Introduction
Beef is one of the most commonly consumed meats worldwide; it has a rich composition of key nutrients such as minerals, fatty acids, and vitamins, and has high protein contents (Hambidge and Krebs, 2007). However, beef is prone to chemical and microbial changes owing to its abundant nutrients and proper environment for microorganisms. Food packaging protects products against the external environment, plays a role as a marketing tool, and provides convenience to customer (Viana et al., 2005; Yam et al., 2005).
Vacuum packaging (VP) and modified atmosphere packaging (MAP) are popular packaging types for improved shelf life and the protection of fresh meat. VP has inhibitory effects on lipid oxidation and microorganisms in low oxygen condition. However, VP could be resulted in an unattractive color and a high deoxymyoglobin content as oxygen concentration (Jeremiah, 2001). Depending on the characteristics of the vacuum film, such as the number of layers, composition of materials, thickness of the film, and oxygen permeability, the maintenance of the nutritional value of meat during storage in the vacuum-packed state differs (Lee, 2010). MAP is a packaging method that maintains a bright red color by changing the gas composition in the packaging film. High contents of oxygen contribute to the red color of beef due to the formation of an oxymyoglobin at the meat surfaces using the MAP technique (Carpenter et al., 2001; Jeremiah, 2001). However, oxygen accelerates oxidative changes in beef; accordingly, it has negative effects on general consumer preference toward beef (Lund et al., 2007).
Vacuum skin packaging (VSP), which involves an upper packaging film and tray, is an advanced method recently applied for retail meat packaging. Meat is placed in trays and wrapped tightly in a heat-shrink film. VSP has various advantages, e.g., fewer wrinkles, slower microbial growth, and a longer shelf life, compared with VP (Vázquez et al., 2004). VSP of beef has been applied globally for decades, but is not yet widely applied in Korea.
In recent years, customers have been purchasing beef that has been subdivided into small portions and packaged by various methods for the retail market because it is convenience for cooking and refrigerated storage (Belcher, 2006). Therefore, meat processing industries produce small-scale or small-packaged beef to meet the consumer’s needs.
Therefore, The objective this study was to investigate the physicochemical, microbiological, and sensory properties of Hanwoo using different packaging methods at a small scale under refrigerated conditions.
Materials and Methods
Beef (eye of round, 1 grade) was obtained from carcasses of Hanwoo, Korean native cattle, at Taewoo Green Food Co., Korea. 200-g samples of beef were packaged by 4 methods: 1) wrapped packaging (WP), 2) 30% O2/70% CO2, referred to as MAP, 3) VP, and 4) VSP. In the MAP group, beef samples were packaged with a tray (M-50; Hongwoo Packaging System Co., Korea) and barrier film (L-sealer film; Hongwoo Packaging System Co.), using an MAP machine (MAP-H2; Hypervac Co., Korea). In the VP group, beef samples were packaged using three kinds of VP film: 1) VP1 was made in a 5-layer film [polyvinylidene chloride (PVDC) with O2 transmission rate of 1.04 mL/m2·day at 23°C, a thickness of approximately 48 µm using a Supra S bag (Schur Flexible, Germany)]. 2) VP2 samples was packaged in a 4-layer film (PVDC), with O2 transmission rate of 1.22 mL /m2·day at 23°C and a thickness of approximately 52 µm using S-ZUR Shrink (Mitsubishi Plastics Inc., Japan). 3) VP3 samples were packaged in a 7-layer film (nylon), with an O2 transmission rate of 26.54 mL/m2·day at 23°C and a thickness of approximately 55 µm using ADMAS® (KwangHee Co. Ltd., Korea). The VP machine was the Fj-880D (Jiwoo Engineering Co. Ltd., Korea). In the VSP group, beef samples were packaged with a skin tray (TPS-3-2, Hongwoo Packaging System Co., Korea), heat shrinkable film (TSE11-100422325, Multipack Korea Co. Ltd., Korea), and a VSP machine (MULTIVAC-T300, Multipack Korea Co. Ltd., Korea). The packaged beef samples were stored at 4±1°C. Sampling was carried out at days 0, 7, 14, 21, and 28.
The pH values were measured in packaged beef samples, using a pH meter (WTW, Germany). The pH was measured after homogenizing 5 g of each beef sample with 25 mL of distilled water. All samples were examined in triplicate.
