Introduction
Immunosuppression is an abnormal physiological condition with temporary or long-lasting immune defects that might increase susceptibility to certain infectious diseases (Geha et al., 2007). Many anti-microbial drugs have been developed and tested for treating immunocompromised person after infection. However, prolonged use of anti-microbial drugs often leads to several adverse effects such as allergic reaction, diarrhea, dysbiosis, and drug-induced liver injury (Mohsen et al., 2020). Therefore, the development of a more effective and safer method is required to treat such immunosuppressive diseases.
Probiotics are heterogenous microorganisms that give health benefits when they are consumed or applied to the host (de Melo Pereira et al., 2018). A recent study has provided evidence that consumption of some probiotics might be an effective therapeutic tool to increase overall immune responses of an immunosuppressed host (Gramajo Lopez et al., 2021). L. plantarum CJLP243 was originally derived from Kimchi, a traditional Korean food. It has been developed as food supplement to enhance helper T1 (Th1) activity (Lee et al., 2011; Won et al., 2011). Consumption of CJLP243 can alleviate the symptom of Th2 prone diseases such as birch pollen-induced allergic rhinitis by rebalancing Th1/Th2 responses and increasing cellular immunity (Choi et al., 2018).
In this study, consumption of three different probiotics of L. plantarum including CJLP243, CJW55-10, and CJLP475 were tested for their ability to induce cellular immunity in a cyclophosphamide (CPP)-induced immunosuppressive mouse model. As a result, the consumption of these probiotics promoted interferon-γ (IFN-γ), interleukin-1β (IL-1β), IL-6, and tumor necrosis factor-α (TNF-α) production, increased NK cell activity, and facilitated immune cell proliferation in immunodeficient mice. Our data suggested that consumption of selected probiotics can be developed as a food additive to induce cellular immunity.
Materials and Methods
Isolation of L. plantarum CJLP243 was described previously (Lee et al., 2011). CJW55-10 was isolated from kimch and CJLP475 was isolated from fermented soybean paste according to a previously established method (Lee et al., 2011). Each probiotics was sub-cultured more than three times prior to in vitro experimental analysis.
C57BL/6 mice (female, 6–8 weeks old) were purchased from Orient Bio (Seongnam, Korea) and acclimated for one week prior to experiments. Mice were fed a standard rodent diet with purified water ad libitum and kept at 20°C–24°C with 40%–60% humidity in Korea University animal facility under a 12 h/12 h light-dark cycle. Mice received proper care in accordance with a protocol approved by the Institutional Animal Care and Use Committee (IACUC) of Korea University (protocol number: KUIACUC-2020-0052).
BM cells were isolated from femurs and tibias of mice and resuspended to 1×106 cells/mL in 30% L929-conditioned DMEM medium supplemented with 20% (v/v) FBS, 5×10–5 M β-mercaptoethanol, 20 mM HEPES, 1 mM sodium pyruvate, and 1% penicillin/streptomycin (Weischenfeldt and Porse, 2008). Culture medium was replenished four days after culture. On day 7, cells were harvested and the degree of BMDM differentiation was measured by flow cytometry analyses after staining with PE conjugated anti-mouse F4/80 (BioLegend, San Diego, CA, USA, cat. #123110) and antigen presenting cells (APC) conjugated anti-CD11b (BioLegend, cat. #101212) antibodies. More than 90% of cells displayed F4/80+CD11b+ phenotype.
BMDMs (5×104 cell/well) were seeded into a 96-well plate in 200 μL of RPMI-10 [RPMI medium supplemented with 10% (v/v) FBS and 1% penicillin/streptomycin]. Each probiotics (6×106 CFU/mL) was co-cultured with BMDMs in a humidified 5% CO2 atmosphere at 37°C for 3 d. As a positive control, lipopolysaccharide (LPS) (10 μg/mL) (Merck, Darmstadt, Germany) was used to treat BMDMs without incubation with each probiotics. As a negative control, vehicle (phosphate buffered saline, PBS) was also used to treat BMDMs. After co-culture, IFN-β (R&D Systems, Minneapolis, MN, USA, cat. #42400-1), IL-1β (Thermo Fisher Scientific, Waltham, MA, USA, cat. #BMS6002), IL-6 (Thermo Fisher Scientific, cat. #BMS603-2), IL-12 (Thermo Fisher Scientific, cat. #BMS616), and TNF-α (Thermo Fisher Scientific, cat. #BMS607-3) secretions from BMDMs were quantified by sandwich ELISA kits according to the manufacturer’s instructions.
