Introduction
Flavor is a combination of aroma and taste, and is one of the main factors that drive consumer accept foods (Maughan et al., 2012). Meat flavor is created by components that are derived from either lean or fat tissues and can be divided into two categories - the general meat flavor of all animal species and the specific flavor of beef, pork, lamb or other species (Myers et al., 2009). Species-specific flavors have been traditionally associated with many factors, such as fat, fat-soluble volatile compounds, and phospholipids (Melton, 1999). And those specific flavors vary in meats from different animal species (Rhee et al., 2005).
Donkey is one of the most important domestic animals and plays a vital role in transporting people and goods across the old world. Today, in many countries, donkeys are multi-purpose, as they not only provide transport, but also milk, blood and meat (Camillo et al., 2018). In China, donkey skin is applied as traditional medicine (Colla Corii Asini) to treat anemia (Shi et al., 2011). Donkey meat has recently been recognized as nutritive food for human consumption with good quality proteins, vitamins, and minerals (Lorenzo et al., 2014). Moreover, the leaner meat, with less fat and consistent quality is favored by current consumers, and thus donkey meat is mainly produced by young animals to avoid undesirable characteristics such as the lack of tenderness (Polidori and Vincenzetti, 2013). However, the consumption of donkey meat in the world is very scarce and limited to some countries, such as China, Spain, Bulgaria, Italy, and several countries in Africa (Aganga et al., 2003; Camillo et al., 2018). Several reports mentioned that meat of some other animal species such as buffalo and goat, with favorable nutritional profile, similar as meat products from donkey can be an alternative in the beef and pork markets (Madruga and Bressan, 2011; Marino et al., 2015; Paleari et al., 2003). Previous study have shown that taste nucleotides, taste activity value (TAV), and the flavor content and amino acids in donkey neck meat were higher than any other part of that animal such as lame, ribs and ridge meats (Li et al., 2019). In order to provide new insights into donkey neck meat flavor components and predict its more possible consumption, the present work was carried out to evaluate the flavor substances in neck meat of four different animals (donkey, swine, bovine, and sheep) and try to find out the main causes of these flavor differences.
Materials and Methods
Donkey meats (Xiaohei Donkey, 2-year-old, male) were obtained from Shandong Dong-E-E-Jiao Co. Ltd. (Shandong, China). The bovine (Charolais, 2-year-old, male), swine (Large White pig, 6-month-old, male) and sheep (Charolais sheep, 1-year-old, male) neck meats were provided and appraised by School of Food Science and Technology, Jiangnan University. All the muscles of the pectoral girdle were picked, aseptically trimmed, and divided to experiment immediately after post mortem.
For the detection of volatile compounds, each sample (5.0 g) with five replicates were weighed, sealed, and placed in a constant temperature water bath at 80°C for 30 min. After cooling to room temperature (20°C), the volatile compound analysis was performed on a fast gas phase electronic nose (model Heracles II made by Scientific System, Inc. SSI) with 2 hydrogen flame (FID) detector and an autosampler. The samples were double-column synchronous analysis with a non-polar column DB-5-FID1 and a polar column DB-1701-FID2 (2 m×1 mm). The analytical condition was as follows: program temperature 10°C/s, injection speed 125 μL/s, injection time held continuously at 13s with temperature 200°C; injection volume 2,000 μL; an initial temperature of search trap was 50°C, temperature of detector was 260°C.
The meat samples were preheated as the above mentioned in electronic nose analysis. The headspace solid phase extraction conditions were as follows: extraction head model 50/30 μm DVB/CAR/PDMS; 250°C, 30 min aging, then extraction head was placed in 80°C heating tray, constant temperature headspace extraction for 50 min. After extraction, the needle was pulled out and inserted into the GC inlet immediately, and the test was completed in 2 minutes. GC-MS was analyzed on a DB-5 capillary column (Agilent J & W GC, 30 m×0.25 mm×0.25 μm) with high purity helium at 1.0 mL/min flow rate. The inlet temperature was 250°C. The GC-MS analysis methods as follows: set initial temperature at 40°C, held for 3 min, then increased to 240°C at a rate of 10°C/min, held for 5 min. The transfer line temperature was kept at 240°C, and the ion source temperature was 240°C. The detector operated in scan mode from 35 to 500 m/z with an ionization voltage of 70 eV.