The color of each packaged beef sample was determined at 4 defined areas on the surface of the sample, using a color meter (Minolta Chroma Meter CR-400; Minolta Co., Japan; illuminate C, calibrated with a white plate, lightness (L*) = +97.83, redness (a*) = -0.43, yellowness (b*) = +1.98). The color of beef was determined 30 min for blooming time after opening the outer packaging and is expressed as lightness (L*), redness (a*), and yellowness (b*) values.
Volatile basic nitrogen (mg%) was estimated to determine the extent of protein deterioration during refrigeration. VBN was measured according to modified methods of Pearson (1968). Briefly, 5 g of sample was mixed with distilled water and filtered with Whatman No.1 (Whatman International, UK). On the inner section, 50 µL of indicator and 1 mL of 0.01 N H3BO3 were set. On the outer section of the Conway microdiffusion cell, 1 mL of filtered sample solution and 50% K2CO3 solution were added. After incubation for 90 min at 37°C, the inner section solution was titrated with 0.02 N H2SO4.
where a indicates the amount (mL) of H2SO4 added to the sample for titration, b indicates the amount (mL) of H2SO4 added to the blank for titration, f represents 0.02 N H2SO4, N represents normality, and S represents the weight (g) of the beef sample.
Lipid oxidation was evaluated in triplicate by the TBARS method of Kim et al. (2015) with modifications. The results are shown as milligrams of malondialdehyde (MDA) per kilogram of packaged beef sample. In brief, 10 g of each beef sample was blended with 50 mL of distilled water for 2 min using a homogenizer (AM-7, Nihonseiki, Kaisha Ltd., Japan) and transferred to a distillation tube. The cup used for mixing was cleaned with 47.5 mL of distilled water and added to the same distillation flask along with 2.5 mL of 4 N HCl and antifoaming agent (KMK-73, Shin-Etsu Silicone Co. Ltd., Korea). The mixture was distilled and 5 mL of 0.02 M TBA in 90% acetic acid was added to a test tube containing 5 mL of the distillate and mixed. The tubes were closed, heated in boiling water for 30 min for chromogen development, and cooled to room temperature. Absorbance was measured at 538 nm against a blank (mixed with 5 mL of distilled water and 5 mL of TBA reagent) using a UV/VIS spectrophotometer (Optizen 2120 UV Plus; Mecasys Co. Ltd., Korea).
A microbiological analysis of packaged beef was performed by determining the total viable count (TVC) as well as counts of psychrotrophic bacteria (PSY), lactic acid bacteria (LAB), coliform, anaerobic bacteria (ANB), Escherichia coli O157:H7, and Salmonella spp.
A total of 10 g of each packaged beef sample was placed in a sterile filter bag (Interscience Laboratory Inc., USA) and 90 mL of 0.1% peptone water was added. Samples were homogenized using a stomacher (IUL Instruments, Spain) for 15 s at room temperature. The homogenates were diluted 10-fold with 0.1% peptone water and spread on media.
The microbiology analyses of packaged beef were depended on Korean Food Standards Codex with some modification. TVC and PSY counts were obtained on tryptic soy agar (Difco Laboratories, USA) and incubated at 37°C for 24 h and 25°C for 72 h, respectively. LAB counts were determined by incubation on Lactobacilli MRS agar (Difco Laboratories) at 37°C for 24 h. Coliforms were counted after incubation on Desoxycholate Lactose Agar (Difco Laboratories) at 37°C for 24–48 h. Anaerobic bacteria were counted after incubation on tryptic soy agar at 37°C for 24–48 h using an anaerobic jar with AnaeroPack–Anaero (Mitsubishi Gas Chemical Co., Inc., Japan). E. coli O157:H7 and Salmonella spp. were conducted according to Ministry of Food and Drug Safety (2015). All microbiology analyses were performed in duplicate and the colony counts are expressed as Log CFU/g.
A sensory analysis was performed in triplicate for each packaged beef sample by sensory panelists. A trained 6-member panel consisting of researchers from the Food Processing Research Center, Korea Food Research Institute was employed to evaluate the packaged beef. Selected panelists followed an established sensory evaluation procedure (Lawless and Heymann, 1998). Each packaged beef sample was evaluated for appearance, color, flavor, and overall acceptability. Each sample was unpacked and evaluated after 30 min for blooming time in order to avoid color difference between before and after blooming (Brewer et al., 2001). The appearance (1=extremely undesirable, 10=extremely desirable), color (1=extremely undesirable, 10=extremely desirable), flavor (1=extremely undesirable, 10=extremely desirable), and overall acceptability (1=extremely undesirable, 10=extremely desirable) of packaged samples were evaluated using a 10-point descriptive scale. This sensory analysis was based on the hedonic test described by Bergara-Almeida and da Silva (2002).
