Introduction
Meat quality is important to consumer satisfaction and enticing them to buy again (Papanagiotou et al., 2013). Previous research indicated that the overall consumer satisfaction of meat was first related to tenderness (O’Quinn et al., 2018). Meat color affects consumer’s purchase decision as consumers prefer bright red meat (Mancini and Hunt, 2005). The water-holding properties of meat is important in store and meat processing (Prevolnik et al., 2010). Thus, the formation mechanism of meat tenderness, water-holding capacity and color has always been the research focus in the area of meat science.
A series of physiological and biochemical changes occur after the animals are slaughtered, of which rigor mortis is the most significant change. As glycolysis is the source of ATP in muscle cells after slaughter, glycolysis determines rigor mortis. Early glycolysis after slaughter is very important to meat quality, too fast or too slow glycolysis will cause heterogeneous meat and bring losses to the meat industry (Lawrie and Ledward, 2006). As the time after slaughter prolonged, muscle undergoes the process of rigor mortis and aging, meat quality attributes continuously change during this process. Based on the development of rigor mortis, meat can be divided into hot fresh meat (pre-rigor) and aged meat (post-rigor). Recent studies have shown that the hot fresh meat and aged meat has different meat quality characteristics. The hot fresh meat has lower cooking loss in comparison with aged meat, but the tenderness was better in aged meat (Xiao et al., 2020). Hence, to explore the formation mechanism of meat quality characteristics at different postmortem phases and enrich meat quality regulatory theory is needed.
Many studies have been carried out on mechanism of meat quality development postmortem, but there are still issues of large variation in tenderness, color deterioration, and drip in the meat industry, indicating that the reason for meat quality difference at different postmortem phases has not been fully understood and the regulatory mechanisms also have not been fully elucidated (Bhat et al., 2018; Mancini and Hunt, 2005).
There have been some reports on the mechanism of protein phosphorylation affects meat quality during postmortem storage (Li et al., 2018; Wang et al., 2019). Acetylation is also a crucial modification of protein lysine ε-amino group that acetyltransferase catalyzes the transfer of acetyl group from acetyl coenzyme A (Ac-CoA) to lysine. The positively charged acetyl group changes the structure of the lysine side chain to regulate protein homeostasis, intracellular signaling and biomolecular interactions (Narita et al., 2018). Jiang et al. (2019) suggested that the dynamic protein acetylation was related to muscle postmortem changes that may affect meat color, drip loss, pH and cooking yield in pork. Studies have found that protein acetylation positively regulated the postmortem glycolysis of muscle (Li et al., 2016; Li et al., 2017b). Furthermore, a proteomic study suggested that pre-slaughter handling may regulate meat color, tenderization, water distribution, and pH via acetylation/deacetylation of glycolysis enzymes (Zhou et al., 2019). Altogether, protein acetylation may involve in the transformation from muscle to meat and affect meat quality formation at different postmortem phases.
Hence, this experiment dedicated to investigate the effects of protein acetylation on meat quality at different postmortem phases (pre-rigor, rigor mortis and post-rigor) in terms of myofibrillar and sarcoplasmic proteins that are the two types of proteins with the highest proportion in muscle tissue. The relationship between protein acetylation (sarcoplasmic proteins and myofibrillar proteins) and meat quality attributes (tenderness, color, and water-holding capacity) in ovine longissimus thoracis et lumborum (LTL) muscles at different postmortem phases were investigated to provide new insight on the variation of meat quality at different postmortem phases.
Materials and Methods
Ten carcasses of crossbred sheep (fat tailed Han×local sheep) were randomly collected from a local slaughterhouse. These sheep were 8 months old and had the same feeding system, batch, genetic background, sex and pre-mortem treatment. The butchery of sheep was carried out in a morning using standard practices of the slaughterhouse. The mean carcass weight was 27.8 kg (range 27.0–28.6 kg). The LTL muscles were collected within 30 min after slaughtering. Superficial fascia and fat were removed before wrapped the LTL muscles with oxygen-permeable membrane and stored at 4°C. At 1 h postmortem, two pieces of LTL muscles from the left side weighed approximately 65 g were cut for pH and color measurement respectively. Afterwards, the two pieces of meat were wrapped and stored at 4°C again for the pH and color measurement at 1 h, 6 h, 12 h, 1 d, 2 d, 3 d, 5 d, and 7 d postmortem. The remaining left-side and right-side LTL muscle were collected at the same postmortem time points that some used for shear force and cooking loss measurements, others were stored at −80°C for myofibril fragmentation index (MFI) and protein acetylation analysis respectively.