Lipids were extracted from the meat as described by Gao and Zhang (2010). For the fatty acid composition analysis after saponification and methyl esterification, the lipid sample (750 μL) was dissolved in 2% sodium hydroxide methanol solution (8 mL), the sample was heated at 80°C, and 15% boron trifluoride methanol (7 mL) was added, then cooled to room temperature. The fatty acids methyl esters were extracted with normal heptane and 1 μL was injected into a gas chromatograph. Chromatographic conditions: Supelco SP-2560 vapor phase capillary column (30 m×0.25 mm×0.2 μm, 100% dicyanopropyl polysiloxane as a stationary phase); helium was used as the carrier gas; 1.0 mL/min constant flow; the column temperature was held at 140°C for 5 min, and then increased with speed of 4°C/min up to 200°C and held for 1 min, then increased with speed of 3°C/min from 200°C to 220°C and hold for 26 min; final inlet temperature was 230°C.
For the free amino acid determination, 5.0 g sample was homogenized and filtered by 0.45 μm microporous membrane, as described by previous report (Kobayashi et al., 2016). An amino acid analyzer was used to amino acid determination. The analytical conditions were: Na-type ion exchange column (4.6 mm×60 nm, 3 um) with lithium salt buffer as the eluent; two-channel detection, at the flow rate of 0.35 mL/min, with a detection wavelength was 420 and 570 nm, respectively; column temperature was 57°C, post-column derivatization, the derivatizing agent was ninhydrin buffer and the reaction temperature was 135°C.
Nucleotides were extracted and analyzed as described by Dai et al. (2011) with little modification. Meat samples (5.0 g) were homogenized with pre-cooled 5% perchlorate (15 mL), then centrifuged at 10,000×g for 5 min. The supernatant was transferred to a 100 mL beaker, 15 mL of pre-cooled 5% perchloric acid was added, shaken, and the supernatant was centrifuged and mixed again, and the above operation was repeated twice. The pH of the supernatant was adjusted to 6.5, and ultrapure water was adjusted to 50 mL volumetric flask. The liquid phase filtered prior to high-performance liquid chromatography (HPLC) injection. The HPLC system consisted of a SPD-20A UV detector and a Galaksil EF-C18Bio column (5×250 mm). The mobile phases were A: 0.05 M NaH2PO4/H2O, B: methanol. The gradient was A: B (100:0 for 0–6 min, 100:95 for 6–7 min, 100:95 for 7–16 min, 0:100 for 16–30 min). Nucleotides of standard substance and samples were detected at 260nm and flow rate was 1.5 mL/min. Each nucleotide was identified with 37 Component FAME Mix standard substance which was purchased from sigma.
Weighing standard product dissolved in ultra pure water to prepare 1,000 μmol/L mother liquor, then diluted to 100, 40, 20, 10, 5 μmol/L, the injection volume was 20 μL. Nucleotide identification was made by comparing the retention time of the solution in the standard. The peak area was taken as the abscissa, and the concentration of each standard was the ordinate, and a standard curve was drawn. The HPLC chromatogram of the mixed standard solution is seen in LI (Li et al., 2019). The peak order and retention time of each individual standard in the meat samples were: guanylate (GMP): 4.939 min; muscle glycosylate (IMP): 5.344 min; hypoxanthine (Hx): 6.513 min; adenylate (AMP): 8.804 min; inosine (HxR): 15.579 min. The gradient concentration of the nucleotide standard was analyzed, and the standard curve for establishing five nucleotides was as shown in follow: GMP: Y=1,2636x+4,211.5, R2=0.9999; IMP: Y=8,147.3x–4,717.9, R2=0.9999; Hx: Y=9,825.1x+ 3,1443, R2=0.9958; AMP: Y=1,5176x–3,953.7, R2=0.9999; HxR: Y=8,825.4x+2,353.6, R2=0.9999.
