Introduction
As the consumer demand for reduced fat diet has been increasing, there has been a growing intake of low- and non-fat dairy foods (Boukid et al., 2021). However, low- and non-fat dairy foods may have textural defects, such as formation of week body and increased whey separation (Saleh et al., 2020). Moreover, a reduction in the fat in dairy foods leads to the deficiency of hydrophobic nutrients including omega-3 polyunsaturated fatty acid (PUFA) (Zimet and Livney, 2009). Omega-3 PUFA including docosahexaenoic acid (DHA) and eicosapentaenoic acid has various health benefits. It can decrease the risk of cardiovascular disease and high blood pressure (Ha et al., 2018; Nain et al., 2021). The oxidation of DHA and DHA-rich ingredients (e.g., fish oil) resulted in the formation of fish-like off-flavor and thereby reducing the food quality during storage in low- and non-fat dairy foods (Hwang et al., 2017; O’Dwyer et al., 2012; Saga et al., 2013). Moreover, high hydrophobicity of DHA with poor aqueous solubility makes it difficult to be applied in low- and non-fat foods. Therefore, it is challenged to develop new methods to overcome the limitations when applying DHA for foods. Delivery systems have been used to exceed those limitations by encapsuling and protecting DHA inside of delivery system.
Various studies have focused on the encapsulation of DHA and DHA-enriched ingredients (e.g., fish oil) in nano- and micro-sized delivery systems, which are nanoparticles (Ha et al., 2018; Zimet and Livney, 2009; Zimet et al., 2011), microparticles (dos Santos Vaucher et al., 2019), nanoemulsions (Hwang et al., 2017), and microemulsions (Cortés et al., 2019).
Among various synthetic- and natural biopolymers, milk proteins including caseins have been used as delivery materials for the delivery of hydrophobic nutrients due to their high nutritional value and various functional properties, such as an ability to bind hydrophobic nutrients, gel forming capacity, stabilization of emulsions, and antioxidant activity (Kimpel and Schmitt, 2015; Livney, 2010). Caseins are the main milk protein component accounting for about 80% of total milk proteins (Livney, 2010; Wang and Zhao, 2022). The use of glucono-σ-lactone (GDL) or starter culture reduces the pH of milk and casein, which can lead to the production of acid gels (Lucey et al., 1997; Luo et al., 2015; Nag et al., 2011). The acid gelation is a way to develop delivery systems since the formation of acid-induced gels can efficiently protect and enhance the stability of probiotics (Nag et al., 2011), curcumin (Khanji et al., 2015) and polyphenols (Bayraktar et al., 2019).
It was reported that caseins can bind hydrophobic nutrients via hydrophobic interactions (Forrest et al., 2005; Semo et al., 2007; Zimet et al., 2011). For the delivery of hydrophobic bioactive compounds including DHA, it is beneficial to enhance the hydrophobicity of delivery materials, which may efficiently encapsulate and protect hydrophobic bioactive compounds inside of delivery systems (Ha et al., 2013; Ha et al., 2018). In this study, we hypothesized that the formation of casein derivative by covalent modifications with hydrophobic fatty acids can increase the hydrophobicity of casein effectively enhancing the oxidative stability of DHA in aqueous-based delivery system. Therefore, it can reduce the formation of fishy-like off flavor during storage in dairy foods.
The aims of this study were to prepare hydrophobically modified casein derivatives and to investigate how production variables, such as degree of modification of casein derivatives and pH, affect the physicochemical characteristics of casein derivative-based delivery systems including DHA.
Materials and Methods
Sodium caseinate, 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), N-hydroxy succinimide (NHS), O-pthalaldehyde (OPA), sodium dodecyl sulfate (SDS), 2-mercaptoethanol (2-ME), 1-anilinonaphthalene-8-sulfonic acid (ANS), GDL were obtained from Sigma-Aldrich (St. Louis, MO, USA).
Hydrophobically-modified casein derivatives were produced by using EDC-mediated coupling reactions (Ha et al., 2013; Ha et al., 2018). EDC can induce covalent conjugations between the amino groups of sodium caseinate and carboxyl groups of linoleic acid while NHS was used to enhance the stability of reaction intermediate (O-acylisourea intermediate). Two hundred milligrams of sodium caseinate were dissolved in 20 mL of deionized water and mixed with various concentration of 10 mL of linoleic acid dispersed in ethanol. Linoleic acid concentration levels in ethanol were 0 mg/10 mL, 17.9 mg/10 mL, 35.9 mg/10 mL, and 53.8 mg/10 mL, which correspond to the molar ratio (charged amount of linoleic acid) of 0%, 10%, 20%, or 30% to sodium caseinate solution, respectively. Then, 5 mL of EDC and NHS dissolved in tetrahydrofuran was added to sodium caseinate/linoleic acid mixture followed by stirring for 12 h at room temperature (molar ratio of EDC:NHS:linoleic acid=1:1:1). To remove unbound linoleic acid and residual reaction chemicals, caseinate/linoleic acid/ EDC/NHS mixtures were dialyzed against 50% ethanol and deionized water for 24 and 48 h using dialysis membrane (3.5 kDa molecular weight cut-off, Thermo Scientific, Rockford, IL, USA) followed by freeze-drying.