To quantify mRNA transcript of inducible nitric oxide synthase (inos), total RNA was extracted from BMDMs using Trizol reagent (Thermo Fisher Scientific) according to the manufacturer’s instructions. It was then reverse transcribed into cDNA using a First-Strand cDNA Synthesis kit with random hexamer and SuperScript RT (Thermo Fisher Scientific). After cDNA synthesis, quantitative real-time PCR was conducted on a Bio-Rad CFX96 Real-Time Detection System (Bio-Rad, Hercules, CA, USA) using QGreenTM 2X qPCR Master Mix (GenDEPOT, Katy, TX, USA). Relative expression levels of inos mRNA transcripts were normalized to mouse Gapdh level. Primer sequences for mouse inos were 5′-CCAGTTGTGCATCGACCT-3′ (sense) and 5′-ATGCTCCATGGTCACCTC-3′ (anti-sense), resulting in a 139 bp product. Primer sequences for mouse gapdh were 5′-ATGGTGAAGGTCGGTGTGAA-3′ (sense) and 5′-GGTCGTTGATGGCAACAATCTC-3′ (anti-sense), resulting in a 100 bp product.
Flow cytometry analyses were performed to determine surface expression of mouse MHC class II (Ab), CD80, and CD86 on BMDMs before stimulation and at 3 d after stimulation with each probiotics. For surface staining, FITC-conjugated anti-Ab antibody (Biolegend, San Diego, CA, USA, rat IgG2b, cat. #107605), PE conjugated anti-mouse CD80 antibody (BD Biosciences, San Jose, CA, USA, hamster IgG, cat. #553769), and PE conjugated anti-mouse CD86 antibody (BD Biosciences, rat IgG2a, cat. #553692) were used. FITC conjugated rat IgG2b antibody (BD Biosciences, cat. #553988), PE conjugated hamster IgG2 antibody (BioLegend, cat. #400908), and PE conjugated rat IgG2a antibody (Thermo Fisher Scientific, cat. #12-4321-81) were used as isotype controls. After staining, BMDMs were analyzed by flow cytometry using a FACSVerse with FACSuite software (BD Biosciences).
To confirm whether CPP treatment causes an immunodeficiency in mice, six of C57BL/6 mice were randomly divided into two groups (n=3 per group). One group of mice was received vehicle (PBS) and the other group of mice was intraperitoneally injected with CPP (150 mg/kg of body weight) in 200 μL of sterile PBS on day 0 and again on day 2. Three days after the first administration of CPP, all mice were sacrificed for monitoring immune cell populations using flow cytometry analyses. For monitoring thymocytes and T cell populations, FITC-conjugated anti-mouse CD3ε antibody (Biolegend, cat. #100714), PerCP-Cy5.5-conjugated anti-mouse CD4 antibody (BD Biosciences, cat. #550954) and APC-Cy7-conjugated anti-mouse CD8 antibody (Biolegend, cat. #100714) were used. For monitoring splenic B cells and NK cells, PE-conjugated anti-mouse B220 antibody (Biolegend, cat. #103208) and APC-conjugated anti-NK1.1 antibody (Invitrogen, Carlsbad, CA, USA, cat. #17-5941-81) were used.
After confirming the immunosuppressive effect of CPP treatment in mice, thirty of C57BL/6 mice were randomly divided into five groups (n=6 per group). One group of mice was received neither CPP nor probiotics and the rest four groups of mice were intraperitoneally injected with CPP (150 mg/kg of body weight) in 200 μL of sterile PBS on day 0 and again on day 2 to establish an immunocompromised animal model. On day 3, each mouse of the three CPP-treated groups started to receive 200 μL of PBS containing each probiotics (CJLP243, CJW55-10 or CJLP475) (1×1010 CFU per mouse) using a gavage needle every day for 14 consecutive days. The other CPP-treated group of mice started to receive 200 μL of vehicle (PBS) without probiotics starting from day 3 until day 17 as a negative control. On day 18, all mice were sacrificed for analyses (Choi et al., 2018).
