Introduction
Meat and meat products are susceptible to microbiological and physicochemical changes under inappropriate temperatures (Rupasinghe et al., 2022). Freezing is a common preservation method used to prolong the shelf life of meat, as it can restrain the growth of microorganisms and delay biochemical activities (Medić et al., 2018). In addition, freezing is followed by thawing at temperatures higher than the freezing point (Bae et al., 2014). However, frozen/thawed meat demonstrates inadequate meat quality attributes compared to non-frozen fresh meat (Kim and Kim, 2017). Specifically, structural damage caused by ice crystals leads to the loss of juiciness and releases pro-oxidants that promote microbial growth and oxidation (Jung et al., 2011). Meat is commonly thawed using cold water, room temperatures, refrigerators, and microwaves, but the longer the thawing time and the higher the temperature, the easier it is for microorganisms to grow, especially at room temperature (Park et al., 2012). However, consumers tend to thaw at room temperature for simplicity and convenience, despite the increased risk of meat quality deterioration. In addition, during distribution and subsequent storage, meat is exposed to temperature abuse, which causes the proliferation of bacteria and acceleration of chemical reactions (Limbo et al., 2010). Although cold chain systems have been developed to address these issues, weak points exist in the meat cold chain, such as temperature abuse at ambient temperature during transport, distribution, and unloading in small businesses. The meat exposed to temperature abuse prior to refrigerated storage showed higher microbial spoilage and improvement of water holding capacity (WHC; Vishnuraj et al., 2014; Zhu et al., 2004). However, there are few reports on the quality of beef loins treated with abusive temperatures prior to freezing. Thus, it is vital to assess whether temperature abuse prior to freezing/thawing influences the quality parameters of beef loins. Metabolites detected by nuclear magnetic resonance (NMR) spectroscopy have previously been employed to provide information on meat quality (Kim et al., 2021). Therefore, the purpose of this study was to examine the effect of temperature abuse prior to freezing and during thawing by comparing samples abused only during thawing in terms of quality, microstructure, and metabolites.
Materials and Methods
Raw beef loins (Longissimus dorsi) from one side of three different steer carcasses were obtained and transferred to a laboratory in an icebox. After removing the fat and connective tissues of the outer part of the beef loins, each muscle was cut into a piece weighing approximately 130 g and packaged in a 155×155×60 mm plastic box (LocknLock, Seoul, Korea). The samples were then randomly divided into three groups (CR, FT, and AFT). The control (CR) samples were placed in a refrigerator at 4°C without an abused temperature; those of FT were stored at –18°C for 6 d, followed by thawing at room temperature (20±1°C) for 1 d and kept at 4°C. AFT samples were exposed at room temperature (20±1°C) for 6 h and stored at –18°C for 6 d, followed by thawing at room temperature (20±1°C) for 1 d and stored at 4°C. Samples from each treatment group were collected to assess quality parameters at 0, 1, 8, and 15 days. The FT and AFT groups were defined as d 0 when the sample thawing was completed.