The degree of modification of casein derivatives, a number of linoleic acid bound to sodium caseinate, was determined by measuring the number of free amino groups using OPA reagent. Twenty-five milliliters of sodium tetraborate buffer (pH 9.5) was mixed with 2.5 mL OPA dissolved in methanol (16.4 mg/2.5 mL) followed by addition of 5 mL of 20% SDS to sodium tetraborate buffer/OPA mixture. Then, 400 μL of 2-ME was added to sodium tetraborate buffer/OPA/SDS mixture and final volume of mixture was adjusted to 100 mL with deionized water. To determine the number of amino groups, 50 μL of unmodified (standard) or hydrophobically-modified sodium caseinate was mixed with 1 mL of OPA reagent. Measurement of the number of amino acids groups at 340 nm was performed using a UV-VIS spectrophotometer (GENESYS10-S, Thermo Spectronic, Rochester, NY, USA).
The surface hydrophobicity of casein derivatives with various charged amount of linoleic acid was assessed by using a procedure represented in Monahan et al. (1995) and Ha et al. (2013). ANS was used as a fluorescence probe for the hydrophobic affinity of protein surface. Freeze-dried casein derivatives were dispersed in Tris-HCl buffer (pH 5.0) and diluted to selected concentration of 0.005%, 0.01%, 0.015%, 0.02%, and 0.25% (w/v) followed by adding 20 μL of 8 mM ANS dispersed in 0.1 M phosphate buffer (pH 7.4) to 3 mL of casein derivative solution. Fluorescent intensity of casein derivative/ANS mixture was evaluated at excitation wavelength of 390 nm and emission wavelength of 470 nm using a spectrofluorometer (Luminescence Spectrometer LS50 B, Perkin-Elmer, Waltham, MA, USA). The R value calculated by following equation was used as an indication of surface hydrophobicity of casein derivatives.
where F is the fluorescent intensity of casein derivatives with ANS and F0 is the fluorescent intensity of ANS solution without casein derivatives.
Casein derivative delivery systems were developed by the use of a modified pH-induced gelation method with GDL (Nag et al., 2011). Five percent (w/v) of casein derivative solution was mixed with DHA dissolved in ethanol and 2% (w/v) GDL solution followed by stirring for 10 min. Final concentration of DHA in casein derivative solution was 25 mg/100 mL. To form delivery system, 10 mL of casein derivative solution including GDL and DHA was added to 40 mL of soybean oil (Sajo Haepyo, Seoul, Korea). It was mixed thoroughly for 5 min in magnetic stirrer and then held for 60 or 70 min to allow to form gel matrix. After holding for 60 and 70 min, pH of sodium caseinates solution reached to 5.0 or 4.8, respectively and aqueous phase (sodium caseinate solution) was turned into micro-sized particles by acid-induced gelation. Casein derivative-based delivery systems formed in water phase were collected by centrifugation at 15,000×g for 1 min and oils at top layer were discarded. After washing 3 times with deionized water to remove residual oil, casein derivative delivery systems were collected and then freeze-dried.
Dynamic light scattering measurement was done with a particle size analyzer (Zetasizer Nano ZS, Marvern Instruments, Malvern, UK) to assess the average particle size, distribution (polydispersity index), and zeta potential values of casein derivative delivery system. It was prepared with a 633 nm HeNe laser, operating at a scattering angle of 173°.
Oxidative stability of DHA during storage in model milk was determined by assessing peroxide, p-anisidine value, and development of volatile compounds (Ha et al., 2018). Model milk was prepared by adjusting pH of distilled water to 6.7 with 0.01 M HCl. Free DHA and freeze-dried casein derivative-based delivery system containing DHA and free DHA were added to model milk and stored at 4°C for 16 d. The development of hydroperoxide (primary oxidation product) and aldehyde (secondary oxidation product) during storage in skim milk was assessed by using peroxide (IDF method 74A, 1991) and p-anisidine value [AOCS method CD 18–90 described by Kargar et al. (2011)], respectively. The formation of volatile compounds during storage was measured by using a GC-mass spectrometry (GC MS-TQ8030, Shimadzu, Kyoto, Japan). An internal standard (3-methyl-3-buten-1-ol, final concentration: 4 mmol/L) for estimating the relative concentration of (E,E)-2,4-heptadienal was added to free DHA and casein derivative delivery system containing DHA in model milk. A solid phase microextraction fiber (Supelco, Bellefonte, PA, USA) was used to obtain volatile compounds from model milk containing DHA during storage. Chromatographic separation was achieved via a DB-5ms column (30 m×0.25 mm i.d.×0.25 μm film thickness, Agilent J&W Scientific, Folsom, CA, USA). The relative concentrations of (E,E)-2,4-heptadienal were evaluated by following equation:
All data were demonstrated as a mean of three replicates. The effects of production variables, such as degree of modi fication of casein derivative and pH, on the physicochemical characteristics of delivery system and oxidative stability of DHA were analyzed by one-way analysis of variance (ANOVA) with a statistical significance of p<0.05. Repeated-measures ANOVA was applied to assess the impacts of production variables, storage time, and their interactions on the oxidative stability of DHA during storage. The statistical analysis system (Version 9.1, SAS Institute, Cary, NC, USA) was used to conduct ANOVA.
