Introduction
Skeletal muscle consists of white and red muscle fibers whose ratio has a directly effect on meat quality. Red muscles with high myoglobin content mainly contain oxidative type I and oxidative/glycolytic type IIA fibers while white muscles contain glycolytic type IIB fibers with low myoglobin content (Morita et al., 1970). Fiber type compositions (FTCs) of skeletal muscles are affected by the location and function of the muscle within an animal. FTC in muscle is a major determinant factor of meat quality because contractile of myofibrils and metabolic properties of muscle are differentiated by types of muscle fiber (Joo et al., 2013). Previous studies have shown that these varying FTCs in porcine muscles are related to pork quality traits such as meat color, water-holding capacity, and tenderness (Choi and Kim, 2009; Jeong et al., 2017; Kim et al., 2013; Kim et al., 2018; Lee et al., 2012; Ryu and Kim, 2005; Ryu et al., 2008). However, few studies have reported the relationship between FTC in porcine muscles and pork taste-traits.
Taste plays an important role in sensory properties of meat together with other palatability traits such as flavor, texture, and juiciness. Meat has many taste-active substances such as free amino acids and adenosine 5’-triphosphate (ATP) metabolites. It is known that nucleotides and glutamic acid contribute to meat tastes, including delicious, umami, and brothy tastes (Nishimura, 1998, Nishimura et al, 1988). These components relative to umami have been considered influential contributors to the oro-sensory (sensory perception on taste) quality of meat (Fuke and Konosu, 1991). Especially, inosine monophosphate (IMP) in pork contributes to the preference of a human panel (Kawai et al., 2002; Okumura et al., 1996). IMP is released from pork muscle in the initial phase of moist heat cooking (Sasaki et al., 2007). According to Sasaki et al. (2005), the concentration of IMP in a pork water-extract varies among genetic species. Based on these findings, it is hypothesized that there are differences in content of nucleotides between porcine muscles because the rate of postmortem metabolism depends on the relative amount of muscle fiber types (Ryu and Kim, 2005; Hwang et al., 2010), resulting in different pork taste.
Many studies have used sensory evaluations to investigate meat quality and taste. However, the subjectivity and low reproducibility of sensory evaluations have often been criticized. For these reasons, an electronic tongue system has been developed to evaluate taste quality and intensity of many foods (Toko, 1996; Toko, 1998). It has been applied to meat and meat products (Chikuni et al., 2010; Okumura et al., 2004; Sasaki et al., 2007). Therefore, the objective of this study was to evaluate taste properties of three porcine muscles using an electronic tongue system in order to understand the relationship between FTC in porcine muscles and pork taste-traits.
Materials and Methods
Three porcine muscles, longissimus lumborum (LL), psoas major (PM), and infra spinam (IS), were excised from carcasses of eight crossbred (Landrace×Yorkshire×Duroc), castrated male pigs. The average live weight of these pigs was 110.5±2.62 kg. Pigs were slaughtered by electrical stunning. Approximately 10±0.5 g of each muscle was taken and frozen in isopentane liquid nitrogen for histochemical analysis within 45 min postmortem. These samples were then stored at –80℃ until subsequent analyses. Meat quality were carried out after 36 h postmortem. For content of nucleotides and electronic taste-traits analysis, the analyses were performed after completing meat quality analysis. Three repetition were conducted and all measurements were made at 48 h postmortem.
Serial transverse muscle sections with entire blocks (1.0×1.0×1.5 cm) mounted onto glass slides were cut into 10 μm thin slice using a cryostat microtom (HM525, Microm GmbH, Germany) at –20℃. The measurement of myosin adenosine triphosphatase (mATP) activities of the samples were based on Brooke and Kaiser (1970) after acid pre-incubation at pH 4.63. The stained sections was examined using an image analysis system (Image-Pro®plus 5.1, Media Cybernetics Inc., USA). The muscle fibers were differentiated into fiber type I, IIA, and IIB according to the nomenclature of Brooke and Kaiser (1970). Approximately 600 fibers per sample were counted and FTC was determined by fiber area percentages which was the ratio of total cross-sectional area of each fiber type to total fiber area measured.