The VBN analysis was conducted according to the micro-diffusion method (Kim et al., 2019). Then, each sample (3 g) was added to 27 mL of distilled water and homogenized for 30 s at 670×g using a homogenizer (T25 basic, Ika, Staufen, Germany). The homogenate was centrifuged at 2,265×g for 10 min (Continent 512R, Hanil, Incheon, Korea) and filtered with filter paper (Whatman No. 1, Whatman PLC, Middlesex, UK). Both 1 mL of filtrate and 50% potassium carbonate (w/w) were placed in the outer section of the Conway cell (Sibata, Sitama, Japan). In the inner section of the Conway cell, 1 mL of 0.01 N boric acid and 100?μL of an indicator solution [0.066% methyl red in ethanol:0.066% bromocresol green in ethanol=1:1 (v/v)] were added together. The Conway cell was then placed in an incubator for 1 h at 37°C, and reacted samples were titrated with 0.01 N of hydrogen chloride. The VBN value was estimated as follows:
0.14: volatile basic nitrogen equivalent to 1 mL of 0.01 N hydrogen chloride
V1: titration volume of sample (mL)
V2: titration volume of blank (mL)
The moisture content of the samples was determined using the AOAC method (Horwitz and Latimer, 2007). The ground sample (3 g) was placed on an aluminum dish and dried in an oven at 110°C for 16 h (DS-520L, Daewon Science, Bucheon, Korea). The water content was calculated using the following equation:
For WHC, the sample (3 g) was chopped and placed on a filter paper and centrifuged at 252×g for 10 min (Continent 512R, Hanil). The WHC was calculated using the following equation:
Vacuum packaged beef loin (80 g) was heated in a water bath for cooking (WB-22, Daihan Scientific, Wanju, Korea) until the central temperature reached 70°C, after which the cooked beef was weighed. Cooking loss was determined using the following equation:
Scanning electron microscopy was performed according to the method described by Shin et al. (2020). Samples of approximately 20×20×20 mm were cut perpendicularly to the muscle fibers. Samples were fixed in a mixed solution (60% ethyl alcohol, 30% chloroform, and 10% glacial acetic acid, v/v) for 24 h under refrigeration (4°C). The fixed samples were dehydrated sequentially using 70% (12 h), 95% (2 h), and 99.5% (2 h) of ethyl alcohol. The samples were submerged in bis(trimethylsilyl)amine in two ten-minute periods. Then, the samples were dried and placed carefully on aluminum stubs with carbon tape and coated with platinum under vacuum for surficial visualization. Images of muscle fibers were obtained using a field emission SEM (AURIGA, Carl Zeiss Microscopy, Thornwood, NY, USA).
NMR analysis was performed using the method described by Kim et al. (2021). Twenty milliliters of 0.6 M perchloric acid was added to 5 g of a sample and homogenized for 1 min at 1,720×g with a homogenizer (T25 digital, Ika). Following centrifugation at 3,083×g for 15 min, the supernatant was placed in another test tube and adjusted to pH 7 with sodium hydroxide. After another centrifugation (3,083×g) for 20 min, the supernatant was filtered to obtain an extract (Whatman No. 1, Whatman PLC). After freeze-drying, the filtered extract (Freezer dryer 18, Labco, Piscataway, NJ, USA) was diluted in 10mM phosphate buffer (pH 7.4) prepared using D2O containing 1 mM TSP. After heating in a water bath (35°C) for 10 min, centrifugation was performed under the same conditions as described above. The supernatant was transferred to a microtube and centrifuged at 17,000×g for 10 min (HM-150IV, Hanil), and 600 μL of the supernatant was transferred to an NMR tube for measurement. The 1H NMR spectrum was acquired using a Bruker 850 MHz Cryo-NMR spectrometer (Bruker Biospin GmbH, Rheinstetten, Baden-Württemberg, Germany), and the analysis was performed using zg30 as a pulse program with a sweep width of 7,812.500 Hz at 128 scans. Metabolites in the spectrum were referenced to the TSP peak at 0 ppm and quantified via pattern integration using Topsin 3.5p7 (Bruker Biospin GmbH).
Each experimental analysis was performed twice for all three replicates. Statistical analyses for the singular effects of the storage method and day were performed using one-way analysis of variance. Significant differences were determined using the Student-Newman-Keuls multiple range test at a significance level of p<0.05, and results were expressed as mean values with standard error of means (SAS 9.4, SAS Institute, Cary, NC, USA).
To identify the differences in metabolites among storage methods, a partial least squares-discriminant analysis (PLS-DA) containing variable importance in projection (VIP) scores within the model were performed using MetaboAnalyst 4.0, according to Lee et al. (2021). The samples were log-transformed and autoscaled before conducting our multivariate analysis.
