Introduction
The novel postmortem process referred to as Rinse & Chill® technology (RC: MPSC Inc., Hudson, WI, USA) entails inserting a sanitized catheter into the carotid artery of an animal immediately upon exsanguination, and a chilled isotonic solution is then infused into the vasculature at a rate up to 10% of the carcass weight. The RC solution pushes the blood out of the carcass through the venous vasculature (jugular veins) and also continues to drain from the carcass similar to normal bleeding (Kethavath et al., 2022; Mickelson and Claus, 2020). In detail, on the kill floor, each carcass is weighed by an automated process control system and the amount of rinse solution needed is calculated. This process requires typically approximately 4 min or less per beef carcass, and the catheter is then removed. The suspended carcass passes through the normal slaughter procedure along connecting rails.
The RC solution is primarily composed of water (98.5%) with a blend of dextrose, maltose, and sodium phosphates. All of the ingredients in the RC solution are approved by the U.S. Food & Drug Administration and are internationally GRAS-listed, common food-grade ingredients. The solution is designed based on the hypothesis: Dextrose (glucose) is the normal substrate in muscle used in muscle metabolism to produce energy. Maltose is simply a disaccharide composed of two glucose units which the muscle utilizes to provide additional glucose for metabolism. Phosphates stimulate energy metabolism and are naturally present in the muscle. Phosphatases also present in the muscle rapidly hydrolyze the phosphates as part of normal muscle metabolism (Kılıç et al., 2020; Sickler et al., 2013). Thus, these substrates are used to enhance the glycolytic metabolism of the muscle (Hunt et al., 2003; Yancey et al., 2001). These substates leave no detectable residues in beef (Hwang et al., 2020) since the muscle is physiologically active at the time of vascular rinsing early postmortem.
Recently, a number of studies have been investigated (Da Cunha Moreira et al., 2018; Hwang et al., 2020; Kethavath et al., 2020; Kethavath et al., 2022) to address the physical and biochemical properties related to carcass vascular rinsing effects on beef meat quality. Therefore, this review is to provide a review of the potential physical and biochemical mechanisms associated with beef carcass vascular rinsing that impact color stability, oxygen consumption, sarcomere length, metabolite residues, and muscle contractile responses.
Effects of Beef Carcass Vascular Rinsing on Meat Quality
Previously published studies have found that the carcass vascular rinsing has a beneficial effect on meat color stability (Fowler et al., 2017; Hunt et al., 2003; Kethavath et al., 2021; Kethavath et al., 2022; Mickelson and Claus, 2020). Common results were more red (higher CIE a* and deoxymyoglobin, lower metmyoglobin) and lighter (higher CIE L*) of the meat color in the triceps brachii (shoulder), longissimus lumborum (loin), and semimembranosus (ham) from a variety of animal species (beef, bison, pork, and lamb). In a study by Da Cunha Moreira et al. (2018), they observed RC lean ground beef resulted in greater redness (CIE a*: 15.75 vs. 13.06; p<0.05), higher deoxymyoglobin (DMb: 1.29 vs. 1.12; p<0.05), and lower metmyoglobin (MMb: 0.94 vs. 1.11; p<0.05) on 7 d display than the beef samples from the non-rinsed carcasses (Table 1). RC appeared lighter and more yellow on 1 d display, but no other differences were seen on day 4 and 7 between the control (CN) and RC. Associated with the increase in lightness, Farouk and Price (1994) suggested that the higher yellowness (CIE b*) determined might be responsible for the increased lightness, as a result of the greater light scattering. Erazo-Castrejón et al. (2019) found that the RC removed 40% more residual blood from the pork muscle compared to the conventional chilled carcasses. RC had lower hemoglobin (Hb: 13.6% vs. 19.1%) and higher myoglobin (Mb: 86.4% vs. 80.9%) than the CN when the percentages of Mb and Hb by weight (relative to each other) were calculated. Thus, the additional blood removal from the carcass might have contributed to the lighter colored meat.
Data from Da Cunha Moreira et al. (2018).