After sacrificing mice, splenocytes from each animal were isolated and seeded into a 96-well plate at 1×106 cells/well in 200 μL of RPMI-10. Each probiotics (6×106 CFU/mL) was then co-cultured with splenocytes in a humidified 5% CO2 atmosphere at 37°C. As a positive control, LPS (10 μg/mL) was used to treat splenocytes without incubation with each probiotics. Three days after incubation, IFN-γ (Thermo Fisher Scientific, cat. #BMS606-2), IL-1β (Thermo Fisher Scientific, cat. #BMS6002), IL-6 (Thermo Fisher Scientific, cat. #BMS603-2), IL-12 (Thermo Fisher Scientific, cat. #BMS616), and TNF-α (Thermo Fisher Scientific, cat. #BMS607-3) secretions from splenocytes were quantified using sandwich ELISA kits according to the manufacturer’s instructions.
To measure immune cell proliferation after challenging with probiotics, splenocytes were incubated with 5 μM of carboxyfluorescein succinimidyl ester (CFSE) at 37°C for 10 min, washed with RPMI-10, and then cultured with each probiotics or LPS. Three days after culture, the proliferation of CFSE-labeled cells was measured by flow cytometry.
To measure NK cell activity from probiotics-treated splenocytes, splenic NK cells were used as effector cells and YAC-1 cells (ATCC, Manassas, VA, USA) were used as target cells. Briefly, splenocytes (1×106 cell/well) from sacrificed mice and YAC-1 cells (1×104 cell/well) were seeded to 96-well plates in 200 μL of RPMI-10 and co-incubated at 37°C for 6 h. The cytotoxicity of target cells (YAC-1 cells) was then measured with an LDH assay kit (Takara Bio, Shiga, Japan) according to the manufacturer’s instructions.
Results and Discussion
Professional antigen presenting cells (pAPCs) play a pivotal role in initiating immune responses. Accordingly, macrophages not only stimulate innate immunity by secreting several cytokines, but also initiate adaptive immunity by providing an activation signal to naïve T cells to become effector T cells (Kambayashi and Laufer, 2014). To test whether selected probiotics including CJLP243, CJW55-10, and CJLP475 could are able to induce macrophage activation, BMDMs were co-cultured with these probiotics. After co-culture, we measured macrophage activity was measured with three parameters. First, iNOS expression levels were measured by quantitative real-time RT-PCR. The iNOS has been shown to play a critical role in activating macrophage function. Its level is augmented after exposure to proinflammatory cytokines such as IL-1β and TNF-α (Ulisse et al., 2001). As expected, LPS-treated BMDMs significantly increased the mRNA level of inos compared to that of vehicle-treated BMDMs (Fig. 1A). Selected probiotics-treated BMDMs also increased mRNA levels of inos by more than 20 folds compared to vehicle-treated BMDMs (Fig. 1A). Especially, CJLP475-treated BMDMs displayed the highest fold increase of inos compared to vehicle-treated BMDMs (Fig. 1A).
Recently, immune modulatory role of probiotics is largely divided into two categories depending on a function of certain strain: Enhancing inflammatory responses or inhibiting inflammatory responses (Bui et al., 2015; Vincenzi et al., 2021). To determine immune modulatory effects of these probiotics, we measured levels of pro-inflammatory cytokines including IL-1β, IL-6, IL-12, and TNF-α. As a positive control, we measured levels of these cytokines from LPS-stimulated BMDMs. As a result, LPS-treated BMDMs secreted enormous amounts of these cytokines compared to vehicle-treated BMDMs (Figs. 1B and C). Compared to vehicle-treated group, IL-6, IL-12, and TNF-α were produced significantly from these probiotics-treated BMDMs (Fig. 1B). However, BMDMs stimulated by these probiotics did not produce IL-1β (Fig. 1B).
