Introduction
Food allergy is type I hypersensitivity reaction mediated IgE antibodies and its prevalence is increasing every year (Sampson, 2004). Food allergies are related to the failure of mucosal immune system to identify the food antigen. Allergic reactions can be alleviated by a shifting from a Th2 to a Th1 immune response and T cell anergy or tolerance induction (Chehade and Mayer, 2005; Oliveira et al., 2015).
Oral administration of antigen can induce systemic antigen-specific unresponsiveness called to oral tolerance. Oral tolerance can be induced by T cell anergy (Weiner et al., 1999) or clonal deletion (Chen et al., 1955a) in mice after administration of a single high dose of antigen or regulatory T cell activation by repeated lower doses (Chen et al., 1955b).
Recently several studies reported the induction of tolerance with oral administration of antigen alone (Ngan and Kind, 1978; Strid et al., 2004). Also, with continuously processed immunotherapy studies for food allergy, oral tolerance induction with peptide and probiotics have been reported, as well. In peptide-oral tolerance induction, oral administration of genetically modified rice seed, that express many T cell epitopes, reduced the Th2 immune response (Takagi et al., 2005). Repeated administration of Lactococcus latis that express β-lactoglobulin inhibited the Th2 immune response concomitant with the induction of a Th1 immune response (Adel-Patient et al., 2005).
Meanwhile, gamma irradiation can alter the structure of an allergen composed of glycoproteins. Gamma-irradiated allergens (e.g., ovalbumin, tropomyosin, β-lactoglobulin) showed a decreased binding ability to allergic human IgE serum (Byun et al., 2000; Lee et al., 2001). The Th2 immune response, as well as, the Th1 immune response was suppressed when immunized irradiated allergen (ovalbumin) was administered to mice (Seo et al., 2007).
This study was conducted to investigate the feasibility of gamma-irradiated allergens to induce tolerance for immunotherapy. We evaluated humoral and cellular immune responses of mice after oral administration of single high dose of gamma-irradiated allergen. Here we showed that administration of gamma-irradiated allergen also induced oral tolerance.
Materials and Methods
OVA (ovalbumin, grade V, Sigma, USA) was dissolved in a 0.01 M phosphate buffered saline (PBS) to 50 mg/mL. OVA solution was irradiated at 1 kGy in a cobalt-60 irradiator (IR-79, Nordion International Ltd., Canada) equipped with 100 KCi activity and operated at a dose rate of 10 kGy/h. Gamma-irradiated OVA solutions were stored at 4℃.
BALB/c mice, 6 wk of age, were purchased from Orient Inc. (Raon Bio, Korea). Induction of tolerance followed the protocol of Kim et al. (2013) with minor modification. Briefly, either native or irradiated OVA (25 mg in PBS) was administrated to mice, intragastrically, in 0.5 ml using a ball-tipping feeding needle. On days 5 and 12 after feeding, mice were sensitized with the mixture of 20 μg of OVA and 2.6 mg of aluminum hydroxide gel (Sigma, USA) to induce a Th2 response.
Native OVA (10 mg/mL) was coated with 0.1 M bicarbonate coating buffer (pH 9.2) overnight at 4. The wells were washed with PBS containing 0.1% Tween 20 (PBST) three times. After a blocking with 2% BSA in PBS for 1 h, mouse serum, obtained from subsequent experiments, was added to the wells for 2 h. Anti-mouse IgM+IgG+IgA (IgG+A+M)-HRP, anti-mouse IgG1 conjugated biotin (BD Bioscience, USA), anti-mouse IgG2-biotin (BD Bioscience, USA), or anti-mouse IgE-biotin (BD Bioscience, USA) was added followed by a washing step with PBS. A streptoavidin-HRP conjugate was incubated for 30 min to detect antibodies labeled with biotin. One 3,3,5,5-tetramethylbenzidine solution (TMB soluble, Calbiochem, USA) was added as a chromogen. The stop reaction was conducted with 0.5 M H2SO4 without a washing step. The absorbance of the plate was read at 450 nm using a microplate reader (Bio-Rad Laboratories, USA).
The spleens were harvested from mice stimulated twice with OVA+aluminum hydroxide gel on day 19, and homogenized in a homogenizer. Single cell suspensions of splenocytes were plated in 96-well tissue culture plates at a final concentration of 1×106 cells/well in a RPMI-1640 medium containing 10% fetal bovine serum, 100 U/mL penicillin and 100 U/mL streptomycin and then cultured at 37℃ in 5% CO2. The cells were stimulated with OVA at a final concentration of 0-25 μg/mL. Culture supernatants were harvested after 72 h and stored at −70℃ until cytokine measurement.