Meat sample (5 g) was homogenized in a 50 mL conical tube with 20 mL of 0.5 M perchloric acid in the ice bath for 1 min based on the modified method of Yang et al. (2002). The extraction mixture was centrifuged at 3,000×g for 15 min at 4℃. Supernatant was filtered with Whatman filter paper No. 1. The residuum was mixed with 10 mL of 0.5 M perchloric acid. The mixture was again homogenized and filtered. The filtrate was neutralized to pH 6 with 5 M potassium hydroxide. The neutralized filtrate was centrifuged (3,000×g at 4℃ for 10 min) and filtered with Whatman No. 4. The filtered supernatant was added with 0.5 M perchloric acid (pH 6.0) to reach final volume of 50 mL. The solution was filtered again through a 0.45 μm filter and stored at –25℃ prior to analysis. Analysis of nucleotides was performed using an Agilent 1100 HPLC system with Eclipse Plus C18 column (4.6×100 mm, 3.5 μm). Mobile phase A consisted of 0.06 M K2HPO4 and 0.04 M KH2PO4 adjusted to pH 7.0. Mobile B consisted 80% methanol and 20% Mili-Q water. Peaks were detected and analyzed at 254 nm with a diode array detector (DAD). HPLC separation was achieved using continuous gradient elution between Phase A and Phase B. The elution program of Phase B was: 0 min (0%), 10 min (0%), 11 min (100%), and 12 min (0%). The program then took 4 min to return to initial conditions and stabilize. Flow rate of the mobile phase was set at 1.2 mL/min with 20 μL injection volume. Total retention time was about 5 min and the gradient was run for 16 min to ensure full separation. All ATP, ADP, AMP, IMP, inosine, and hypoxanthine in samples were identified by comparison with retention time of standards.
Color measurements of different porcine muscles were performed using a colorimeter (Minolta CR -300, Minolta Co., Japan) that was standardized with a white plate (Y=93.5, X=0.3132, y=0.3198) before measuring. Color parameters were expressed as L* (lightness), a* (redness), b* (yellowness), polar-coordinate Chroma (C*), and hue angle (h°).
Water-holding capacity (WHC) of all samples was evaluated by released water (RW) % and cooking loss (CL) %. CL % was determined as described by Hwang et al. (2010) by weight different before and after cooking. RW % was based on method of Joo (2018). Approximately 3.0 g of meat sample was placed on a filter-paper between two thin plastic films. A load of 2.5 kg was then applied for 5 min. After accurately removing the compressed meat sample, the damp filter-paper and two plastic films were rapidly weighed. The percentage of RW % was calculated as follows: RW %=[(damp filter-paper and plastic films weight)–(filter-paper and plastic films weight)/meat sample weight]×100.
Warner–Bratzler shear force (WBSF) values were measured using an Instron Universal testing Machine (Model 4400, Instron Co., USA) with a V shaped shear blade. Samples (1.0 cm–diameter cores obtained from muscle) were cooked to have an internal temperature of 70℃ for 30 min. Peak force was obtained using 100 N load cell tension applied at a crosshead speed of 250 mm/min. The full-scale load was 50 kg.
An electronic tongue system (INSENT SA402B electric taste sensing system, INSENT, Tokyo, Japan) was used to determine pork taste-traits. This system is composed of five taste sensors of polymer membranes fixing different lipids. These sensors, CA0, C00, AE1, AAE, and CT0, are designed to respond to individual tastes of sourness, bitterness, astringency, umami, and saltiness, respectively (Toko, 1996; Toko, 1998). Fresh 30 mM KCl solution containing 0.3 mM tartaric acid (corresponding to saliva) was used as the reference solution (RS). It was also used to rinse electrodes after every measurement. Vr (membrane potential in reference solution) is the potential when the electrode is dipped into the RS for the first time and Vs (membrane potential in sample solution) is another potential for the sample solution or suspension. Vr’ (new membrane potential in reference solution) is the new potential of the RS when the electrode is dipped into the RS again. CPA (change of membrane potential caused by adsorption) is the difference (Vr’–Vr) between potentials of the RS before and after sample measurement. It represents aftertaste. Each measuring time was set at 30 sec. Electrodes were rinsed after each measurement.
Each porcine muscle sample was measured after electric potentials of all membranes had been stabilized in standard pork taste (SPT) solution. A synthetic solution containing 0.02% lactic acid (sourness), 0.4% monosodium glutamate (umami), 0.001% quinine hydrochloride (bitterness), 0.05% sodium chloride (saltiness), and 0.8% sucrose (sweetness) was used as SPT solution. Sample solutions were prepared by extracting ground porcine muscles (100 g) with 400 mL hot water (1:4, w/v) of 95℃ for 10 min followed by centrifugation at 3,000×g for 10 min before analysis. All measurements were made at room temperature of 28℃.
All experimental data were analyzed by analysis of variance (ANOVA) procedure of statistical analysis systems (SAS, 2002). Duncan's multiple range test was used to determine significant differences among means at 5% level of significance (SAS, 2002). Pearson correlation coefficients were used to determine the relationship between FTC and taste-traits of electronic tongue system using partial correlation coefficients (SAS, 2002).
Results and Discussion
A clear difference in FTC was observed among three porcine muscles (Table 1). IS muscle had significantly higher proportion of type I fibers than LL and PM muscles (p<0.05). There was no significant difference in the proportion of type I fibers between PM and IS muscles. The proportion of type IIA fibers was significantly lower in LL muscle than that in other muscles (p<0.05). The proportion of type IIA was not significantly different between PM and IS muscles. There were significantly differences in the proportion of type IIB fibers among LL, PM, and IS muscles. LL muscle had the highest proportion of type IIB fibers while IS muscle showed the lowest proportion of type IIB fibers among three porcine muscles.