Results and Discussion
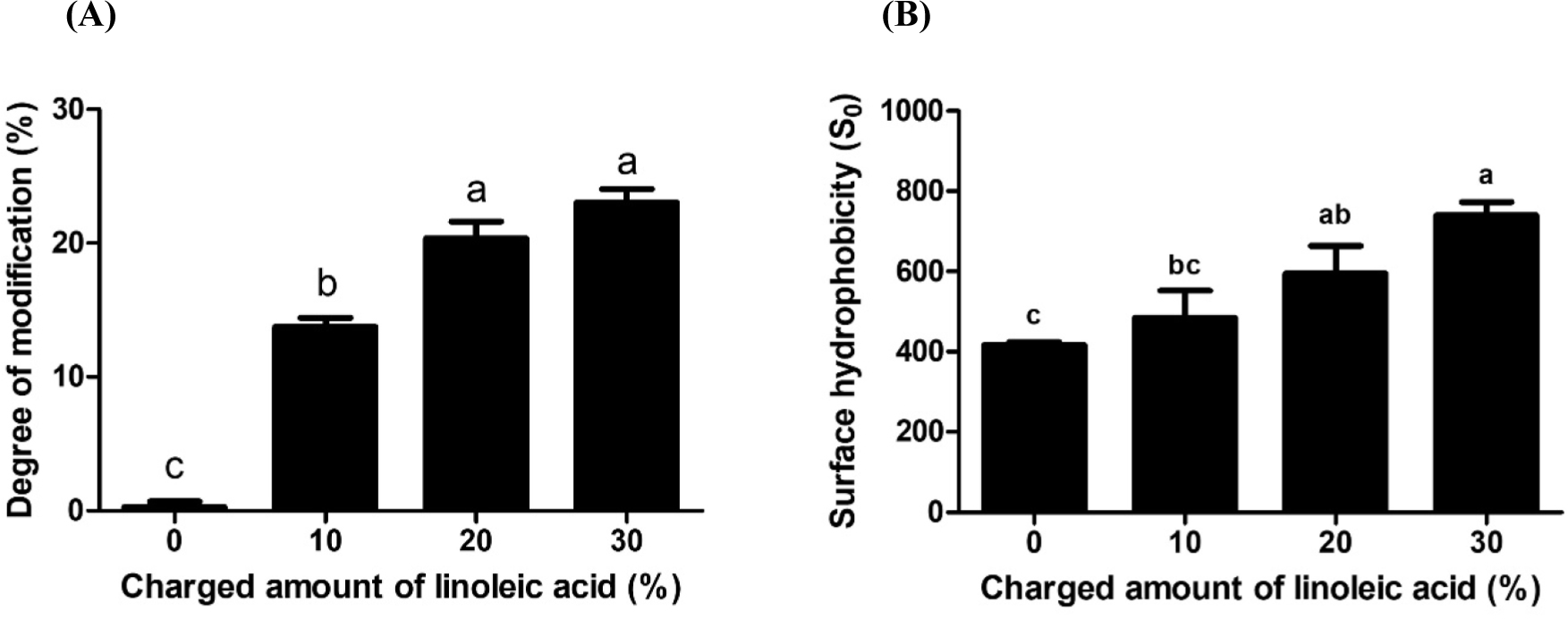
To enhance the hydrophobicity of caseins, the hydrophobic fatty acid, linoleic acid, was covalently attached to caseins using EDC and NHS. When EDC without NHS was used, less than 2% of amino groups of casein was covalently linked with the carboxyl groups of linoleic acid (data not shown). It can be due to the poor stability of reaction intermediate that is acylisourea intermediate. On the other hand, when both EDC and NHS were used, the degree of modification, the number of amino acids of caseins bound to carboxyl groups of linoleic acid, was increased up to 23% (Fig. 1). The effects of charged amount of linoleic acid on the degree of modification of casein derivative were presented in Fig. 1. As the charged amount of linoleic acid was increased from 0% to 30%, an increase in the degree of modification from 0% to 23% was observed indicating that more linoleic acids were covalently attached to amino groups of caseins. Casein derivatives with increasing charged amount of linoleic acid had more linoleic acid residues on the surface of casein derivative. Those more hydrophobic sites on the surface of casein derivative led to an increase in the hydrophobic affinity for ANS resulting in increased surface hydrophobicity (Fig. 1B).