The pH meter (testo 205, Testo, Lenzkirch, Germany) was performed a 2-point calibration in buffers at pH value of 4.00 and 7.00 before measurement. The average pH value was calculated by taking the mean of three separate positions of each LTL muscle samples.
The determination of meat color was following the published literature (Li et al., 2017a) using CM-600D colorimeter (Konica Minolta Holdings, Tokyo, Japan). For each sample, four random points were selected for color measurement. The color parameters including lightness (L*), redness (a*) and yellowness (b*) were read directly from the instrument and averaged for statistical analysis.
The reflectivity values from 360 nm to 740 nm were obtained from the colorimeter for the calculation of reflectance values at 474 nm, 525 nm, and 572 nm (Li et al., 2017a). The Kubelka–Munk K/S values were calculated by K/S = (1 − R)2/(2 R), R = % reflectance. The relative content of myoglobin redox forms was calculated by the equation deoxymyoglobin (DeoxyMb %) = [1.5 − (K/S474) / (K/S525)], metmyoglobin (MetMb %) = [2 − (K/S572) / (K/S525)] and oxymyoglobin (OxyMb %) = [1 − (K/S610) / (K/S525)] (AMSA, 2012; Li et al., 2018).
MFI was measured according to Wang et al. (2018) but with minor changes: (1) muscles were homogenized three times for 30 s using dispersing machine (IKA Labortechnik, Staufen, Germany), (2) half MFI buffer was used for the second resuspending; and (3) the suspension of myofibrils was diluted five times before protein concentration determination.
Cooking loss and shear force were measured by the methods described by Hopkins et al. (2010) and Holman et al. (2015). A certain weight of muscle (73.5±12.1 g) was collected from the LTL for cooking loss measurement at each postmortem time point and weighed as m1. The samples were soaked in 71°C water using cooking bags. Thirty-five minutes later, the samples were moved into cold running water for 30 min. Afterwards, drying the meat pieces with filter paper and reweighed as m2, cooking loss was calculated by (m1 – m2) / m1 × 100%.
The cooked blocks were stored at 4°C–5°C overnight to measure shear force. Twelve cuboidal strips (length 4 cm, width 1 cm, height 1 cm) were cut off parallel to the fiber orientation. Afterwards, the V-slot blade (TA. XT plus® texture analyser, Stable Micro Systems, Nottingham, UK) was set to 1 mm/s crosshead speed to measure the peak force (Newtons) required to cut the cuboidal strips.
The determination of the total level of acetylated sarcoplasmic and myofibrillar proteins was done according to Li et al. (2016). Briefly, the concentration of sarcoplasmic protein and myofibrillar proteins was measured by BCA (bicinchoninic acid) assay kit, and then take a certain amount of protein solution to run electrophoresis. Finally, the relative content of the acetylated proteins in the total proteins is determined by the acetylated lysine antibody using Western blotting, which is recorded as the total level of acetylated proteins. The detailed method is as follows.
Frozen samples (1 g) were homogenized (3×15 s with 15 s break on ice between bursts) in 6 mL pre-chilled extraction buffer (0.6057 g Tris, 0.0771 g DTT, pH 8.3, one tablet protease inhibitor per 50 mL) by dispersing machine. The sarcoplasmic and myofibril proteins were separated by centrifugation at 10,000×g for 30 min (4°C). The sarcoplasmic protein concentration of supernatant was measured using the BCA method and then adjusted to 4 mg/mL. Dissolve the pellets (myofibrillar proteins) with 15 mL 5% SDS (sodium dodecyl sulfate) solution firstly, and then heated for 20 min at 80°C. Afterwards, the myofibrillar protein concentration was adjusted to 4 mg/mL. Equal volume of loading buffer was added into the diluted supernatant and pellet solution. Finally, the solution was heated in 100°C for 10 min and then refrigerated at –80°C until gel electrophoresis.