Results and Discussion
VBN can act as an index of muscle food freshness, as it is associated with the degradation of protein to basic nitrogen caused by microbial metabolism and endogenous proteolysis (Kruk et al., 2011). The maximum acceptability of beef was recommended as 16.5 mg VBN per 100 g (Byun et al., 2003). Table 1 indicates that the VBN values in all samples increased significantly with storage time. The FT and AFT samples had higher VBN values than the CR samples (p<0.05). Ultimately, CR had values below 20 mg/100 g throughout the study period, whereas FT and AFT surpassed the spoilage criterion by 30.80 mg/100 g and 30.33 mg/100 g at 15 d, respectively. In addition, FT and AFT had already exceeded 7 Log CFU/g in the number of total aerobic bacteria, whereas CR reached 6.22 Log CFU/g on day 8 (data not shown). This result is unacceptable for the distribution and consumption of AFT and FT after 8 d (Chai et al., 2017). This result also supports the higher VBN values in FT and AFT than in CR, as bacterial metabolism helps produce basic nitrogen from proteins, especially at moderate temperatures (Kruk et al., 2011). In addition, the high activity of the enzymes catalyzed by the abused temperature may accelerate the formation of VBN (Tak et al., 2005). Furthermore, ice crystals formed in FT and AFT can induce the release of enzymes, leading to an increase in protein degradation (Lee et al., 2021). Therefore, both abuse prior to freezing and during thawing at room accelerated the formation of VBN when compared to no temperature abuse. However, there were no significant differences between FT and AFT, thereby suggesting that abuse prior to freezing did not have a greater effect on VBN formation.
In general, the improvement of WHC is related to the degree of protein degradation which is dependent on the storage period and temperature. The increased protein concentration caused by protein degradation induces water inflow for equilibrium between extracellular and intracellular compartments (Kristensen and Purslow, 2001; Zhu et al., 2004). However, WHC is negatively affected by structural damage caused by ice crystals and protein denaturation (Leygonie et al., 2012). This indicates that the improvement of WHC by protein degradation in FT and AFT can be inhibited by cell disruption caused by ice crystals. Several studies also showed that water content was not always correlated with drip loss during storage (Kristensen and Purslow, 2001). In the present study, there was no significant difference of WHC and moisture contents between the control and treatment groups during the storage period (Table 1).
WHC and cooking loss are not always positively correlated because cooking loss is affected by several factors, such as initial pH and cooking temperature (Liu et al., 2010). Table 1 shows that cooking loss decreased with storage time for both refrigerated and frozen/thawed meat (p<0.05). In addition, the cooking losses of FT and AFT were lower (p<0.05) than that of CR on days 8 and 15. This may have occurred because substantial water in frozen/thawed beef samples was already released via drip loss prior to cooking loss (Zhang and Ertbjerg, 2018).
The microstructures of the CR, FT, and AFT of the beef loin samples following 15 d of storage are shown in Fig. 1. The status of the microstructure is related to eating quality (Leygonie et al., 2012), and differences in the fiber structure are noticeable among CR, FT, and AFT. The structure of CR sustained a structural connection and uniform morphology between the muscle bundles compared to FT and AFT. However, especially in AFT, the two treatments appeared to cause structural destruction, thereby indicating more degradation of the myofibrils via proteolysis than CR (Sotelo et al., 2004). Because free calcium ions released by ice crystals accelerate the activity of calpain which contributes to protein degradation (Zhang and Ertbjerg, 2018). From this result, thawing with temperature abuse affected the loss of integrity of muscle fiber, which could influence the texture negatively, such as severe tenderness (Khan et al., 2016).