The effects of pH on the physicochemical characteristics of casein derivative delivery systems, such as particle size, polydispersity index, zeta-potential value and DHA encapsulation efficiency, were presented in Fig. 2. There was an increase in the size of delivery systems from 6.5 to 9.4 μm when pH was decreased from 5.0 to 4.8 (Fig. 2A). Since the isoelectric pH of caseins was about 4.6, there are stronger aggregations between casein derivative molecules at pH 4.8 through electrostatic attractions than that of pH 5.0 (McClements, 1999). It resulted in the formation of larger particles at pH 4.8 (McClements, 1999). No significant differences were observed in the polydispersity index values of delivery systems formed at 4.8 and 5.0 (Fig. 2B). The negative surface charges of delivery systems was significantly (p<0.05) increased with increased pH from 4.8 to 5.0 (Fig. 2C). Due to the isoelectric pH of casein is about 4.6, sodium caseinate had more negative charges at pH 5.0 than pH 4.8, which can lead to the formation of sodium caseinate delivery systems with higher negative charges at pH 5.0. Manufacturing pH did not significantly affect the encapsulation efficiency of DHA (Fig. 2D). Since delivery system had much smaller size with higher negative charges at pH 5.0 than pH 4.8, optimum pH was set at 5.0 for further experiments.
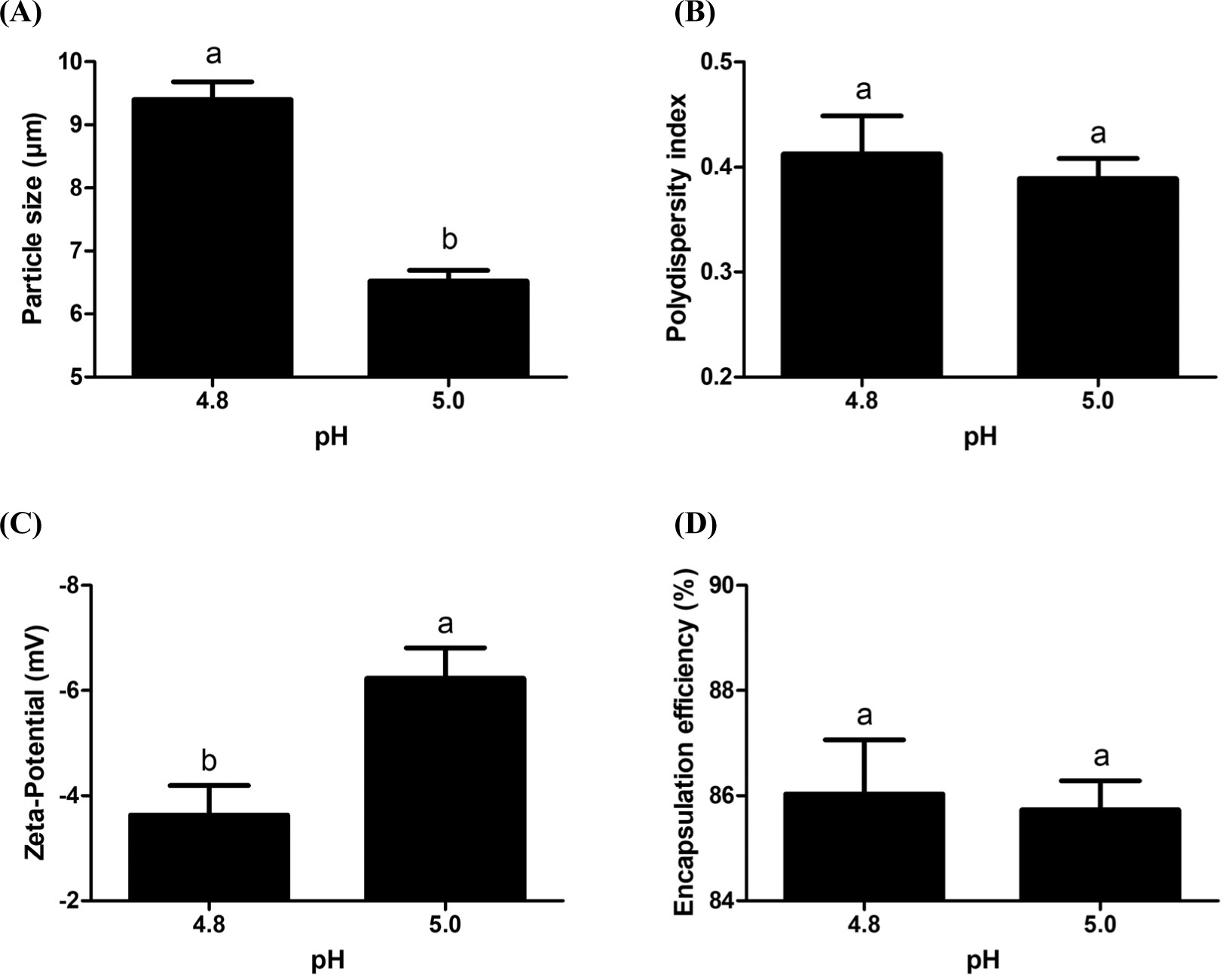
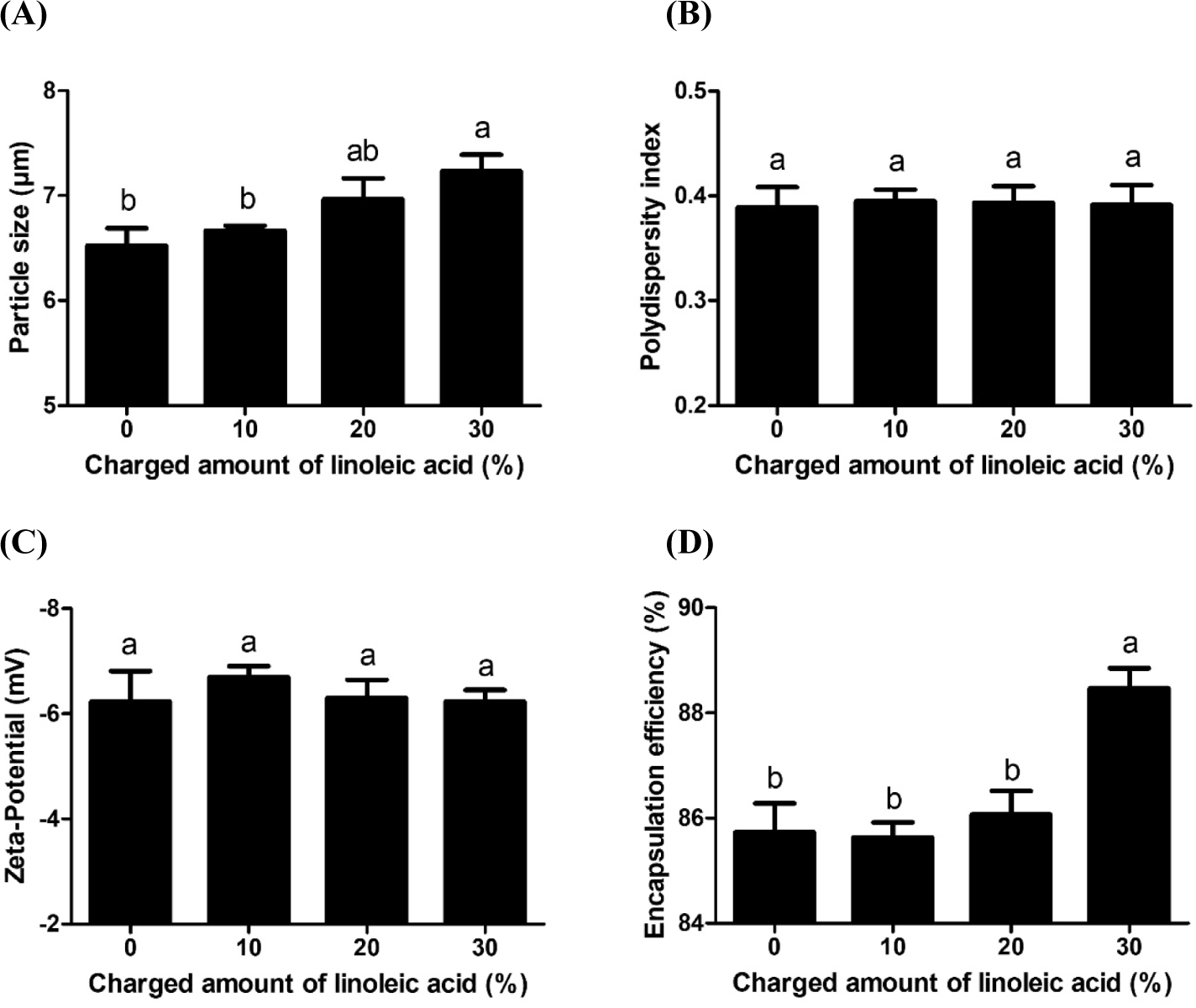
Fig. 3 revealed that increasing charged amount of linoleic acid significantly (p<0.05) enhanced the size and DHA encapsulation efficiency of casein derivative-based delivery systems. Since casein derivatives with higher charged amount of linoleic acid had more hydrophobic linoleic acid moieties and higher surface hydrophobicity (Fig. 1), it could increase hydrophobic attractions between casein derivative molecules and hydrophobic DHA. This can lead to an increase in the size of delivery systems from 6.5 to 7.2 μm (Fig. 3C) and DHA encapsulation efficiency from 85.7% to 88.5% (Fig. 3D).