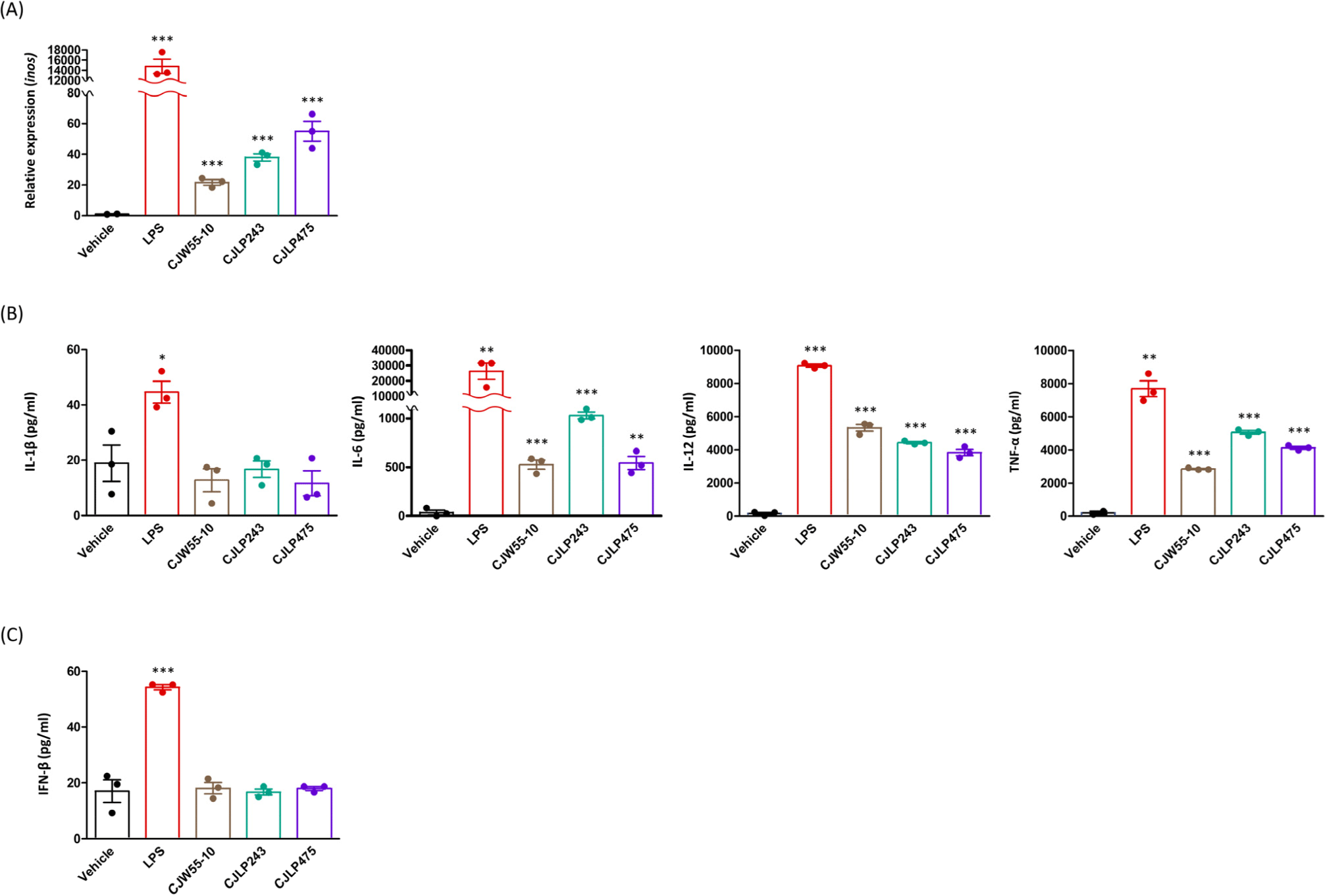
Notably, IL-12 is an essential cytokine to initiate Th1 immune response. It is currently developed as a potential immunological tool to destroy certain cancer (Nguyen et al., 2020). Similar to our results, previous observations have also indicated that certain probiotics strains can induce IL-12 production by macrophages (Shida et al., 2006). In addition, previous observations have demonstrated that probiotics can enhance anti-microbial responses by inducing type I IFNs (Gutierrez-Merino et al., 2020). Therefore, we also measured the production of IFN-β by BMDMs after stimulation with these probiotics. However, these probiotics did not stimulate macrophages to produce type I IFN (Fig. 1C).