Myoglobin oxidation is one of the major non-microbial factors which result from lipid oxidation and induce quality deterioration in muscle foods. RC tended to slow down the myoglobin oxidation process with the greater DMb and lower MMb compared to CN during the display periods (Table 1). These results could be related to sodium phosphates in the RC solution that act as antioxidants and inhibit lipid oxidation by chelating metal ions (Fe2+, Cu2+, etc.). Wu et al. (2022) reported that the reduced Mb forms (DMb) have lower pro-oxidant ability in muscle foods when compared to their oxidized form (MMb). In addition, Hb and Mb are the most abundant heme proteins and contribute to accelerating lipid oxidation in postmortem muscles. With more blood being removed by RC, besides the additional amount of hemoglobin that is reduced in the vasculature, RC would also remove non-heme iron. Non-heme iron has the potential to cause oxidation of lipid and myoglobin. This is also closely associated with the flavor which is negatively affected by the hemoglobin and non-heme iron. Yancey et al. (2001) reported that steaks (semitendinosus) from the RC beef carcasses had a lower cardboard flavor (rancidity) compared to the non-rinsed samples. A greater beef flavor was identified in cooked ground beef from the RC cattle than the non-rinsed carcasses. Thus, the enhanced blood removal and sodium phosphates likely contributed to the meat redness, an extension in meat color stability, and the sensory quality.
Oxygen consumption represents the ability of postmortem muscle to consume oxygen. In postmortem muscle, there is competition for oxygen among mitochondria, oxygen-consuming enzymes, myoglobin, microorganisms, and lipid and protein oxidation (Ramanathan and Mancini, 2018). In particular, the competition between myoglobin and mitochondria is the key factor that influences the formation of bright cherry red color (oxymyoglobin). If mitochondrial activity outcompetes myoglobin for oxygen, this will result in a darker, deoxygenated meat color (predominant DMb) due to limited oxygen supply to myoglobin (Mancini et al., 2009). In addition, if the oxygen level in the meat is very low, this promotes MMb formation.
RC ground beef tended to have a greater oxygen consumption as the amount of residual oxygen was lower than CN, but no difference was observed in the ground loin (Kethavath et al., 2020; Table 2). However, Kethavath et al. (2021) found that RC had greater oxygen consumption (RC 4.56% vs. CN 5.18% O2; p<0.05) immediately after removing the PVC film and vacuum packaging of the ground pork shoulder. Other studies that just compared muscles (non-rinsed muscles) described that the psoas muscle had greater oxygen consumption than the longissimus muscle, suggesting that this muscle predominantly consisted of red fibers, and accordingly greater oxidative metabolism was than the longissimus (Ke et al., 2017; Mohan et al., 2010). In another study (Ramanathan and Mancini, 2018), greater oxygen consumption can contribute to enhanced color stability. This likely involves mitochondrial production of NADH and other reducing equivalents related to maintaining myoglobin in ferrous form that is associated with the formation of deoxymyoglobin and oxymyoglobin.
| Treatment | Oxygen consumption (%) | Sarcomere length (μm) |
|---|---|---|
| CN | 2.58a | 1.42a |
| RC | 2.45a | 1.80b |
Oxygen consumption on ground beef loin from Kethavath et al. (2020); sarcomere length on beef loin muscle from Kethavath et al. (2022).
RC promptly removes more oxygen from oxygen-carrying hemoglobin by the enhanced blood removal that stimulates the rate of postmortem anaerobic glycolysis. Perhaps this shift from aerobic metabolism to anaerobic metabolism facilitates preserving mitochondria activity reflected in the greater oxygen consumption in the RC muscles. The aerobic metabolism through the TCA cycle induces production of more NADH that helps maintain the heme iron in the ferrous state (Kethavath et al., 2020). With greater oxygen consumption and generation of NADH would facilitate formation of deoxymyoglobin which is a more stable oxidative state than oxymyoglobin. Therefore, RC has the potential to positively affect color development and color stability (limit oxidation) in beef that is anaerobically packaged.
When muscles enter rigor mortis, sarcomere shortening can occur, depending on chilling conditions (e.g., rate, temperature), carcass suspension methods, and glycolytic metabolism within the muscle fibers that will have effects on postmortem energy metabolism, pH development, and proteolytic activity (Warner et al., 2014). It is well established that when a carcass temperature declines below 10°C early postmortem while the pH is still above 6 and the presence of adenosine 5’ triphosphate (ATP), the muscle can shorten, caused by excessive release of calcium ions from the sarcoplasmic reticulum (Davey et al., 1976; Tornberg, 1996). In contrast, a rapid drop in pH while a carcass temperature is still warm (35°C–42°C) can lead to negative effects on color (pigment denaturation) and reduction in water holding capacity due to the less physical space for water to bind to the contractile proteins of actin and myosin (Hopkins et al., 2014; Mickelson and Claus, 2020; Warner et al., 2021).