Estimation formula of umami nucleotide content as followed: X=C×V×m (Wen and Wu, 2010), and a general linear models (GLM) procedure was also used. X: target substance content (mg/100 g); C: concentration of target substance (mg/mL); V: sample’s volume (mL); m: weight (g).
Taste activity value (TAV) = C/T (Chen and Zhang, 2007a). C: weight; T: odour threshold.
Results and Discussion
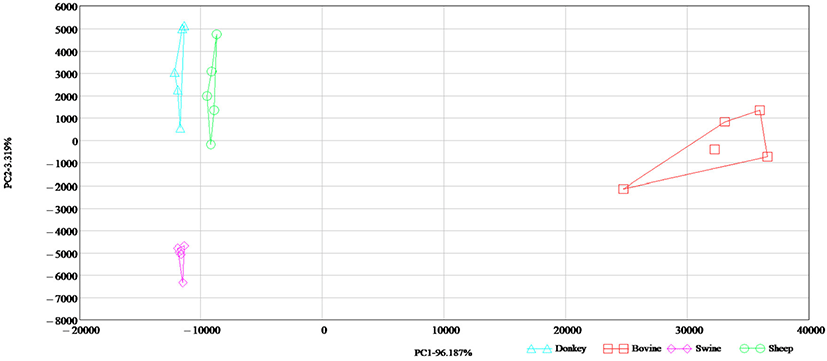
Flavor volatile components of different animals’ neck meat was assessed by Heracles II fast gas phase electronic nose. Furthermore, the principal component analysis (PCA) was performed with flavor compounds in different samples; so as to linearly convert the raw data vector of the sample, and change the coordinate axis to achieve the purpose of distinguishing the sample. Possible flavor differences among the samples of neck meat were analyzed according to the retention index designation library (Fig. 1). It was reported that the unidirectional distance of the sample in the horizontal and vertical coordinates in PCA plot represented the difference between the samples. Indeed, if the lateral distance of the two samples is larger, it indicates that there is a large difference between the samples, and vice versa; since the longitudinal distance has a small contribution rate between samples, the influence on the difference between samples was also small (Huang et al., 2018; Tian and Sun, 2008), and more than 70% to 85% of the overall contribution rate can be tested experimentally. It can be seen that principal component 1 (PC1) contributed to 96.19% of the data variability while principal component 2 (PC2) contributed to the 3.32% of the variability. And these two principal components explained 99.51% of the variability. Therefore, most of the valid information of representative samples can be compared (Xin et al., 2014), and the quadrilateral regions where the four samples are located do not overlap each other, indicating that the electronic nose can effectively distinguish the flavor differences between the four different meat necks. It can be obviously seen that PC1 is the main flavor difference component, which has a great influence on the overall flavor. The distance between the neck meat of donkey and bovine is larger, that is, the difference between PC1 is larger, and the difference between the neck meat of donkey and swine is smaller in the abscissa, indicating that the PC1 has less difference. The difference in ordinate distance is large, but because of its small contribution rate, the overall flavor difference between the neck meats of donkey and swine is small. Thus, in general, the difference in flavor between the neck of donkey, bovine and sheep is more significant, and slightly lower difference in flavor between the neck meat obtained from donkey and swine.
