384 UV-VIS Spectrophotometer (Molecular Devices, USA); all of the results are expressed as mg malonaldehyde (MDA)/kg of pork.
We performed microbial analysis with the aid of 3 M Petrifilms (3M Health Care, USA). Pork samples (25 g) were blended with 225 mL distilled water, and serially diluted. In total, 1 mL amounts of the dilutions were spread on Petrifilm Aerobic Count Plates (3M Health Care) to determine total bacterial counts (TBCs). The plates were incubated at 37±1℃ for 24-48 h. Escherichia coli was incubated on Petrifilm E. coli/Coliform Count Plates (3M Health Care) for 24-36 h under identical conditions. Bacterial colonies were counted and expressed as log10 colony-forming units (CFU)/g of meat. Counts were obtained on days 0, 1, 3, 7, 10, and 14.
All of the data are expressed as means standard deviations (SDs). Differences among samples in terms of procyanidin level or number of storage days were evaluated using a Tukey’s multiple comparison within SAS software (SAS Institute, USA). P values less than 0.05 were considered statistically significant. All of the experiments were performed in triplicate or quadruplicate, with replicates.
Results and Discussion
We measured the surface color lightness (L*), redness (a*), and yellowness (b*) of pork loin packaged with procyanidins for various storage periods (Table 2). The L* values increased (all p values < 0.05) in both procyanidin-treated and procyanidin-untreated samples during storage. Notably, however, the color change in procyanidin-treated pork was less than that of the control after both 1 and 3 d of storage.
All values represent means±SD. n=7-8.
a-cMeans in the same column with different superscripts are significantly different (p<0.05).
A,BMeans in the same row with different superscripts are significantly different (p<0.05).
a, b, c: storage effects. A, B: procyanidin effects.
The a* values generally did not differ among samples by storage period. However, small changes (p<0.05) were affected by procyanidins (at 1, 3 and 10 d of storage). The b* values were higher (p<0.05) at later times than initially; however, such changes were generally not significant. In general, procyanidin treatment and storage time affected the color parameters of pork loins, in partial agreement with the data of previous studies (Bekhit et al., 2011; Mitsumoto et al., 2005). The a* value did not change in the control group during storage, but the a* values of procyanidin-treated samples increased gradually. Thus, neither redness nor yellowness changed rapidly immediately after procyanidin addition, but meat patties could become oxidized during storage. In a previous work, the a* values of pork patties and beef were affected by addition of green tea and oregano (Camo et al., 2011; Jo et al., 2003). Color increases can result from color introduced by procyanidins (Malien-Aubert et al., 2002). Our results are in partial agreement with those of previous studies (Nam and Ahn, 2002). Addition of procyanidins to meat potentially prolongs shelf life by preserving red color, a characteristic that is valued by consumers. Thus, it is reasonable to assume that meat color correlates with quality and freshness (Lorenzo et al., 2014).
Changes in the pH of pork loin during storage are shown (Table 3). The pH of control samples did not significantly change. Storage wrapped in films with 0.1% and 0.3% (w/w) procyanidins showed pH decreases (p<0.05), as previously reported during storage with a polyphenol (Qin et al., 2013). The pH affects water-holding capacity (WHC) in meat (Parlak et al., 2011). pH values exerts major influences on this capacity, drip loss, and color (Brewer et al., 2006; Byrne et al., 2002). Thus, pH is an important indicator of meat quality and freshness. The high quality meat has ultimate pH at the range of 5.4-5.6 (Wêglarz, 2010). Overall, the meat quality enhanced by the procyanidin level and storage periods.
All values represent means±SD. n=7-8.
a-cMeans in the same column with different superscripts are significantly different (p<0.05).
A,BMeans in the same row with different superscripts are significantly different (p<0.05).
a, b, c: storage effects. A, B: procyanidin effects.
The VBN values are shown in Table 4. VBN is an important index of meat freshness (Min et al., 2007). The VBN values increased significantly (p<0.05) in both procyanidin-treated and procyanidin-untreated meat during storage. The shelf life in pork can be improved by the addition of procyanidin (Jeong et al., 2015). The VBN content has a tendency to increase due to amino acid decarboxylase for microbiological effect along with an increase in the storage period (Jung et al., 2010). However, the VBN values were less (p<0.05) in procyanidin-treated than control pork at 7, 10 and 14 d. Polyphenols and flavonoids attenuated proteolysis in human (Pandey et al., 2009). Overall, procyanidins reduced the VBN values of meat.
All values represent means±SD. n=7-8.
a-eMeans in the same column with different superscripts are significantly different (p<0.05).
A,BMeans in the same row with different superscripts are significantly different (p<0.05).
a, b, c, d, e: storage effects. A, B: procyanidin effects.