Kethavath et al. (2022) conducted research with beef loins and determined the sarcomere length, wherein sarcomeres in the beef loin of RC were approximately 27% longer (Table 2) than in the CN. They noted that electrical stimulation was not applied to the CN or RC carcasses to accelerate rigor mortis and help avoid cold shortening. In the absence of electrical stimulation and since the muscles were excised from the carcass at 24 h postmortem, perhaps rigor was not yet completed, and the muscles were capable of continuing to shorten. In contrast to the control, Kethavath et al. (2022) found that RC accelerated the rate of anaerobic glycolysis and the pH declined before conditions known to cause cold-induced occurred. In RC the pH was below 6 when the temperature reached approximately 10°C–15°C (>12 h postmortem) whereas in the control the pH was 6.3. This finding confirms those by Devine and Gilbe (2014) who suggested that the ideal temperature for meat to prevent cold shortening is approximately 15°C at rigor mortis (>12 h postmortem without stimulation).
A potential alternative hypothesis maybe related to RC stimulating early release of calcium from the sarcoplasmic reticulum and limiting the ability of this membrane system to adequately resequester calcium (Mickelson and Claus, 2020). When carcasses are vascularly rinsed, the appendages typically extend and stiffen. This physical change may be related to the released calcium or perhaps the physical effect of the pressure of the rinse solution during the application. After the vascular rinsing terminates the appendages mostly returned to their pre-rinsed anatomical position. Once the muscle has limited ATP, the sarcoplasmic reticulum would not be able to resequester the calcium and the muscle would be locked into rigor thereby the muscle would be unaffected by temperatures below 10°C. Another aspect of RC is that the early release of calcium could encourage more desirable conditions for calpain activity and affect the contractile mechanism. Calpains are known to increase in activity when calcium is available and cause tenderization. Several studies related to the beef carcass vascular rinsing have shown improvement in tenderness, wherein tenderness was improved by 20% in cow striploin steaks (Hite et al., 2019), 24% in bison steaks (Mickelson and Claus, 2020), 56% in steaks from light dairy cows, and 58% in steaks from lean dairy cows (Kethavath et al., 2022). Thus, the chilled isotonic RC solution appears to counterbalance potential unfavorable changes related to a more rapid pH decline, as evidenced by greater sarcomere length in the beef loin. In addition, despite the more rapid pH decline in the carcass, use of the chilled RC solution and its effect on efficiently removing heat out of the carcass helps protect the meat pigments from being denatured and improves the red color stability.
Contractile system of the muscle can be stimulated depending on the release of calcium. Generally, muscle contractions are accelerated as postmortem metabolism proceeds under anaerobic conditions. As a result, lactate and hydrogen ions which are the end products of anaerobic glycolysis are accumulated in the muscle (Matarneh et al., 2017). The muscle pH normally lowers from approximately 7.2 in living muscle to an ultimate pH between 5.5 and 5.7. The rate and extent of postmortem metabolism is a critical factor affecting protein functionality and meat quality attributes such as color, texture, flavor, water holding capacity, and shelf-life (Hopkins et al., 2014; Warner et al., 2021). However, in spite of its importance, the rate and extent of muscle metabolism and its role during postmortem glycolysis have not been clearly established associated with vascular rinsing effects.
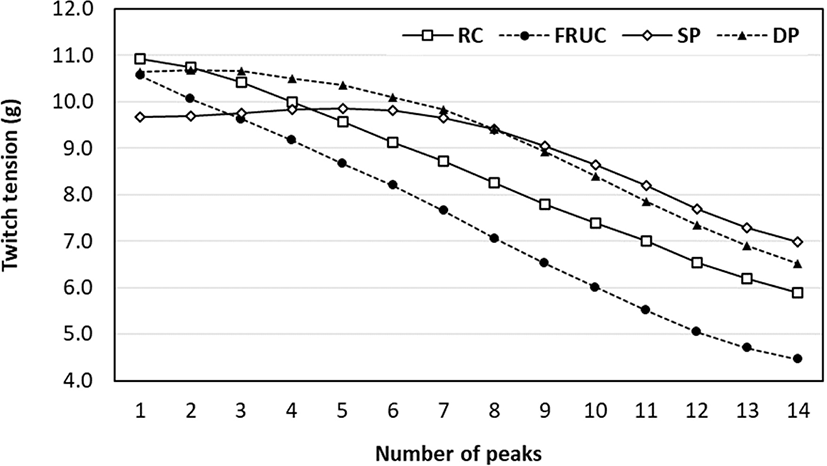
Determination of contractile force responses in pre-rigor muscles were studied to understand if metabolic activity could be modulated by various glycolytic substrates. Recently, a preliminary study was done by Da Cunha Moreira et al. (2019) who determined muscle contractile responses to electrical stimulation from the beef sternomandibularis muscles exposed to various substrate solutions (Fig. 1). It was demonstrated that the contraction force decreased as time increased. Faster declines in contraction force were observed using the standard Rinse & Chill® solution as well as fructose, whereas the forces were maintained longer and decreased slowly with the use of dipotassium phosphate and sodium phosphate. Regarding the faster decline in force, this is likely due to the increased rate of postmortem glycolysis. The accelerated release of calcium then contributes to a rapid muscle contraction and an increase in the rate of muscle metabolism and the rate of pH decline (Huff-Lonergan and Lonergan, 2005). Besides, the cold temperature of the solution likely affects the ability of the terminal cisternae of the sarcoplasmic reticulum to store calcium when carcasses are vascularly rinsed. Matarneh et al. (2017) stated that at lower temperatures, the sarcoplasmic reticulum actively pumps calcium ions out of the sarcoplasm while calcium sequestering is impaired, thereby promoting the cytosolic concentration of calcium. Thus, this result confims RC leads to early release of calcium ions from the sarcoplasmic reticulum, having a direct effect on activation of the muscle shortening (Devine et al., 2014).