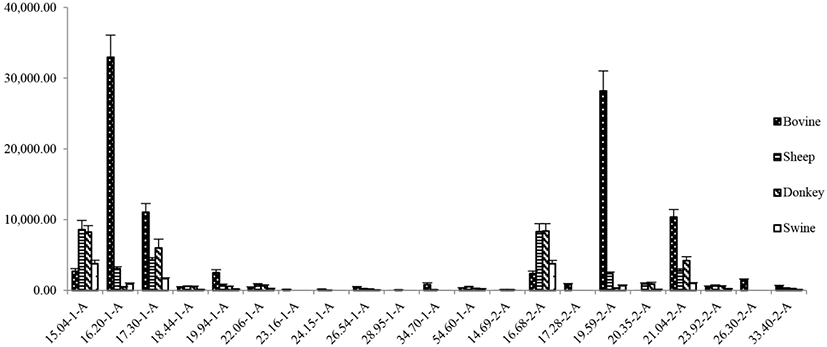
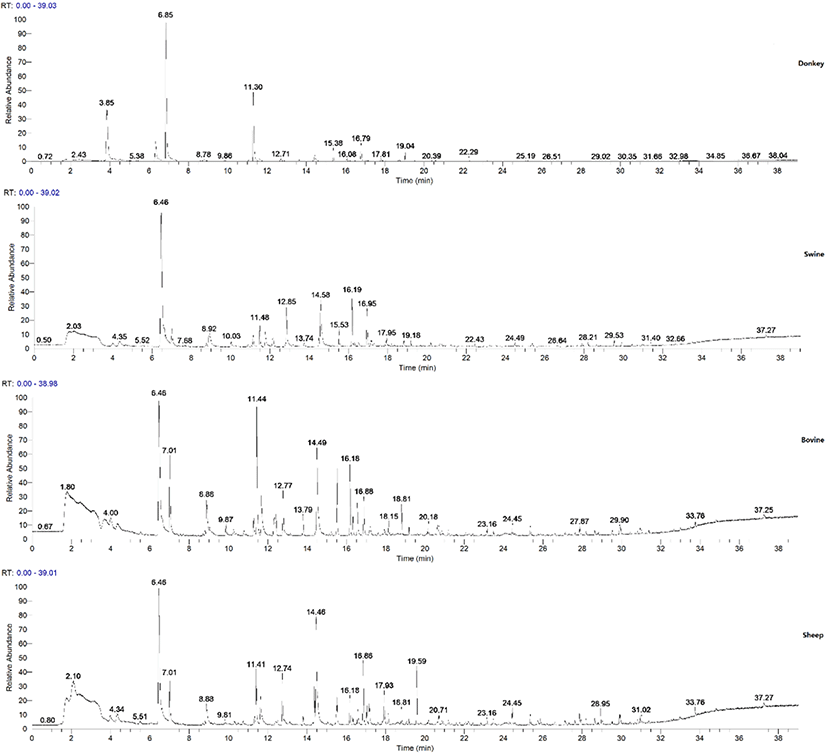
It was shown in Fig. 2 that the number 1 indicated the retention time under the DB-5-FID1 column and the number 2 indicated the retention time of the DB-1701-FID2 column. Comparing the chromatograms of two different polar columns, the flavor components shown the difference initially identified, with the retention time on 15.04-1-A and 16.88-2-A, 16.20-1-A and 19.59-2-A, 17.30-1-A and 21.04-2-A, 18.44-1-A and 23.92-2-A, 26.54-1-A and 33.40-2-A. Different flavor components were listed in Table 1, and the aldehydes were in the highest amount. The GC–MS analysis (Fig. 3) was applied on verifying the results in the electronic nose analysis and analyzing the composition of the volatile components and the results were shown in Table 2, Table S1, Table S2 and Table S3. A total number of 73 compounds were identified in four samples. Among them, 25 volatiles [n-hexanal (0.52 mg, 46.85%), heptanal (0.07 mg, 6.31%) and octanal (0.07 mg, 6.31%)] were detected in donkey neck meat; 40 volatiles in swine neck meat, mainly including n-hexanal (1.79 mg, 38.74%), nonanal (0.50 mg, 10.82%), and n-caproic acid vinyl ester (0.35 mg, 7.58%); 42 volatiles were detected in the neck of sheep, mainly including hexanal (0.96 mg, 24.12%), nonanal (0.70 mg, 17.59%) and 2,3-octanedione (0.26 mg, 6.53%); and 33 volatiles in bovine neck meat, mainly including n-hexanal (1.69 mg, 28.89%), nonanal (0.90 mg, 15.38%) and octanal (0.43 mg, 7.35%). Previous work on meat flavor (Xie et al., 2008; Zhao et al., 2017) corroborated with these results that the aldehydes accounted for the largest (Table 3), followed by the alcohols and hydrocarbons; considered as the key volatile flavor compounds in neck meats. The result was consistent with the possible flavor differences in Table 1. Among the aldehydes, n- hexanal is the most volatile flavor in the neck of the four neck meats, known for its clear fragrance of grass, mainly derived from the oxidative decomposition of oleic acid, linoleic acid and arachidonic acid. Alcohols is the second largest flavor compounds in donkey neck meat, and the alcohols is mainly produced from the automatic oxidation of fat in the meat. The majority of those alcohols is 1-octene-3-ol which belongs to the unsaturated alcohols and the nature of low odor thresholds. Thus, it could be easily detected and contributed a lot to the oil odor of the donkey neck meat. The different content of hydrocarbon compounds