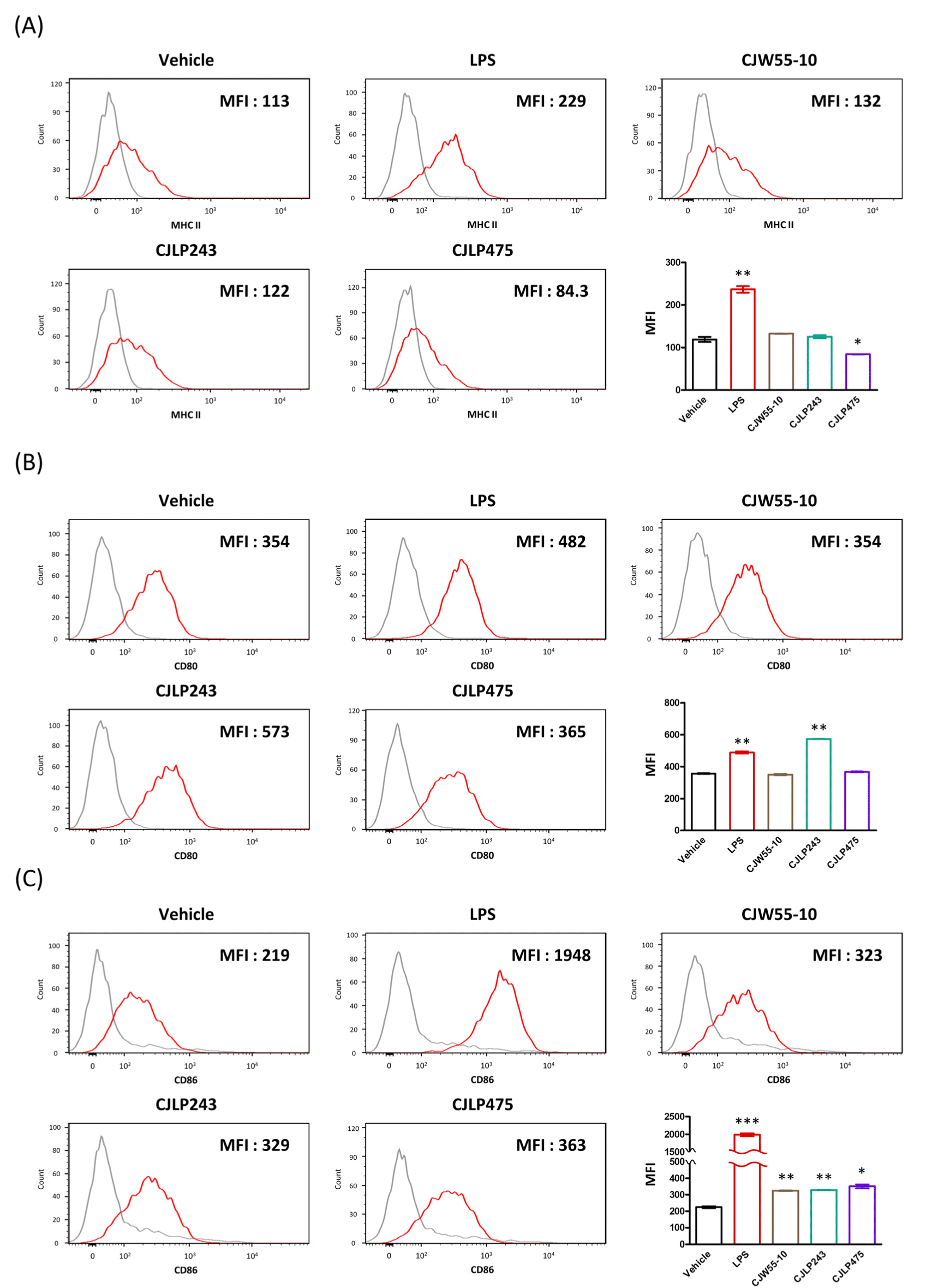
Two signals are required for stimulation of Naïve T cells to turn into effector T cells. The interaction between peptide antigen with MHC molecules on pAPCs and T cell receptor on naïve T cells delivers the first signal. The second signal is mediated by the interaction between CD80 or CD86 on pAPCs and CD28 on naïve T cells (Kambayashi and Laufer, 2014). Therefore, we measured expression levels of MHC class II (Ab) and co-stimulatory molecules (CD80 and CD86) to determine whether cell surface expression levels of these molecules were up-regulated on macrophages after stimulation with these probiotics. LPS-stimulated macrophages displayed increased expression levels of all three molecules (Ab, CD80, and CD86) (Fig. 2). Treatment with none of these probiotics led to increase expression of Ab on macrophages (Fig. 2A). However, the surface expression of either CD80 or CD86 was increased statistically when macrophages were treated with these probiotics (Figs. 2B and C). Combined evidence strongly suggest that macrophages stimulated with these probiotics can enhance immunity by increasing the production of iNOS, proinflammatory cytokines, and co-stimulatory molecules.