Western blotting analyses of acetylated proteins were performed according to the standard procedure. Ten microliters sample was separated on 12% Mini-PROTEIN TGX Precast Gels. Set the voltage at 70 V to pass the sample through the concentrated gel, and then maintain the voltage at 110 V to pass the sample through the separation gel. Proteins on gels were transferred to nitrocellulose membrane at 100 V for 200 min.
After blocked for 1 h at room temperature, the membrane was incubated with first antibody (PTM Bio, Hang Zhou, China) overnight at 4°C, and incubated with second antibody (CST, Danvers, MA, USA) for 1 h or so at room temperature. Acetylated proteins were visualized using enhanced chemiluminescence (ECL) kit.
One-way ANOVA was performed for comparisons of postmortem time points by SPSS Statistic 21.0 (IBM, Chicago, IL, USA). The principal component analysis (PCA) was performed by Origin 2018 software (OriginLab, Northampton, MA, USA). The significance was determined by the Duncan’s Test at the 5% confidence level. The results were expressed as average and SD.
Results
As shown in Table 1, pH declined significantly from 1 h to 12 h postmortem (p<0.05) and then remained stable until 3 d postmortem. The pH values decreased significantly again (p<0.05) after 3 d postmortem, and reached the minimum value on 5 d postmortem, which increased significantly from 5 d to 7 d postmortem (p<0.05). The pH value of 7 d still lower than that of 6 h postmortem (p<0.05).
The L*, a*, and b* values of LTL muscles stored at 4°C were shown in Table 1. The L* and b* increased from 1 h to 2 d postmortem, moreover, the L* and b* of 2 d were higher than 1 h, 6 h, and 12 h (p<0.05). The L* decreased significantly from 3 d to 7 d postmortem (p<0.05). The b* decreased significantly from 3 d to 5 d postmortem (p<0.05). Similarly, the a* increased significantly from 1 h to 12 h postmortem and reached the maximum value at 12 h postmortem (p<0.05). The a* value decreased from 2 d to 7 d postmortem, which was lower on 7 d postmortem than that on 2 d, 3 d, and 5 d postmortem (p<0.05).
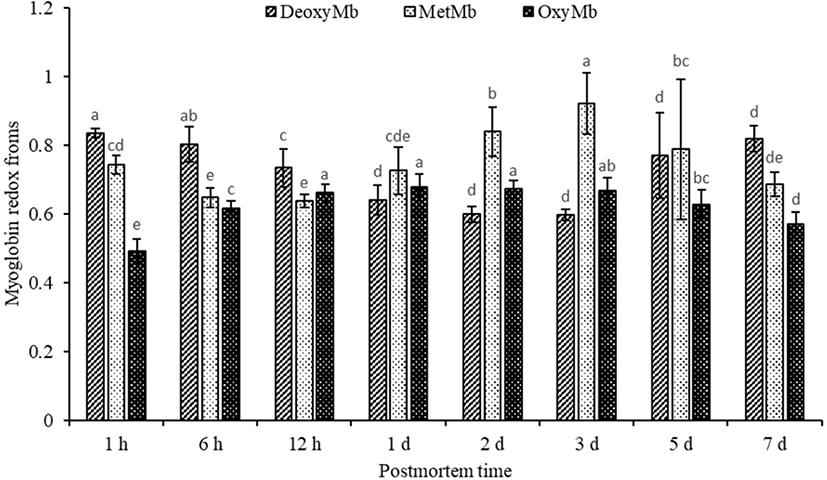
The percentage of DeoxyMb, MetMb and OxyMb were shown in Fig. 1. The DeoxyMb decreased from 6 h to 1 d and increased from 3 d to 5 d postmortem (p<0.05). The relative content of MetMb decreased significantly from 1 h to 6 h postmortem (p<0.05). The MetMb increased from 1 d to 3 d postmortem, and reached the maximum on 3 d (p<0.05). Furthermore, the MetMb content decreased significantly again from 3 d to 7 d (p<0.05). The relative content of OxyMb increased significantly from 1 h to 12 h postmortem (p<0.05). It was noted that the OxyMb of 7 d postmortem were lower than all other time points except 1 h postmortem (p<0.05).