Metabolites in meat are related to flavor, either directly or indirectly, and they act as substrates for chemical reactions that form compounds during cooking (Kim et al., 2020). The metabolites found in this study, including hypoxanthine, isoleucine, inosine, leucine, phenylalanine, tyrosine, and valine, are associated with a bitter taste, with glucose and alanine having a sweet taste, alanine, lactate, phenylalanine, and tyrosine conveying a sour taste, and carnosine, anserine, glutamate, and inosine 5’-monophosphate having an umami taste (Oh et al., 2019). Thus, changes in metabolites in beef loin may be attributed to their nutritional value and sensory acceptability to consumers. Therefore, PLS-DA and VIP scores were processed based on metabolite quantification using NMR to differentiate between the control and treatment groups on day 15 (Fig. 2; r2=0.937, Q2=0.673). PLS-DA score plots revealed distinct clustering between the control and treatments, thus indicating that they had different quality traits on the last storage day (Fig. 2a). According to the VIP score, the intensity of the measured scores was high, in the order of anserine, glucose, inosine, phenylalanine, glutamate, and creatine, which were represented by variables with high contribution (>1 score) in the PLS-DA model (Almeida et al., 2013; Fig. 2b). Specifically, three metabolites with high VIP scores (anserine, glucose, and inosine) were much higher than other metabolites and were higher in CR than in FT and AFT. Anserine (β-alanine-3-methyl-l-histidine) is a bioactive compound with antiaging, antioxidation, and neurotransmitter functions (Jung et al., 2013). Anserine can be reduced during storage as it hydrolyzes to 1-methyl-histidine and β-alanine from anserine (Shumilina et al., 2016). Glucose is involved in the Maillard reaction and provides sweetness and a meaty flavor. The glucose in meat is the primary substrate for bacteria to grow, and organic acids produced from glucose contributed to off-odors during storage (Casaburi et al., 2003). Inosine is a metabolite produced by the enzymatic reaction of inosine 5’-monophosphate (IMP), an indicator of freshness (Aliani et al., 2013). In the VIP scores, inosine was highest in the CR group compared to the other treatments. This is because the inosine in the treatments was already degraded to hypoxanthine according to the NMR profile (data not shown). The rate of spoilage depends on the concentration of glucose and amino acids in meat, resulting the principal precursors of microbial metabolites responsible for spoilage (Casaburi et al., 2003). Accordingly, bacteria and high activity of enzymes by temperature abuse stimulated more changes in metabolites. Less positive taste-related metabolites were formed in FT and AFT than in CR. Therefore, beef samples subjected to temperature abuse may have had a worse quality than CR because of the high formation of metabolites that may have acted as a negative flavor.
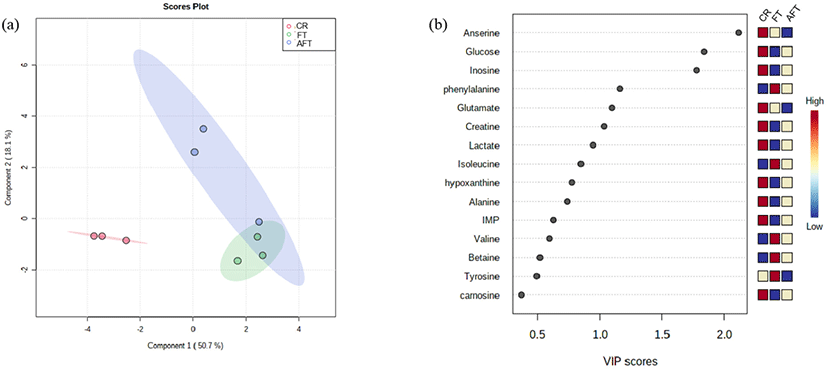
Contrary to the tendency for amino acids to increase more in AFT than in FT, several amino acids (phenylalanine, isoleucine, valine, betaine, and tyrosine) of the samples were significantly higher in FT than AFT on day 15. We found that the temperature abuse prior to freezing influenced the metabolites of AFT to be more varied than those of FT.
Conclusion
Based on the results obtained in this study, we conclude that temperature abuse on frozen/thawed beef loin influenced quality changes when compared with the control and that no more than 8 d of storage is acceptable when VBN values are considered, as per the recommendation of Byun et al. (2003). Furthermore, the difference between FT and AFT was not clearly shown in VBN, WHC, and cooking loss, differentiation was observed slightly through SEM images and detected metabolites. This study indicates that minimizing exposure at room temperature prior to freezing and during thawing is also important in preventing quality deterioration of beef.