All experiments were replicated three times of each samples. All experimental results for packaged beef during refrigerated storage are expressed as means±standard deviation. A two-way analysis of variance and Duncan’s multiple range tests were performed to determine significant differences among groups, using PASW version 18 (IBM Inc., USA) (2009).
Results and Discussion
The pH and color of packaged beef during refrigerated storage are shown in Table 1. The pH values of all packaged beef tend to decrease during storage (p<0.05). The decreases in pH as storage time increased of packaged beef stored refrigerated condition were consistent with the results of Vázquez et al. (2004) and Kameník et al. (2014). The decrease in the pH of packaged beef during storage can be attributed to microbial metabolites, such as lactic acid or CO2 mainly produced by LAB (Paneras and Bloukas, 1994).
The lightness (L*) values of beef in MAP were significantly higher (p<0.05) than those of beef in other packaging types. The L* values for packaged beef were not significantly change except MAP and VSP during refrigerated storage. The L* values of MAP were higher than other packaging. VP3 showed lower L* value than VP1 and VP2. VSP was significantly higher than VP until day 7 (p<0.05), but was similar or lower after that. According to Lagerstedt et al. (2011), L* values of VSP in longissius dorsi were significantly higher than those of VP until day 14 and tended to be similar beginning at day 21. The redness (a*) values for WP and MAP significantly increased and decreased before and after 7 days of storage (p<0.05). The a* values of MAP samples decreased rapidly during refrigerated storage and reached a minimum at day 28. VP3 showed a high a* values until 14 days of storage, but decreased thereafter. VP3 has higher a* value in initial storage than in other vacuum films with different oxygen permeability, but resulted in higher microorganisms in the late of storage. The a* value of VSP were significantly higher or no difference compared to VP1 and VP2 until day 14, but it was significantly lower thereafter. In this study, a* values of VSP was less than VP during refrigerated storage. However, Taylor et al. (1990) reported that the red color of meat in VSP showed stable retention during refrigerated storage for 28 days. The a* patterns is decreased for MAP and increase or maintain for VP and VSP during refrigerated storage were similar to those reported by Li et al. (2012). The yellowness (b*) values tended to decrease significantly after 7 day for all packaging methods (p<0.05). The b* values for MAP samples were significantly higher than those for other packaging methods during refrigerated storage. The color of beef is influenced by oxygen. VP and VSP samples were in the vacuum-packed state, lacking oxygen. Therefore, myoglobin does not become oxymyoglobin, resulting in a dark-red color with a high a* value. In contrast, WP and MAP enable metmyoglobin production, resulting in a decrease in a* values. In beef, differences in the a* value influence consumer acceptance (Font-i-Furnols and Guerrero, 2014). In total, VP and VSP showed a higher a* values than WP and MAP. The a* values of VSP were higher than other packaging until day 14 of storage, and after that VP had higher a* values.
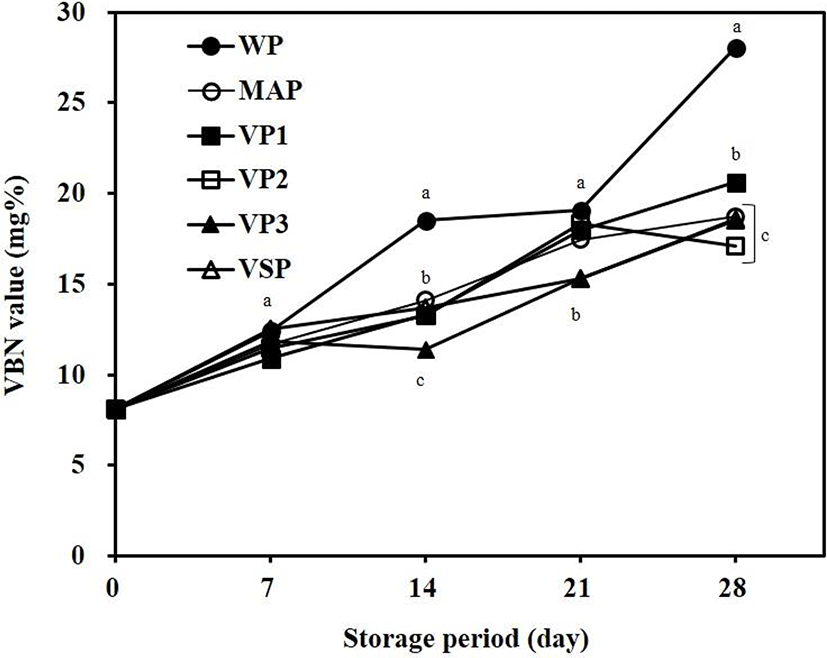
VBN is an indicator of protein decay in meat products. The protein in meat is hydrolyzed to amino acids, peptides, and inorganic nitrogen, increasing levels of volatile bases and adenosine monophosphate (AMP) by amino acid decarboxylase, enzymes, and microorganisms (Kohsaka, 1975). The VBN value of meat is considered to indicate decay if it exceeds 20 mg% (Choi et al., 2002). Fig. 1 shows the changes in VBN values for packaged beef during refrigerated storage. VBN values for all packaged beef samples increased during storage. VBN values were higher for WP samples than for other packaged beef during refrigerated storage. WP showed VBN values of about 20 mg% on day 14, which has a sensory influence. The VBN value for VP1 was 20.61 mg%, which was significantly higher than VP2 and VP3 at day 28 of storage (p<0.05). In the other packages, the VBN values did not exceed 20 mg% during the storage period. The increase in the VBN value seems to be proportional to the growth of microorganisms in packaged beef (Jay, 1992).