The effect of encapsulation of DHA in casein derivative-based delivery system on the oxidative stability was presented in Fig. 4. Repeated-measures ANOVA results exhibited that both treatment (encapsulation in delivery system) and storage time (15 d) had a significant effect on peroxide value (Fig. 4A), p-anisidine value (Fig. 4B), and development of (E,E)-2,4-heptadienal (Fig. 4C). A significant (p<0.001) increase in peroxide value, p-anisidine value, and development of (E,E)-2,4-heptadienal was observed with storage time. PUFAs including DHA are easily oxidized resulting in the production of primary oxidation product (hydroperoxide) and secondary oxidation products (aldehydes and (E,E)-2,4-heptadienal). During storage in model milk, noticeable increases in the primary and secondary oxidation products were observed after 6-d storage and gradually increased to 15 d. However, when DHA was encapsulated in casein derivative-based delivery system, peroxide value, p-anisidine value, and development of (E,E)-2,4-heptadienal, a major compound related to fishy-like off-flavor and a potential marker for DHA autoxidation (Venkateshwarlu et al., 2004), were significantly (p<0.0001) reduced compared with that of free (unencapsulated) DHA. It indicates that the use of casein derivative-based delivery systems could efficiently protect DHA from oxidative rancidity during storage. Caseins have been reported to increase the oxidative stability of omega-3 PUFAs (Gallaher et al., 2005; Nielsen and Jacobsen, 2009) due to their antioxidant activity by chelating potential metal pro-oxidants, especially iron. Phosphoseryl groups in caseins are key components responsible for the intense binding properties to transition metals (Diaz et al., 2003; Vegarud et al., 2000). Moreover, the formation of delivery systems by acid-induced gelation may provide physical barrier for DHA, which can contribute to shielding effects reducing the autoxidation of DHA.