As shown in Table 1, the MFI of LTL muscles always increased within 7 d postmortem (p<0.05). MFI at 5 d and 7 d postmortem were higher than that at all other timepoints (p<0.05).
The shear force did not change from 1 h to 12 h postmortem (Table 1, p>0.05). Furthermore, the shear force reached the maximum value on 1 d postmortem, which was higher than that at all other timepoints (p<0.05). It was noted that no significant difference in shear force was observed between 2 d and 3 d, 5 d, and 7 d postmortem (p>0.05). According to the change of shear force, the pre-rigor period was 1 h–12 h postmortem, the process of rigor mortis was 12 h–2 d postmortem, whereas the stage of the post-rigor was 2 d–7 d postmortem.
The cooking loss of 1 h, 6 h, 12 h, and 1 d was lower than 3 d postmortem (Table 1, p<0.05). No significant difference in cooking loss was observed between 3 d and 5 d, 5 d and 7 d postmortem (p>0.05), but the cooking loss of 3 d postmortem was higher than that on 7 d postmortem (p<0.05).
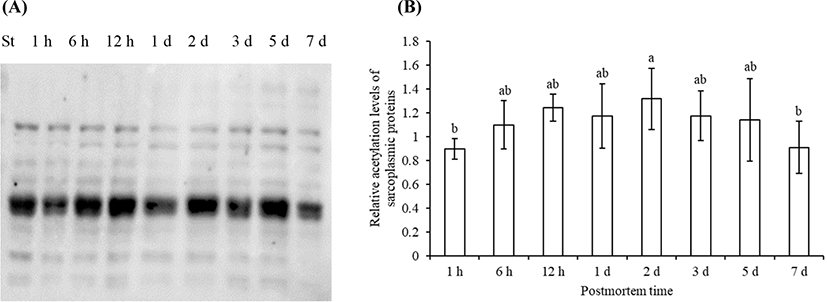
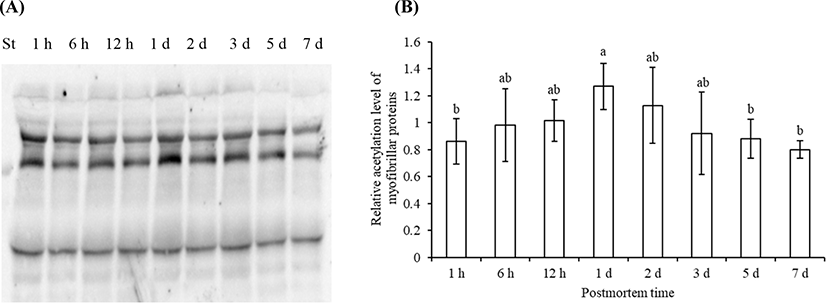
The acetylated sarcoplasmic proteins (Fig. 2A) and myofibrillar proteins (Fig. 3A) were visualized by Western blotting. The total level of acetylated sarcoplasmic proteins and myofibrillar proteins both showed a trend of first increased and then decreased (Fig. 2B, Fig. 3B). The total level of acetylated sarcoplasmic proteins was significantly higher on 2 d postmortem than that at 1 h and 7 d postmortem (p<0.05). The total level of acetylated myofibrillar proteins was significantly higher on 1 d postmortem than that at 1 h, 5 d, and 7 d postmortem (p<0.05).
The relationship between the total level of sarcoplasmic and myofibrillar protein acetylation and pH, color, cooking loss, shear force, and MFI at different storage stages were investigated by PCA. The PCA applied to the data matrix from 1 h to 12 h postmortem (Fig. 4A) showed that the cosine angles among total level of acetylated sarcoplasmic proteins and cooking loss, L*, a*, b*, OxyMb were less than 90°. The total level of acetylated sarcoplasmic proteins showed a positive correlation with L*, a*, b*, OxyMb, and cooking loss, whereas these variables were negatively correlated with pH. The total level of acetylated myofibrillar proteins were positively correlated with shear force and MFI.