Joo et al. (2013) have indicated that histochemical characteristics of muscle fiber depend on muscle location and function in animals. The higher proportion of type IIB fibers in LL muscle was similar to results of Kim et al. (2013) and Realini et al. (2013). The higher proportion of type I fibers in IS muscle was expected because deep muscles involved in maintaining posture are more oxidative and containing more type I fibers than more superficial muscles involved in rapid movements (Joo et al., 2013). Since this FTC variation between muscles is directly related to metabolic and contractile properties, there could be considerable variations in pork quality characteristics and taste-traits between various porcine muscles within a carcass.
As expected, significant differences in pork quality traits were observed among three porcine muscles (Table 2). LL muscle had significantly higher L* values while IS and PM muscles had significantly higher a* and b* values compared to other muscles (p<0.05). RW % was significantly higher in LL muscle than that in PM and IS muscles (p<0.05), although there was no significant difference in cooking loss % among the three muscles. There were significant differences in WBSF values among three muscles (p<0.05), with IS having the highest WBSF value, followed by LL and PM muscle.
A few studies have been conducted to compare pork quality traits between porcine muscles in relation to FTC (Kim et al., 2018; Realini et al., 2013; Ruusunen and Puolanne, 2004). Most of these studies involving pork quality traits and FTC have been done mainly using the longissimus dorsi muscle. It is well known that variation in pork quality is related to heterogeneity in glycogen depletion between different muscle fiber types (Klont et al., 1998). Generally, glycolysis and onset of rigor mortis are faster in white muscles than those in red muscles. Therefore, the higher L* value in LL muscle is expected because it has a high glycolytic capacity (Joo et al., 2013; Kim et al., 2013). The higher a* value in IS and PM muscles in this study was similar to results of Realini et al. (2013), reporting that muscle redness was positively correlated with type I fiber but negatively associated with type II fiber. It is certain that the higher RW % in LL muscle is due to higher proportion of type IIB fibers because an increase in the proportion of large IIB fibers causes poor water-holding capacity of pork (Kim et al., 2013). Also, the higher WBSF value in IS muscle is probably due to higher proportion of type I fibers. Fiber type I has a positive correlation with shear force values in contrast fiber type IIB has a negative correlation to meat tenderness (Kim et al., 2013; Ryu and Kim, 2005).
Contents of nucleotide compounds in three porcine muscles are shown in Fig. 1. IMP showed the highest content (148–447 mg/100 g), followed by inosine (28–39 mg/100 g), hypoxanthine (7.1–18.6 mg/100 g), ADP (5.7–8.0 mg/100 g), AMP (2.0–2.6 mg/100 g), and ATP (1.1–2.2 mg/100 g) in porcine muscles at 48 h postmortem. There were significantly differences in contents of all nucleotide compounds among three muscles (p<0.05). IS muscle had the highest IMP content while LL muscle had the lowest IMP content (p<0.05). In contrast, LL muscle had significantly higher hypoxanthine content than PM and IS muscles (p<0.05). IS muscle had significantly higher ATP and ADP contents but lower inosine content than LL and PM muscles (p<0.05).
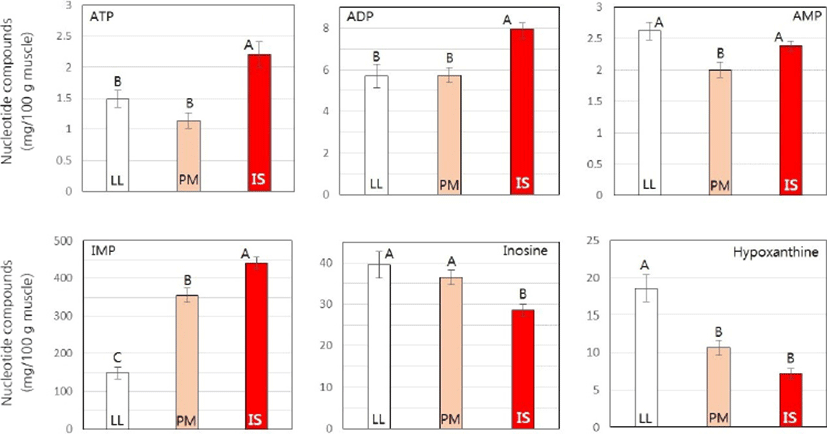
Many studies have shown that IMP contributes to meat taste properties such as delicious, umami, and brothy tastes (Fuke and Konosu, 1991; Kawai et al., 2002; Nishimura et al., 1988; Shi et al., 2017). The IMP content relative to umami has been considered an important contributor to the oro-sensory quality of meat (Fuke and Konosu, 1991). Okumura et al. (1996) have reported that IMP in pork can positively influence the panelist preference. According to Sasaki et al. (2007), IMP is released from the pork muscle during the initial phase of moist heat cooking. The concentration of IMP in a water-extract of pork muscle is different among genetic pig species (Sasaki et al., 2005). In the present study, it was clearly shown that the concentration of IMP was significantly different among three porcine muscles which had different FTCs. These three porcine muscles also had different concentrations of hypoxanthine related to bitter taste (Ozogul et al., 2010). Therefore, it is easily expected that taste-traits are also different among these three muscles due to different concentrations of nucleotides. In this regard, our previous studies have shown differences in sensory evaluation scores among various bovine muscles with different FTCs (Hwang et al., 2010; Jung et al., 2015; Jung et al., 2016).