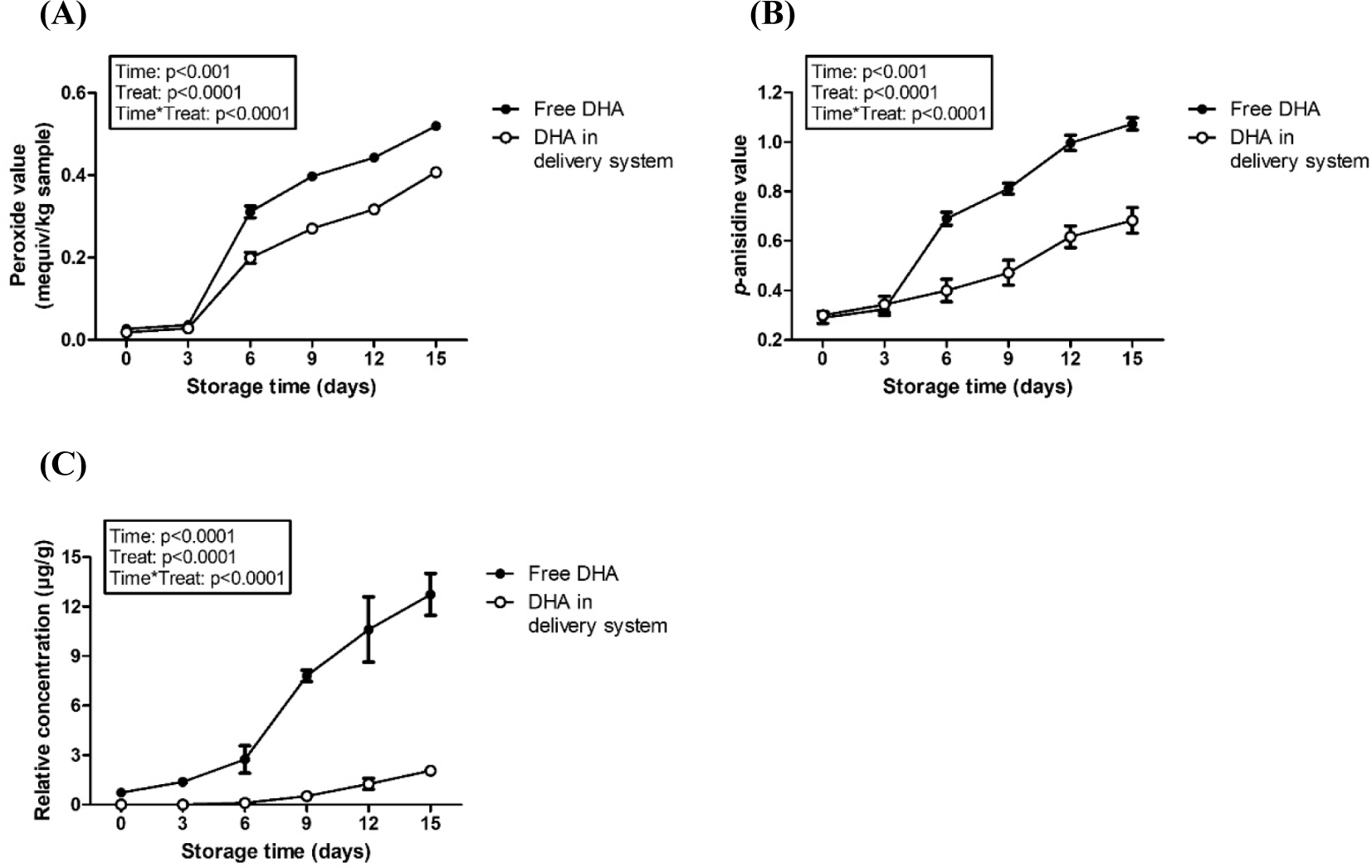
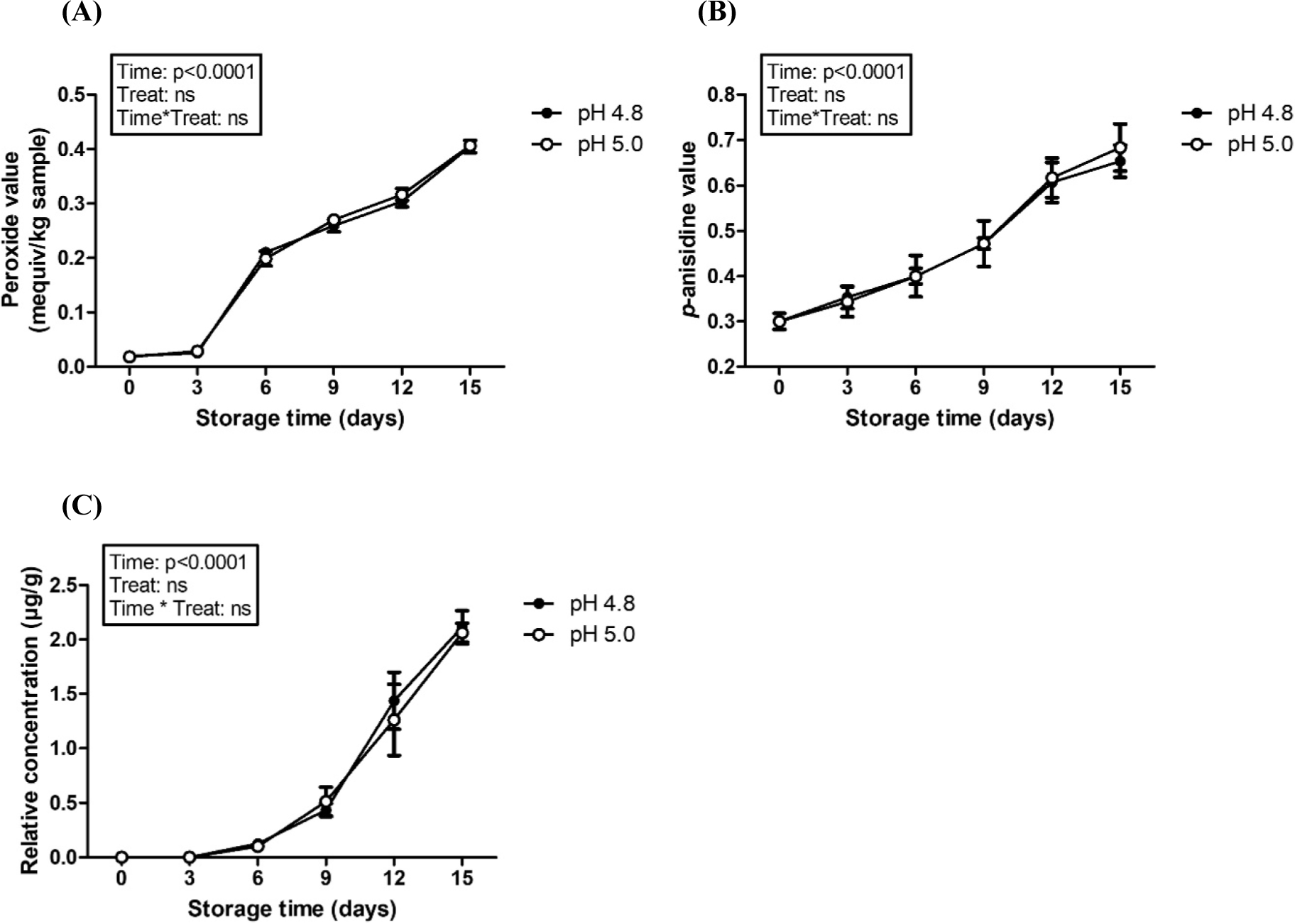
Fig. 5. presents the impacts of manufacturing pH on the oxidiative stability of encapsulated DHA in casein delivery system. Repeated-measures ANOVA results exhibited that storage time significantly (p<0.0001) affected peroxide value, p-anisidine value, and formation of (E,E)-2,4-heptadienal while there are no significant effects on treatment (pH). Although pH had a significant effect on the size and zeta-potential value of casein derivative-based delivery systems (Fig. 2), its protective effects from the oxidative rancidity of DHA were not significantly affected by altering pH.