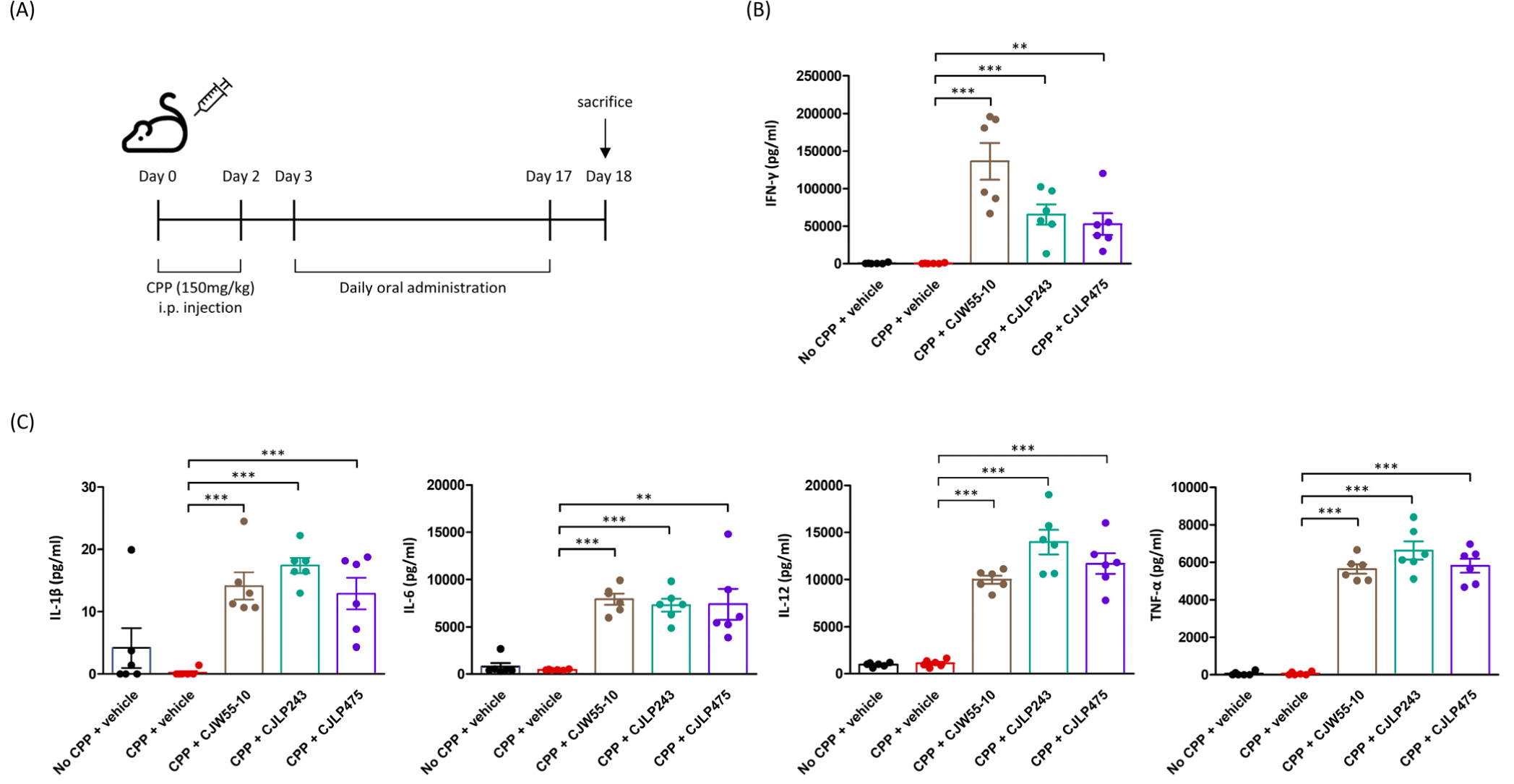
Since we found a stimulatory effect of these probiotics on macrophages in vitro, we further determined whether these probiotics could induce cell-mediated immunities in immunodeficient mice (Jimenez-Valera et al., 2003). CPP-treated mice are widely used in an immunocompromised animal model to test immunostimulatory role of several biological reagents. Treatment with CPP causes both myelopenia and leukopenia with reduced number of splenocytes within 5 d (Jimenez-Valera et al., 2003). To confirm the immunosuppressive effect of CPP in mice, we injected CPP (150 mg/kg) to mice twice with an interval of two days. Three days after the first administration of CPP, all mice were sacrificed and analyzed the changes of immune cell populations in thymus and spleen. As shown in Table 1, the numbers of thymocytes and T cells in thymus was severely reduced in CPP-treated mice compared to vehicle-treated mice. Also, the numbers of B cells and NK cells in spleen were significantly decreased in CPP-treated mice compared to vehicle-treated mice (Table 1). These results clearly showed that CPP treatment induces immunodeficiency in mice.