Changes in TBARS levels during storage are summarized in Table 5. The TBARS values increased significantly (p<0.05) in both procyanidin-treated and procyanidin-untreated meat at all of the storage periods. However, the values of procyanidin-treated samples gradually became significantly lower (p<0.05) than those of untreated controls at 7, 10 and 14 d. Previous studies showed that lipid oxidation in meat were dose-dependently inhibited by antioxidants (Calatayud et al., 2013; Correia and Jordão, 2015). We found that TBARS values generally increased during chilled storage, because the packaging films were permeable to oxygen (Hugo et al., 2009). The TBARS levels in meat treated with a grape seed-derived procyanidin extract (GSP) were significantly less than control values; the GSP suppressed proinflammatory cytokine production, lipid peroxidation, and antioxidant enzyme activity (Lei et al., 2014). Tomato paste significantly (p<0.05) reduced TBARS levels (compared to control) because lycopene is an antioxidant (Kim et al., 2013). The TBARS value of meat packaged in film containing GTEs was significantly less than that of meat wrapped in control film (Nisa et al., 2015). The TBARS value of meat wrapped in chitosan film incorporating GTE was lower than those of meat wrapped in control film (Siripatrawan et al., 2012). Thus, procyanidins exert antioxidant properties when incorporated into edible films, as previously described.
All values represent means±SD. mg MDA.kg-1. MDA = Malondialdehyde. n=7-8.
a-cMeans in the same column with different superscripts are significantly different (p<0.05).
A-CMeans in the same row with different superscripts are significantly different (p<0.05).
a, b, c: storage effects. A, B, C: procyanidin effects.
Changes in aerobic bacterial and E. coli populations during storage are summarized in Fig. 1(a), 1(b). The numbers of aerobic bacteria significantly increased (p<0.05) in both the procyanidin-treated and procyanidin-untreated groups. Procyanidins reduced (p<0.05) aerobic bacterial numbers (compared to the control group). Changes in E. coli/coliform numbers followed a similar trend. E. coli populations increased in both procyanidin-treated and procyanidin-untreated groups during storage. However, procyanidins reduced the E. coli populations compared to those of untreated controls, suggesting that procyanidins are natural antimicrobials. These results are similar to those previously described (Dai et al., 2012). Phenolics were shown to exert strong antibacterial activities (Widsten et al., 2014). A chitosan-tea polyphenol film also extended the shelf life of pork (Qin et al., 2013). We found that a procyanidin film inhibited growth of all aerobic bacteria and E. coli.
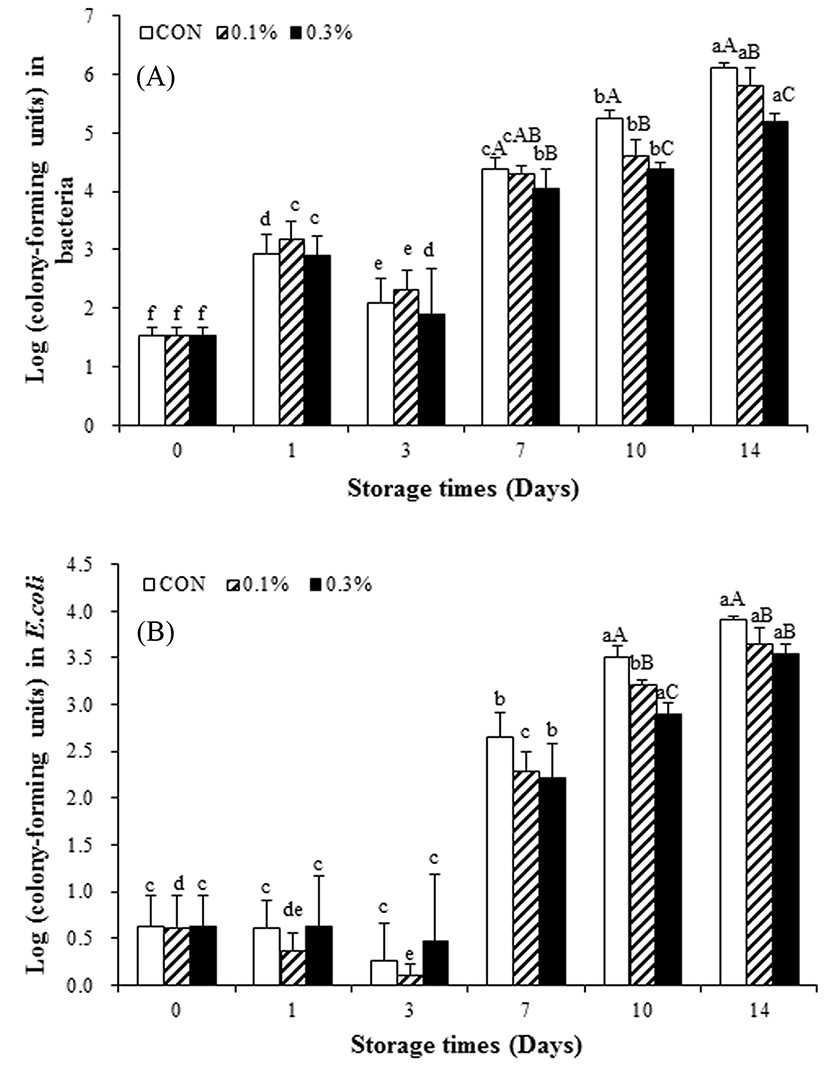
Conclusion
We explored whether edible films containing procyanidins enhanced preservation of pork meat. Such film indeed enhanced meat quality and prolonged shelf life, decreasing VBN and TBARS values, and microbial populations. However, the effect of procyanidins on color may be perceived as negative. Procyanidins may be very useful natural antioxidants and antimicrobials for incorporation into storage films. Edible films containing procyanidins improve meat quality and extend shelf life by attenuating lipid oxidation and reducing microbial populations.