Splenocyte proliferation was detected by the MTT assay. Following spleoncyte incubation with OVA for 72 h, 30 μL of 5 mg/mL MTT in PBS was added to each well and the plate was further incubated at 37℃ for 2 h. The plate was then centrifuged and the medium was removed. Onehundred μL of dimethylsulfoxide (DMSO, Sigma, USA) was then added. After incubation at 37℃ for 5 min, the absorbance was measured at 540 nm by microplate reader.
Supernatants cultured for 72 h were evaluated by the BD OptEIATM kit for mouse IL-2, IL-4, IL-6, and IFN-γ following the manufacturer’s instructions (BD Biosciences, USA). Detection limit of the cytokines of the assays was 1.6 pg/mL for IL-2, 15.6 pg/mL for IL-4 and IL-6, and 31.2 pg/mL for IFN-γ.
Results and Discussion
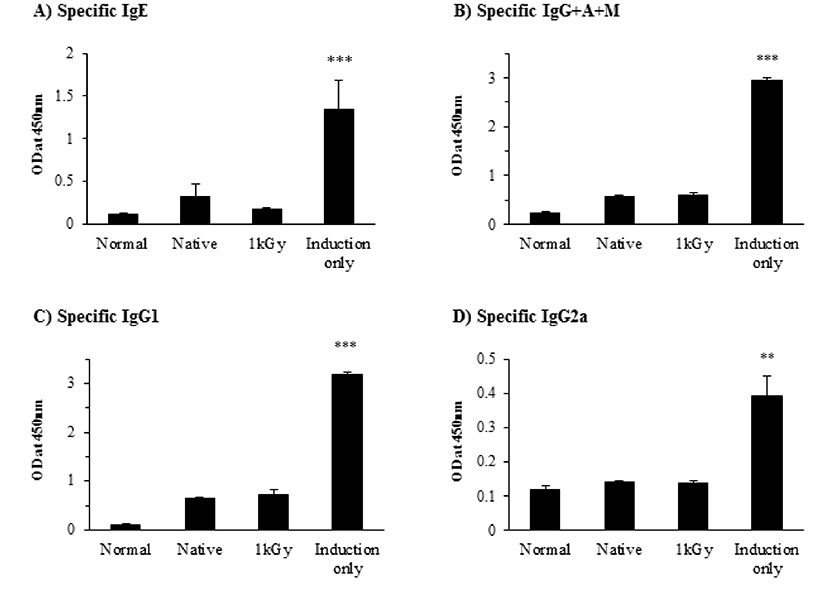
We administered a single high-dose of intact OVA, gamma-irradiated OVA, or PBS to mice, and induced allergic response two times with i.p. intact OVA and alum. An OVA-specific antibody response was evaluated to investigate whether oral administration of gamma-irradiated OVA induces oral tolerance as shown in Fig. 1. Administration of OVA irradiated at 1 kGy showed a similar effect obtained from inducing a result with intact OVA. OVA-specific IgE producion was significantly decreased in mice fed irradiated OVA compared with those fed intact OVA (p<0.05). Although OVA-specific IgG+A+M, IgG1 and IgG2a levels were decreased, there was no statistical significance. Low OVA-specific antibody-production indicates anergy or clonal deletion. OVA irradiated at 1 kGy showed a similar response to intact OVA concomitant with a lower IgE response.
Murine splenocytes of mice with allergic response only showed a high proliferative activity. Murine splenocytes harvested from mice fed intact or irradiated OVA exhibited very low proliferation despite stimulation with intact OVA for 72 h. In addition, there was a significant difference between groups fed intact OVA and those fed irradiated OVA (Fig. 2, p=0.054).