Next, we orally administered CJLP243, CJW55-10, or CJLP475 (1×1010 CFU per mouse) every day for two weeks starting from one day after the final injection of CPP (Fig. 3A). One day after the final administration of these probiotics, we measured immune parameters to determine whether oral uptake of these probiotics could induce an antigen-specific cell mediated immunity of immunocompromised mice (Fig. 3A). To measure immune parameters, splenocytes from each experimental mouse were rechallenged with these probiotics. Cytokine secretion levels, NK cell activity, and proliferation of immune cells were then measured. Results indicated that treatment with these probiotics enhanced the secretion of Th1-type cytokines (IFN-γ and IL-12) in immunodeficient mice than in vehicle-treated mice (Figs. 3B and C). Production levels of pro-inflammatory cytokines including IL-1β, IL-6, IL-12, and TNF-α were augmented in these probiotics-treated mice than in vehicle-treated mice (Fig. 3C).
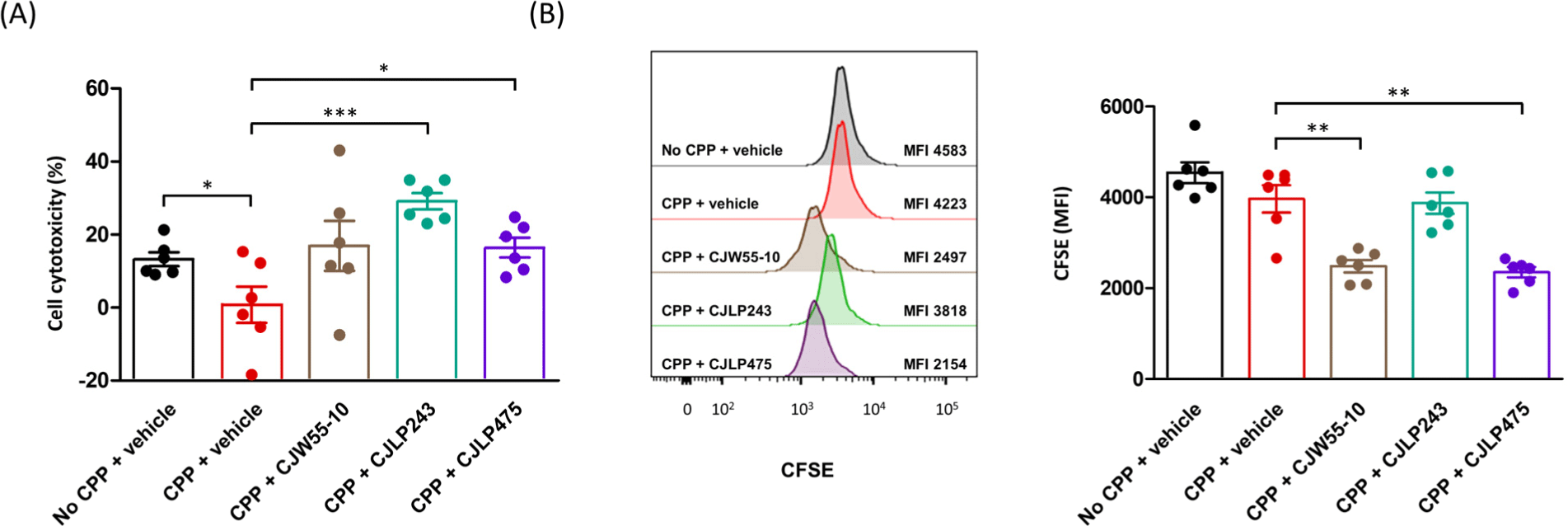
Furthermore, target cell killing activity of NK cells was increased in CJLP243 or CJLP475-treated mice than in vehicle- treated mice (Fig. 4A). Enhanced immune cell proliferation was observed in CJW55-10 or CJLP475-treated mice than in vehicle-treated mice (Fig. 4B). Taken together, these results indicate that consumption of selected probiotics can induce an antigen-specific cellular immune responses in immunocompromised individuals.