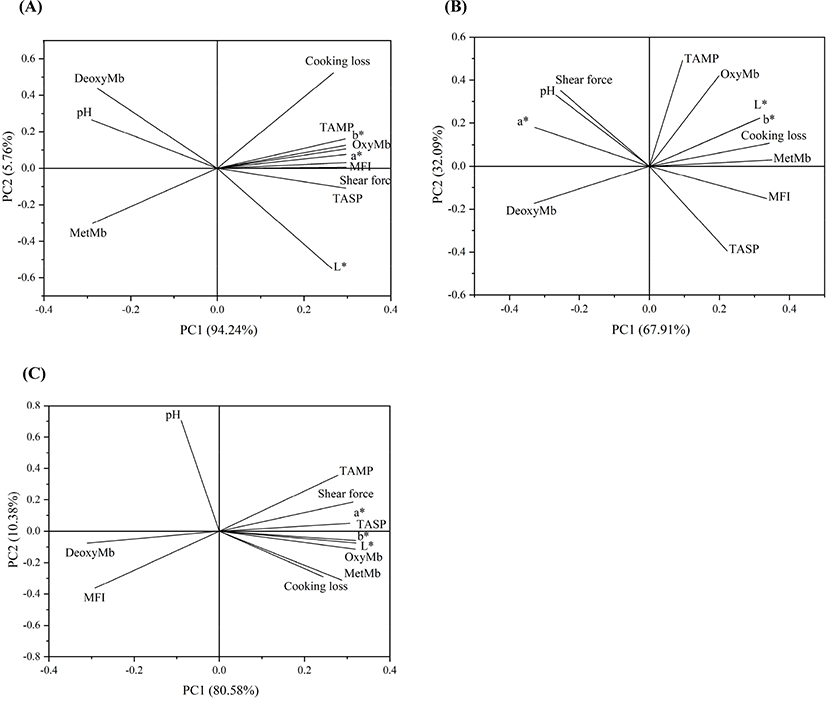
The PCA applied to the data matrix from 12 h to 2 d postmortem (Fig. 4B) suggested that the total level of acetylated sarcoplasmic proteins showed a positive correlation with cooking loss, L*, b*. Whereas these variables were negatively correlated with pH. It was noted that the a* was positively correlated with pH and negatively correlated with the total level of acetylated sarcoplasmic proteins. Furthermore, the total level of acetylated myofibrillar proteins were positively correlated with shear force.
The PCA applied to the data matrix from 2 d to 7 d postmortem (Fig. 4C) suggested that the total level of acetylated sarcoplasmic proteins was positively correlated with cooking loss, L*, a*, b*. Whereas pH was negatively correlated with these variables. In addition, the total level of acetylated myofibrillar proteins were positively correlated with shear force and the MFI was negatively correlated with shear force.
Discussion
The pH decreased gradually within 12 h postmortem indicating that the glycolysis has finished at the pre-rigor phase. The value of L* increased during the pre-rigor period and then decreased, which could be explained by the firstly increased and then decreased tendency of drip loss. Changes in water-holding capacity result in changes in meat surface moisture, which in turn affects L* (Mungure et al., 2016). The value of a* increased gradually because of the increased OxyMb content from the period of pre-rigor to rigor mortis. However, with prolonged storage time, oxidation of OxyMb to MetMb and lipid oxidation gradually increased, therefore the value of a* declined until 7 d postmortem (Bekhit et al., 2007). Similarly, the value of b* showed a firstly increased and then decreased tendency, this may be interpreted as the changed ratio of OxyMb/myoglobin postmortem (Lindahl et al., 2001). MFI is inversely related to shear force and is usually used to indicate the tenderness of meat (Culler et al., 1978; Olson et al., 1976). The MFI increased during the whole postmortem storage, which could be interpreted as the proteolytic breakdown of myofibrillar proteins (Hopkins et al., 2000). It can be inferred from the Table 1 that the meat reached maximum rigor period on 1 d as the shear force reached the maximum value. Afterwards, as the myofibrillar protein breakdown, and the muscle ultrastructure destruction, the muscle reached post-rigor period and the shear force decreased gradually, which was similar with previous results (Wheeler and Koohmaraie, 1994). With the change of pre-rigor to rigor mortis, the thick filaments combine with thin filaments to form an irreversible cross bridge, which results in the contraction of muscle spatial structure and increase in cooking loss consequently. From rigor mortis to post-rigor, the protein degradation and disruption of muscle integrity led to an increase in cooking loss (Abdullah and Qudsieh, 2009).