in the donkey and other animal’s neck meat inevitably led to their different aroma profiles. Hydrocarbons are common volatile components, but with a higher aromatic threshold or even no odor, they have little contribution to flavor. In agreement with the electronic nose PCA analysis results and compared with the swine neck meat, donkey neck meat has analogous amount of alcohols, though slightly higher number of aldehydes, and less flavor contribution from hydrocarbon; it can be seen that the flavors of swine and donkey neck meat are similar.
Fatty acid composition profile of donkey and three other animals’ neck meat was shown in Table 4. Donkey neck meat appeared to have the highest level (39.00%) of oleic acid; followed by palmitic acid (25.32%) slightly higher than the other three samples. Linoleic acid accounted for 15.83% in donkey neck meats, significantly higher than bovine and sheep and slightly higher than swine ones. It appeared that the fatty acids in donkey neck meats were mainly unsaturated fatty acids, accounting for 63.18%. The oxidation of enzymes in fat oxidation is the primary oxidation reaction of unsaturated fatty acids under the catalysis of lipoxygenase. The primary hydroperoxides form secondary oxidation products by homogenization or β fission decomposition, such as aldehydes and ketones make great contribution to meat flavor (Chen and Zhang, 2007). Oleic acid and linoleic acid are the two most abundant unsaturated fatty acids in the donkey neck meats, two sums for 54.83% and higher than those of thee others. The n-hexanal and other aldehydes detected in this experiment, such as heptanal and non-aldehyde were the main volatile oxidation products of unsaturated fatty acids. N-hexanal was considered to have unpleasant rancidity and grass odor (Kobayashi et al., 2016), mainly from oleic acid, linoleic acid and arachidonic acid oxidation. The decomposition of hydroperoxide produced in the process was consistent with the high content of unsaturated fatty acids in the donkey neck meat. Heptanal has odors with salty taste, barbecued fat, oily aroma and citrus aroma (Huang et al., 2018), derived from the oxidative decomposition of linoleic acid. The oxidative decomposition of arachidonic acid derived from nonaldehyde (Tian and Sun, 2008) give a strong fat taste and citrus fragrance. The 1-octene-3-ol, a high-content unsaturated alcohol in the donkey neck, is a product of auto-oxidation of linoleic acid and produce a “mushroom” flavor (Luo et al., 2010).