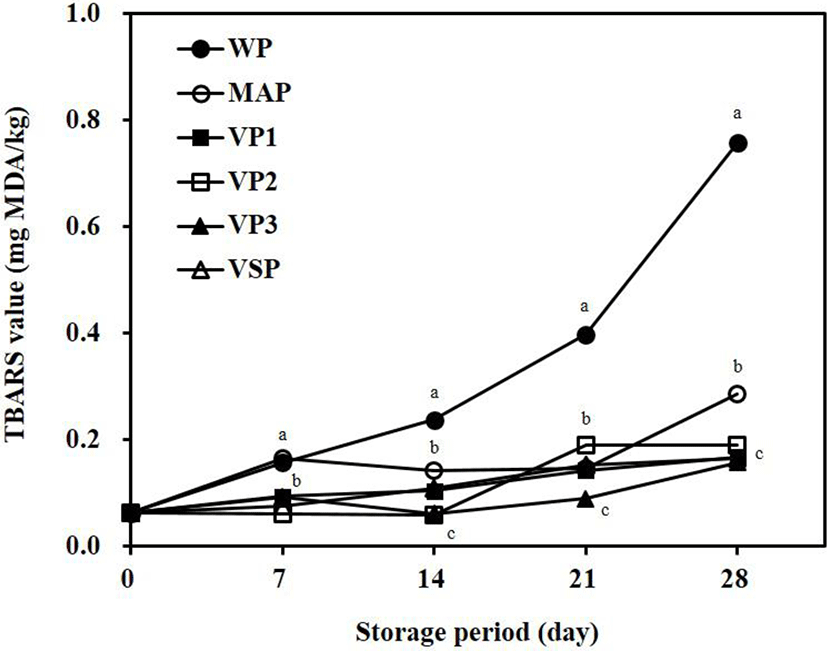
Rancidity is a type of chemical spoilage that occurs in meat and has negative effects on the color, taste, and flavor of meat (Kohsaka, 1975; Faustman and Cassens, 1990). If the TBARS value is higher than 0.5 mg MA/kg, a rancid odor can be detected (Choi et al., 2011; Sheard et al., 2000). Fig. 2 shows changes in the TBARS value for packaged beef during refrigerated storage. On day 28, the TBARS value for WP was 0.76 mg MA/kg, which was higher than those for other packaged beef samples. TBARS values for WP and MAP were higher than those for VP and VSP due to the direct contact of beef samples with oxygen. These results were similar to the TBARS values for MAP and air packaging of minced beef meat during refrigerated storage for 14 days reported by Esmer et al. (2011). Lorenzo and Gómez (2012) reported that TBARS values for vacuum packaging were significantly lower than those for air packaging and MAP using ostrich meat during refrigerated storage (p<0.05).