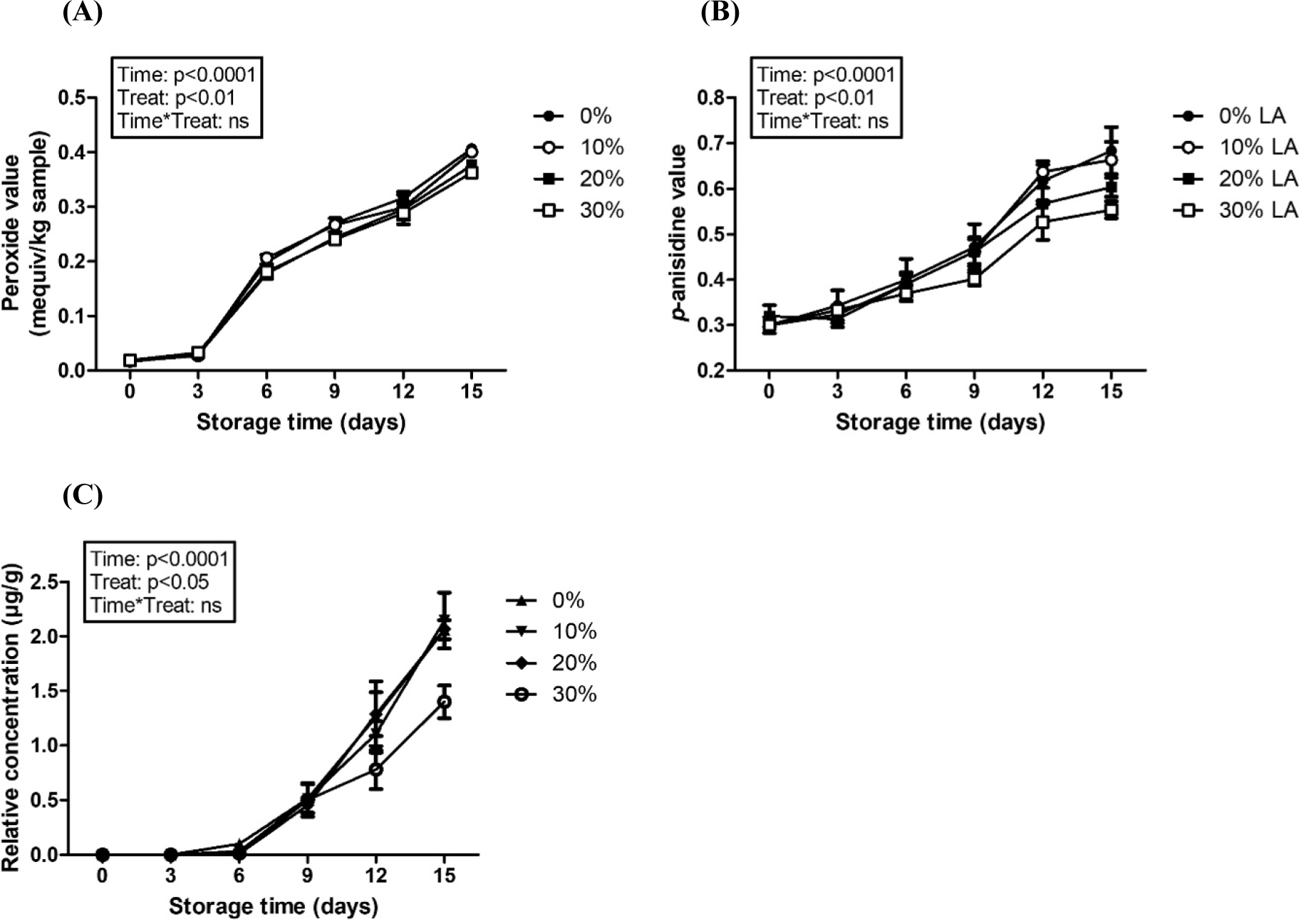
Effects of charged amount of linoleic acid on the oxidative stability of encapsulated DHA were shown in Fig. 6. Similar to Figs. 4 and 5, repeated measured ANOVA results revealed that storage time significantly (p<0.0001) affected the peroxide value (Fig. 4A), p-anisidine value (Fig. 4B), and development of (E,E)-2,4-heptadienal indicating that the oxidation of DHA tended to be increased with storage time. The production of primary oxidation product (hydroperoxide) and secondary oxidation product (aldehydes) was markedly increased after 3-d storage while a significant increase on the fishy-like off-flavor compound, (E,E)-2,4-heptadienal was found after 6-d storage (Fig. 6). Repeated-measures ANOVA presented that hydrophobically modification of sodium caseinate by covalent attachment of linoleic acid with various charged amount of linoleic acid had a significant effect on peroxide vale (p<0.01), p-anisidine value (p<0.01), and development of (E,E)-2,4-heptadienal (p<0.05) during storage in model milk for 15 d (Fig. 6). After 16 d of storage, an increase in the charge amount of linoleic acid from 0% to 30% resulted in a reduction in the formation of (E,E)-2,4-heptadienal from 2.1 to 1.4 μg/g. It indicates that increasing the number of hydrophobic linoleic acid residues attached to sodium caseinate by increasing charged amount of linoleic acid enhanced the hydrophobicity of casein derivative-based derivatives, which can lead to a reduction in the oxidative rancidity of DHA. An increase in hydrophobicity of sodium caseinate could enhance the hydrophobic attractions between sodium caseinate molecules and DHA and may reduce the mobility of DHA and diffusion of prooxidants resulting in a reduction of DHA oxidation (Kellerby et al., 2006). Similar result was reported in a previous study for the hydrophobically modified chitosan/β-lactoglobulin nanoparticles (Ha et al., 2018).
Conclusion
In conclusions, hydrophobically-modified casein derivatives with various degree of modification were produced by modulating charged amount of linoleic acid. Casein derivative-based delivery systems with a size ranging from 6.5 to 9.4 μm were successfully manufactured using acid-induced gelation method with GDL. More than 85% of DHA was encapsulated in casein derivative-based delivery system. During storage in model milk, encapsulated DHA in casein derivative-based delivery system exhibited better oxidative stability than free (unencapsulated) DHA indicating that casein derivative-based delivery system could efficiently protect DHA against autoxidation. In addition, the charged amount of linoleic acid was a key factors affecting the encapsulation efficiency and oxidative stability of DHA during storage in skim milk. Our results suggest that casein derivative-based delivery systems have great potential for the application of DHA to functional dairy foods.