Free amino acids are great contributors of flavor and taste in meat products by generating volatile compounds through Maillard reaction and Strecker degradation (Dashdorj et al., 2015). Furthermore, amino acids are also the basic substances that constitute the protein needed for animal nutrition, and participate in many physiological activities of the human body. The content and taste parameters of free amino acids in the neck meats were shown in Table 5. The type and content of amino acids determine the nutritional value of food proteins. As can be seen from Table 5, the main amino acids in the donkey neck meats were alanine (Ala), lysine (Lys), glutamic acid (Glu), glycine (Gly), and serine (Ser). And low-content amino acids were asparagine (Asp), leucine (Leu), and arginine (Arg). Other less abundant amino acids were tyrosine (Tyr), proline (Pro), phenylalanine (Phe), valine (Val), isoleucine (Ile), histidine (His) and threonine (Thr). Among them, 6 kinds of essential amino acids and 9 of non-essential amino acids were detected. The concentration of essential amino acids in neck meat samples were donkey>bovine>swine>sheep (donkey: 42.55 mg/100 g, bovine: 7.37 mg/100 g, swine: 7.13 mg/100 g, sheep: 5.28 mg/100 g), indicating that donkey neck meat is nutritious and could better provide necessary amino acids for human consumption which body cannot synthesize. The total amount of amino acids in donkey neck meats is the highest when compared with three other types of animals, indicating that the taste of the donkey neck meat may be more delicious. Among them, the sweet amino acid Ala and the umami amino acid Glu content in the donkey neck meat are significantly higher than other meats. The umami taste in meat products mainly depends on the content of umami amino acids. The composition and concentration of free amino acids such as Asp, Ala, Gly and other umami amino acids play an important role in enhancing savory or umami taste. Besides, different amino acid combinations will form different flavors with characteristic properties (Arnau et al., 1998; Estévez et al., 2003). For instance, Gly, Ser, Pro, Thr and Ala for the sweet and savory taste (Rico et al., 1991); mainly umami amino acids including Asp and Glu; those containing a sour taste such as His, Asp, Pro; Arg, Phe, Try, His, Val, Met, Ile, Tyr and Lys has the bitter taste (Arnau et al., 1998; Rico et al., 1991). As it can be seen from Table 6 the highest proportion of amino acids in the donkey neck meats was the sweet amino acid, while the bitter taste amino acid content in the bovine, swine and sheep neck meat was relatively high. In all neck meats, the proportion of umami amino acids was relatively low. In comparison, it was found that the proportion of sweet amino acids in the donkey neck meats was significantly higher than that in the neck of other meats. The sum of the sweet and umami amino acids in donkey neck meat was also higher than that of other meats, especially the sweet amino acid Ala and the umami amino acid Glu were significantly higher than other animal neck meats, indicating that the sweet umami amino acid contributed a lot to the overall taste of donkey neck meats.
Umami taste is described as savory, brothy, or beefy, which is triggered by the flavor-potentiating compounds, such as 5’-inosine monophosphate (IMP) and 5’-guanosine monophosphate (GMP) and by the synergism of these nucleotides with L-glutamate (Dashdorj et al., 2015). The umami taste of L-glutamate can be significantly enhanced by 5’-ribonucleotides and the synergy is a property of this taste quality (Zhang et al., 2010). The ATP rapidly drops within a few hours after slaughter by getting converted into ADP, AMP, and other derived compounds by the action of several enzymes. Both ADP and AMP act as intermediate compounds, and then they decrease to negligible values (Batlle et al., 2001. For example, AMP is deaminated into IMP, and this compound is progressively changed into inosine and hypoxanthine in the fresh meat within few hours after slaughter depending on muscle type (Ishiwatari et al., 2013; Zhang et al., 2010). In this study, HPLC was used to analyze the contents of five kinds of flavor nucleotides of guanylate, inosine, hypoxanthine (Hx), adenylate and inosine in the neck of four kinds of meat (Table 7). As shown in Table 7, the nucleotides in the neck of the donkey were mainly IMP and inosine HxR, while the contents of AMP, GMP and Hx are relatively low. After slaughtering, cooling and maturating, ATP is decomposed into ADP, AMP, IMP, etc. IMP was the most abundant in samples of donkey neck meat, indicating that of ATP was rapidly