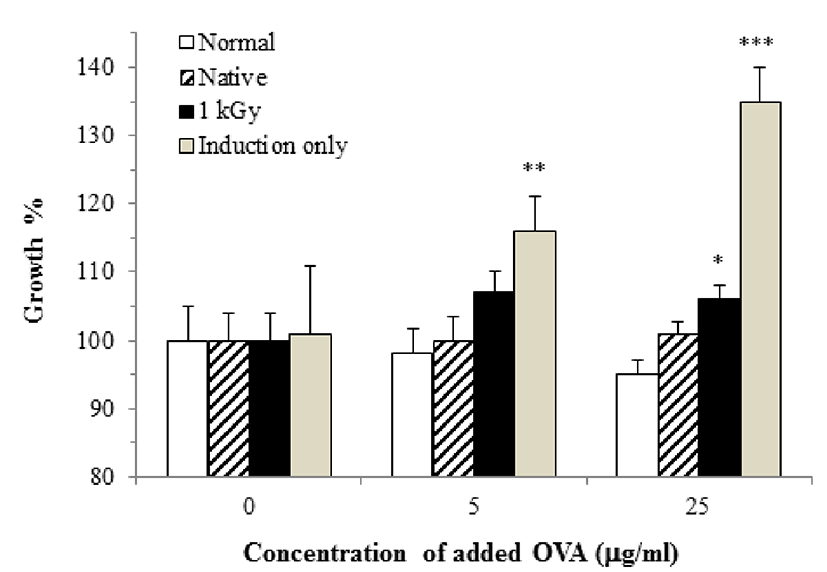
T cell anergy inhibits the development and proliferation of effector T cells due to the absence of co-stimulatory activity. In other cases, T cell proliferation can be inhibited by clonal deletion though apoptosis (Abbas and Lichtman, 2003). Pape et al. (1997) demonstrate that the T cell number increased in mice exposed to a tolerant antigen compared to naïve mice. Asai et al. (2002) showed that long-term administration of a high-dose antigen results in T cell unresponsiveness. Therefore, our results suggest that both intact and irradiated OVA induce weak T cell proliferation due to anergy or deletion.
Cytokine production of splenocytes by Th2 induction after administration of irradiated OVA was evaluated (Fig. 3). Cytokine regulation plays a central role in peripheral tolerance development against orally administrated antigen (Kim et al., 2013). Induction of tolerance for orally consumed antigen occurs by transient T cell activation and clonal proliferative responses (Gûtgemann et al., 1998). As described in the results section regarding T cell proliferative activity, IL-2 stimulates T cell proliferation had increased IL-2 production in mice fed irradiated OVA compared to mice fed intact OVA. Increased IL-2 production supports a small amount of splenic T cell proliferation in mice fed irradiated OVA compared to those fed intact OVA. Therefore, oral tolerance to antigen in mice fed intact or irradiated OVA may not be a result of clonal deletion, but anergy (Kearney et al., 1994).
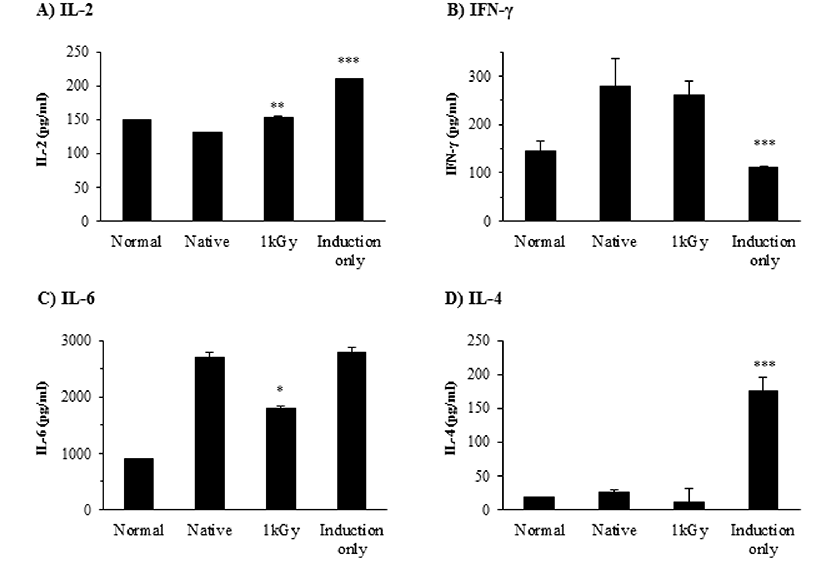
IFN-γ decreased in the group fed irradiated OVA, but not significantly. Oral tolerance by high dose OVA administration did not show a Th1 response from IgG2a and IFN-γ results. IL-6 and IL-4 have a synergistic effect in stimulating B cells to produce IgE (Borish and Steinke, 2003). The level of IL-6 was decreased in the group fed irradiated OVA compared with intact OVA-fed mice. IL-4 showed similar results to intact OVA-fed, irradiated OVA-fed, and non-fed groups (P=0.423).
Conclusion
Taken together, tolerance responses to gamma-irradiated OVA showed similar responses concomitant with a lower Th2 immune response compared with intact OVA treatment. Gamma-irradiated OVA as tolerance-inducing antigen can generate oral tolerance. Therefore, radiation technology shows feasibility as a biomedicine in allergic disease.