A study has revealed that chilling method did not impact (p>0.05) metabolite residues (phosphorus, sodium, and glucose; Table 3) in the ground beef loin (Hwang et al., 2020). They also reported that their results were similar to numerous studies (Czerwonka and Szterk, 2015; Flowers et al., 2018; Garmyn et al., 2011; Mateescu et al., 2013), wherein phosphorus and sodium were naturally found in loin muscles from conventionally chilled beef carcasses. In detail, the results of phosphorus were: CN=1,667 μg/g; RC=1,661 μg/g. The concentrations of phosphorus naturally found in beef were 1,727 μg/g in steer, 1,945 μg/g in cow, 2,167 μg/g in bull, and 2,022 μg/g in cattle, respectively. The sodium content was 711 μg/g in CN and 655 μg/g for RC. The levels of sodium naturally present in beef were 352 μg/g in steer, 530 μg/g in cow, 510 μg/g in cattle, and 533 μg/g in bull, respectively. The glucose contents (CN 6.81 μmol/g vs. RC 7.49 μmol/g) were very similar to those reported by Rhoades et al. (2005) in the beef M. sternocephalicus pars mandibularis which contained 6.54 μmol/g. Antonelo et al. (2020) found that the glucose was 4.11 μmol/g in the beef loin. Falowo (2021) noted that there are large variations in mineral content of muscle foods which are mainly affected by pre-mortem (nutrition, species, breed, sex, age at slaughter, muscle types, etc.) and post-mortem (processing methods, methods for the determination of minerals) factors.
| Treatment | Phosphorus (μg/g) | Sodium (μg/g) | Glucose (μmol/g) |
|---|---|---|---|
| CN | 1,666.91a | 710.77a | 6.81a |
| RC | 1,661.46a | 654.58a | 7.49a |
| SE | 77.74 | 30.33 | 0.47 |
Data from Hwang et al. (2020).
Although the RC solution used to rinse out the blood from the vasculature contains some substrates. At the time of early postmortem and vascular rinsing, pre-rigor muscle is still physiologically active. These substrates are metabolized by the muscle, leaving no detectable residues compared to meat from non-rinsed carcasses. The carcasses are vascularly rinsed at no more than 10% of the carcass weight with a cold isotonic solution, and the solution is allowed to freely drain. Therefore, these findings confirmed that after postmortem storage, no differences in glucose and phosphorus residuals between the non-rinsed beef and the RC beef exist. Based on the inherent amount in sodium naturally found in beef, and the rinse solution is allowed to drain, the minor contribution associated with the phosphates does not result in a difference in the sodium content of the beef.
Conclusion
The primary focus of this review was on beef associated with vascularly rinsing carcasses. This review has provided a more comprehensive understanding of the potential biochemical mechanisms on how vascularly rinsing, Rinse & Chill®, modulates glycolytic activity and its effects on various meat quality outcomes. Results based on early published research combined with the recent studies revealed that Rinse & Chill® has the ability to stimulate the rate of glycolysis early postmortem that facilitates the decline in pH. Beef carcasses vascularly rinsed have improved color stability and tenderness, and leave no detectable rinse solution saccharides or phosphate residues in the meat. Future research that explores an understanding of the effects of this process on sarcoplasmic reticulum functionality would be beneficial as well as a deeper understanding of how the impact on mitochondrial activity. In addition, an understanding of the effects of differences in the glycolytic status among different beef animals at the time of harvest would contribute to the understanding of the process that could lead to further optimization of desired meat quality outcomes.