Finally, we compared absolute numbers of splenocytes in experimental mice to determine whether the induction of antigen-specific cellular immune responses by consumption of these probiotics could recover absolute numbers of splenocytes into normal steady status of immune homeostasis. Compared to that of CPP untreated mice (No CPP+vehicle), CPP treated mice (CPP+vehicle) exhibited significantly reduced absolute number of splenocytes (Table 2). Consumption of these probiotics in CPP treated mice increased substantial number of total splenocytes, but the numbers of splenocytes in these animals were not statistically significant compared to CPP treated mice (CPP+vehicle) (Table 2). This result might indicate that the induction of antigen-specific cellular immune responses is not enough to reconstitute the steady state of immune homeostasis in immunocompromised mice.
| Group1) | Total cell number (×107) | p-value2) |
|---|---|---|
| No CPP+vehicle | 8.19±0.33 | 0.022 |
| CPP+vehicle | 5.43±0.97 | |
| CPP+CJW55-10 | 6.45±0.95 | 0.468 |
| CPP+CJLP243 | 6.13±0.89 | 0.607 |
| CPP+CJLP475 | 7.56±1.23 | 0.203 |
In the present study, we evaluated an immunomodulatory effect of three different stains from L. plantarum in immunocompromised mice. After oral-administration of these probiotics in immunodeficient mice, we re-challenged with selected probiotics and monitored antigen-specific cellular immune responses. Combined results clearly indicated that the consumption of selected probiotics has an immunostimulatory role in immunocompromised individuals. Similar to our results, several previous observations demonstrated that the administration of certain probiotics in CPP-treated mice increased phagocytic activities of macrophages, the migration of neutrophils into periphery, and killing activities of both NK cells and cytotoxic T cells (Bujalance et al., 2007; Jang et al., 2013; Salva et al., 2014). Therefore, treatments of selected probiotics might have a potential beneficial effect in immunodeficient persons. However, it is not clear whether consumption of these probiotics enhances overall immunity against certain pathogens since our model is focused on monitoring antigen-specific immune responses in immunocompromised mice. Further study will be required to evaluate the beneficial effects of consumption of selected probiotics after challenging with specific pathogens in immunodeficient mice.
Many studies including our study supported evidence that probiotics could modulate cellular immune responses in mammals. Major components of probiotics which modulate the immune responses might be strain-specific carbohydrates or lipid components (Crump et al., 2020; Sukhithasri et al., 2013). Therefore, the interaction between certain pattern-recognition receptors and strain-specific molecular patterns might initiate the immune modulation. Indeed, several strain-specific molecular patterns from various bacterial cell walls have been identified as powerful immune modulators after interaction with several pattern recognition receptors including toll-like receptors, nucleotide-binding oligomerization domain-like receptors and C-type lectin receptors (Li and Wu, 2021). Identification of major molecular ingredients of CJW55-10, CJLP243 or CJLP475 which stimulate the signal transduction of specific pattern recognition molecules is useful for the development of novel immunostimulants.