Protein acetylation is one of the major post-translational modifications in both prokaryotes and eukaryotes (Drazic et al., 2016). The changes in the acetylation of sarcoplasmic and myofibrillar proteins in postmortem muscles was investigated in this study. Since acetyl coenzyme A (Ac-CoA) was the main acetyl donor, it was proposed that the changes in total level of acetylated sarcoplasmic and myofibrillar proteins in postmortem muscles may be mainly affected by changes in Ac-CoA content (Kato, 1978; Poleti et al., 2018; Říčný and Tuček, 1980). Termination of blood supply in muscle tissue after slaughter may lead to an increase in the content of Ac-CoA at the pre-rigor phase, and then as the cessation of metabolism, the content of Ac-CoA decreased (Kato, 1978; Poleti et al., 2018; Říčný and Tuček, 1980). Therefore, the protein acetylation increased at the pre-rigor phase and then decreased with the exhaustion of Ac-CoA in the LTL muscles.
The pH value and total level of acetylated sarcoplasmic proteins showed a strong negative correlation at the pre-rigor and the rigor mortis phase, but weakened at the post-rigor phase. Previous research showed that protein acetylation can increase the activity and stability of glycolytic enzymes, thereby affecting the glycolysis rate postmortem (Li et al., 2017b; Xiong and Guan, 2012). For sarcoplasmic proteins, many acetylated glycolysis and glycogen metabolism enzymes include glycogen phosphorylase, pyruvate kinase and glyceraldehyde-3-phosphate dehydrogenase were identified, which showed a decreased acetylation level within 24 h postmortem (Jiang et al., 2019; Li et al., 2017b). Glycogen phosphorylase, and phosphofructokinase are glycometabolic rate-limiting enzymes in glycolysis. Thus, protein acetylation positively regulated the glycolysis process and accelerate the pH decline rate at early postmortem. And with the decrease of glycolytic enzyme acetylation level, its promotion of the glycolytic process weakens. In addition, research showed that pH was related to all other meat attributes and could reflect the overall quality of meat (Kang et al., 2019). In summary, sarcoplasmic protein acetylation negatively regulated the pH value postmortem and be associated with the overall meat quality by controlling glycolysis at early postmortem.
Meat color was governed by the interactions between myoglobin and various external and internal factors (Mancini and Hunt, 2005; Suman and Joseph, 2013). In this study, the total level of acetylated sarcoplasmic proteins showed a positive correlation with a*, b* and OxyMb, a negative correlation with DeoxyMb at the pre-rigor phase. The reason could be that myoglobin acetylation increase its oxygen binding capacity and oxygen content was higher in the early postmortem, which in turn leads to an increase in a* (Jiang et al., 2019; Lindahl et al., 2001; Suman and Joseph, 2013). The positive relationship between total level of acetylated sarcoplasmic proteins and b* value at the pre-rigor phase possibly because both of a* and b* are related to myoglobin forms (Suman and Joseph, 2013). The more myoglobin oxygenation resulted in more redness and higher ratio of OxyMb/myoglobin, which will result in more yellowness (Lindahl et al., 2001). However, although acetylation increased its oxygen binding capacity of myoglobin, myoglobin was mainly oxidized to produce MetMb due to the decrease of oxygen content with the extension of postmortem time (Suman and Joseph, 2013). Thus, a* showed a strong negative correlation with the total level of acetylated sarcoplasmic proteins and MetMb, and a weak negative correlation with OxyMb at the rigor mortis phase. The total level of acetylated sarcoplasmic proteins decreased with the exhaustion of Ac-CoA at the post-rigor phase, thus the oxygen binding capacity of myoglobin decreased. As a result, a* and b* value decreased due to the gradually decreased OxyMb content and increased lipid oxidation (Bekhit et al., 2007; Jiang et al., 2019). Thus, a* and b* value positively correlated with the total level of acetylated sarcoplasmic proteins at the post-rigor phase. In summary, sarcoplasmic protein acetylation improved meat color by increasing myoglobin oxygen binding capacity at early postmortem. At the same time, the values of a* and b* were negatively correlated with pH at the pre-rigor phase. The reason could be that the high pH values increased the meat surface oxygen consumption rate, which inhibited the formation of OxyMb cladding (Aalhus et al., 2001; Simmons et al., 2008). Therefore, sarcoplasmic protein acetylation regulated the a* and b* of LTL muscles by controlling the glycolysis and myoglobin function at early postmortem.