degraded into IMP under the action of endogenous enzymes. However, due to the very slow degradation of IMP, that was accumulated (Han et al., 2017), indeed, the content of IMP was higher than that of other odorants. Therefore, IMP and AMP are the most important nucleotides for the taste of donkey neck meat. Studies have shown that there is a synergistic effect between IMP and AMP in enhancing the umami taste of food (Wu et al., 2014). Comparing the total amount of taste nucleotides in the neck of four kinds of meat, it can be found that swine>donkey>sheep>bovine, indicating that the taste of the donkey neck meat may be better than samples from sheep and bovine. The content of IMP and Inosine were higher in both donkey and swine, and the content of IMP in the donkey neck was higher than that in the swine neck. IMP is a very strong freshener and is the main flavoring substance. IMP contributes mostly to the “umami” taste and has been widely used as a flavor enhancer to increase palatability (Dashdorj et al., 2015). It can be inferred that the taste of the donkey neck meat is superior to other neck meats. GMP and IMP are the most representative umami nucleotides (Mau et al., 1997). Their flavor thresholds are 12.50 mg/100 g and 25.00 mg/100 g, respectively, by calculating the neck meat of donkey, swine, bovine, and sheep. The TAV was used to determine the degree of influence of flavor nucleotides on the taste of the meat. The results were shown in Table 8. A TAV value greater than 1, indicating that the taste of the nucleotide can be perceived by people taste, and a TAV value of less than 1 cannot be perceived. Table 8 showed that the TAV values of GMP in the neck of the four meats were all below 1, indicating that AMP has no significant contribution to the sweetness characteristics of the neck’s meats. The TAV value of IMP in the donkey and swine was greater than 1, indicating that IMP has a significant contribution to the umami characteristics of the donkey neck and swine neck. In donkey neck meat, ATP is rapidly degraded to IMP under the action of endogenous enzymes, but IMP degradation is very slow, IMP is mainly accumulated in animals (Xin et al., 2014) so the content of IMP is higher than other related substances. IMP is the main flavor nucleotide of donkey and swine neck meat, while the IMP in bovine and sheep neck meat had a TAV value of <1, which is not the main flavor nucleotide.
| Taste nucleotide | Donkey | Swine | Bovine | Sheep |
|---|---|---|---|---|
| GMP | 0.73±0.13a | 0.39±0.09b | 0.80±0.12a | 0.26±0.04c |
| IMP | 2.86±0.21a | 2.19±0.09b | 0.13±0.01d | 0.34±0.03c |
The simultaneous presence of taste amino acids and taste nucleotides can produce a synergistic effect and significantly improve the umami taste of meat foods. In terms of taste amino acids, the sum of the specific gravity of sweet and umami amino acids in donkey neck meat were higher than that of other animal neck meats. The content of sweet amino acid Ala and umami amino acid Glu are prominent, that is, compared with the neck meat of other samples, the taste of the donkey neck meat was more sweet; in terms of taste nucleotides, the TAV value of IMP is donkey meat>swine>sheep>bovine studied have shown that Glu and Ala can synergize with IMP to enhance the umami taste, and the presence of IMP can also enhance the sweetness intensity of sweet amino acids. In the end, IMP and the sweet umami amino acid represented by Ala and Glu were umami substances in the donkey neck meat, and they synergistically responsible for its more delicious taste. In summary, the donkey neck meat has a certain degree of nutritional value and significant taste characteristics, which is an important basis for people to lay their choice on the donkey neck meat.
Conclusion
Our investigation demonstrates that the aldehydes are the main volatile flavors in the donkey neck meat, and particularly n-hexanal, the most abundant volatile flavor, is mainly derived from the oxidative decomposition of oleic acid, linoleic acid and arachidonic acid. The comparison with unsaturated fatty acid in donkey, beef, pork and lamb neck meat, evidenced that aldehydes in donkey neck meat were more abundant than other animals due to higher unsaturated fatty acid contained, which significantly influenced meat flavor. According to the analysis of nonvolatile flavor substances that confirmed that donkey neck meat possesses unique flavor, attributed to presence of umami substances such as IMP, Ala and Glu inside. Therefore, there is significant difference between donkey meat and other meats (swine, bovine and sheep) in flavor and nutrients contents.