Sensory evaluation results of three porcine muscles using the electronic tongue system are presented in Fig. 2. When relative taste-traits intensity values were computed from the SPT solution, all three porcine muscles showed negative values for sourness, astringency, and saltiness but positive values for umami, richness, and bitterness. All taste-traits intensity values except bitterness were significantly different among the three muscles (p<0.05). IS muscle had significantly higher umami and richness values but lower sourness value than LL and PM muscles (p<0.05). Sourness and astringency values of LL muscle were significantly higher than those of IS muscle (p<0.05). There were no significant differences in umami, sourness, or astringency values between LL and PM muscles. The saltiness values of LL muscle was significantly lower than that of IS or PM muscle (p<0.05).
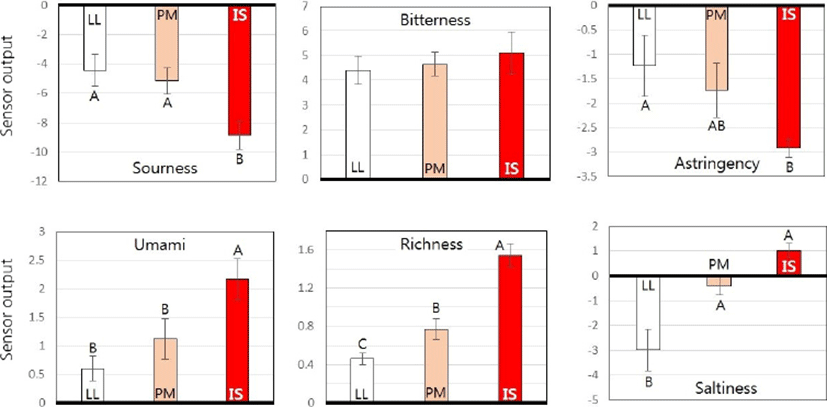
The higher umami value in IS muscle was expected because IS muscle showed the highest IMP content (Fig. 1). In contrast, umami value of LL muscle was lower than that of IS muscle due to the lowest IMP content in LL muscle. These results confirmed results of others showing that IMP content was related to umami taste of meat (Fuke and Konosu, 1991; Nishimura et al., 1989). Our data also showed that IS muscle had higher richness value reflecting persistent intensity of umami taste. The strong umami intensity in IS muscle is probably due to its higher proportion of type I fibers. In contrast, higher sourness and astringency values in LL muscle are due to its higher proportion of type IIA fibers. According to Chikuni et al. (2010), muscle fiber types are strongly related to differences in sour taste, with fast-type muscle having significantly lower pH due to its higher lactic acid content than slow-type muscle. In the present study, FTC in muscle not only affected sour taste, but also astringency taste.
Correlations of electronic taste-traits with FTC and nucleotide compounds are presented in Table 3. These results showed that contents of type I and type IIB fibers were inversely correlated to electronic taste-traits. The proportion of type IIB fiber was positively correlated with sourness and astringency but negatively correlated with saltiness. Interestingly, similar tendencies were found between hypoxanthine content and taste-traits. In contrast, the proportion of type I fiber was positively correlated with umami, richness, and saltiness but negatively correlated with sourness and astringency. IMP content showed correlation tendencies with electronic taste-traits similar to the proportion of type I fiber. These correlations clearly showed that sourness and astringency tastes were increased with increasing content of type IIB fibers in porcine muscles due to increase of hypoxanthine content. These results suggest that umami and richness tastes are increased with increasing content of type I and IIA fibers because of increased IMP content in porcine muscles.
Conclusions
The different composition of muscle fiber types were clearly observed among three porcine muscles. These differences affected content of nucleotide compounds and electronic taste-traits. Pork sourness and astringency tastes were increased with increasing content of type IIB fibers in porcine muscles due to increase of hypoxanthine content. Pork umami and richness tastes were also increased with increasing contents of type I and IIA fibers due to increase of IMP content in porcine muscles.