Cooking loss was positively correlated with the total level of acetylated sarcoplasmic proteins throughout the seven days postmortem. This may be because acetylation changes the protein charge state, and the internal hydrophobic groups such as sulfhydryl group is exposed. The exposed hydrophobic groups increase the hydrophobicity of the protein, and then leads to the easy aggregation of the protein and the formation of precipitation (Srisailam et al., 2002). Precipitation of sarcoplasmic proteins on myofibrils can reduce the electrostatic repulsion between filaments, leading to increased moisture loss (Eikelenboom and Smulders, 1986). Furthermore, previous research showed that the decrease in muscle water-holding capacity was related to muscle contraction, degradation and changes of temperature and pH postmortem (Lawrie and Ledward, 2006). pH was associated with the myofibrillar protein breakdown and actomyosin dissociation during postmortem storage (Starkey et al., 2016; Wu et al., 2014). As protein acetylation has been proved to be involved in the energy metabolism postmortem, sarcoplasmic protein acetylation affected cooking loss by changing pH in muscle. Moreover, the L* showed a positive correlation with cooking loss at rigor mortis and post-rigor. This could be explained by the kept increasing water-holding capacity of fresh meat after slaughtering, causing moisture to leak out on the surface of the meat, increasing L* (Mungure et al., 2016).
Tenderness is recognized as the most critical meat quality attribute as variation of tenderness is the most common cause of unsatisfied meat (Jeremiah, 1982). Several acetylated myofibrillar proteins involved in rigor mortis had been identified, which indicated that protein acetylation may affect the postmortem tenderization process (Foster et al., 2013; Jiang et al., 2019). In all the three different rigor periods, shear force showed a positive correlation with the total level of acetylated myofibrillar proteins, while the correlations at the pre- and post-rigor phase were higher than that at the rigor mortis phase. Abe et al. (2000) reported that actin acetylation facilitated its weak interaction with myosin. Viswanathan et al. (2015) reported that the acetylation of actin could alter electrostatic associations between tropomyosin and myosin, attenuate tropomyosin’s inhibition of binding of actin and myosin, and thereby enhances actomyosin associations. Furthermore, the acetylation of myosin could decrease the Michaelis constant (Km, the concentration of substrate at which the reaction takes place at one half its maximum rate) of the actin-activated ATPase activity and increased the interaction with actin (Samant et al., 2015). At pre-rigor phase, in addition to the increasing actomyosin content due to the reduction of ATP, the acetylation of myofibrillar protein also contributed to the inhibition of actomyosin dissociation. While at post-rigor phase, as the total level of acetylated myofibrillar proteins decreases, its inhibitory effect on actomyosin dissociation is weakened. Thus, the total level of acetylated myofibrillar proteins showed a positive correlation with shear force at the pre- and post-rigor phase. The correlations between shear force and the total level of acetylated myofibrillar proteins at the rigor mortis phase was lower than that at the pre- and post-rigor phase. Probably because the binding of myosin and actin has reached the maximum at rigor mortis phase, the rigidity of muscle was in a slowly changing state. Shear force was positively correlated with MFI at the pre-rigor phase, negatively correlated with MFI at the rigor mortis and post-rigor phase. This could be because the increase in MFI has a smaller effect on muscle tenderization than the increase in shear force caused by the combination of myosin and actin to form an irreversible cross bridge at the pre-rigor phase (Culler et al., 1978). In summary, myofibrillar protein acetylation negatively regulated tenderness by inhibiting actomyosin dissociation, especially in the early and late postmortem.
Conclusion
The regulatory effect of protein acetylation on meat quality is mainly reflected in the early postmortem (1 h–12 h). In the early postmortem period, acetylation of sarcoplasmic protein negatively regulates pH and water-holding capacity, and positively regulates meat redness; acetylation of myofibrillar protein negatively regulates tenderness.