A microbiological analysis of packaged beef is summarized in Table 2. The Ministry of Food and Drug Safety (2014) recommends that beef has a TVC of less than 7 Log CFU/g. The initial TVC was less than 2 Log CFU/g for all packaged beef samples. For WP, TVC increased rapidly until day 21, exceeding the TVC limit. For MAP, TVC was significantly higher than those for other packaging at day 7 (p<0.05), but was maintained at 4.87~5.98 Log CFU/g for day 28. Nassu et al. (2010) reported that the growth of microorganisms could be inhibited by an extended lag phase and generation time when carbon dioxide concentrations exceed 20% in MAP. For VP1, TVC increased steadily and exceeded the limit at day 28. However, for VP2, TVC was low throughout the storage period. For VSP, TVC values were significantly lower at all storage days compared to those for other packaging types and were less than 5 Log CFU/g at day 28 (p<0.05). Lorenzo and Gómez (2012) reported that overwrap packaging results in a higher TVC than those of MAP and VP in foal meat.
PSY, typically Pseudomonas spp., are related to meat spoilage (Ercolini et al., 2009; Jay et al., 2003). Initial PSY counts in packaged beef were less than 2 Log CFU/g. The PSY counts for WP, MAP, VP1, and VP3 were close to or exceeded to 7 Log CFU/g during refrigerated storage. For VP2 and VSP, PSY counts were significantly lower than those for other packaging at day 28 (p<0.05). Especially, the difference of PSY counts of VP1, VP2, and VP3 depended on oxygen transmission rate. According to Kameník et al. (2014), PSY counts for VSP were lower than those for MAP and VP samples in beef and pork meat until day 21 during storage at 2.0±0.5°C.
LAB acts as spoilage bacteria that shorten the shelf life of meat (Nychas et al., 2008; Pothakos et al., 2015). The initial LAB count was less than 2 Log CFU/g (Table 2). For WP, LAB increased to 7 Log CFU/g at day 21. For MAP and VSP, LAB counts were significantly less abundant during the storage period (p<0.05). For VP1 and VP3, LAB were significantly more abundant than for other packaging methods at day 28 of storage (p<0.05).
Coliform serves as an indicator of hygiene status in food (Yalçin et al., 2001). According to the Ministry of Food and Drug Safety (2014), the recommended standard coliform count is less than 3 Log CFU/g in beef. For packaging except WP, MAP, and VP1, coliform counts did not exceed the standard value during the storage period.
Anaerobic bacteria are able to grow without oxygen. For VSP, the number of ANB was significantly lower than that for VP for all storage periods, except on day 7 (p<0.05). These results indicated that aerobic bacteria, anaerobic bacteria, and coliform counts were lower for VSP than for traditional vacuum packaging in beef, consistent with the results of Vázquez et al. (2004).
Salmonella spp. and E. coli O157:H7 were not detected in any packaged beef samples during entire storage period (data not shown).
In summary, beef samples in WP were the first to lose microbiological safety from day 14 to day 21. In MAP, microorganisms did not grow more than WP during storage. However, the growth rate of microorganisms in the initial storage period was faster than other packaging methods and there were many PSY and coliform compared to VP and VSP. Among VPs, VP1, and VP3 were not suitable for long-term refrigerated storage of beef. Compared with VP2 and VSP, VSP showed significantly low microbial growth than VP2. Therefore, VSP was the best microbiological safety in beef among the experimented packaging methods.
The sensory evaluation results for packaged beef are presented in Table 3. For all packaging types, except for WP, no significant changes in sensory characteristics according to storage period were observed. On day 28 of storage, VP samples received significantly higher scores than those of other packaging with respect to appearance (p<0.05). There were no significant differences in the color of packaged beef until day 14 for each packaging type (p>0.05). However, there was a significant difference from day 21 (p<0.05). With respect to color, WP and VSP scores were lower than VP scores. The flavor scores for VSP beef samples decreased on day 21, while those for MAP and VP samples were maintained until day 28. There was no significant difference in overall acceptability from 7 day of storage. After day 7 of storage, MAP and VP samples showed the highest scores, followed by VSP and WP samples. Lagerstedt et al. (2011) reported significantly higher scores for beef steak tenderness, flavor, and juiciness using VP and VSP than MAP during 21 days of refrigerated storage (p<0.05).
Conclusion
The physicochemical, microbiological, and sensory properties of Hanwoo packaged at a small scale by various packaging methods during cold storage were evaluated. The pH values tended to decrease for all packaged beef. The a* values for VP and VSP was stable. Microbiological analysis of packaged beef showed that microbiological safety of VSP was higher than other packaging until day 28 of refrigerated storage. VSP also showed low TBARS and VBN values during refrigerated storage with VP. In sensory analysis, VSP showed acceptable value until day 14. Therefore, we can conclude that VSP is suitable for Hanwoo small-scale packaging. The results of this study will help determine the shelf life of Hanwoo according to the packaging method.













