Introduction
Eggs are animal protein sources that contain essential nutrients in the form of selenium, B vitamins, provitamin A, amino acids, folic acid, and fatty acids (Titcomb et al., 2019). Egg production is influenced by climate, feed, and the health of the laying hens. The use of carotenoids in animal feed has been investigated in many research studies, specifically yellow carotenoids (apocarotenoic esters), which help to increase the orange color of egg yolks (Faruk et al., 2018). Carotenoids are a category of essential pigments (red, yellow, and orange) that are widely found on the earth. Based on their composition, carotenoids are classified into two main subclasses: xanthophylls, which include lutein, β-cryptoxanthin, and zeaxanthin; and carotenes, for example, α-carotene, lycopene, and β-carotene. Carotenoids are found in different plants, microalgae, bacteria, and fungi (Nabi et al., 2020). Plants synthesize carotenoids de novo; they also develop naturally in food products and require no metabolic modification (Marounek and Pebriansyah, 2018).
Carotenoids have various biological functions, including increasing production, antioxidant, anti-inflammatory, antibacterial, and immunomodulator (Arain et al., 2018; Milani et al., 2017; Nabi et al., 2020). Carotenoids can increase poultry performance, including egg production and weight, and decrease feed conversion ratio (FCR). These processes occur through molecular mechanisms (Milani et al., 2017). Carotenoids also effectively improve the color of egg yolks. Egg yolk is a source of provitamin A, which is the forerunner of retinol. Retinol and its metabolites, for example, retinoic acid, play a role in eye function, immunity, brain function, tissue repair, and protein digestion (Brossaud et al., 2017). Because of these benefits, carotenoids are used extensively in the diets of laying hens to boost the internal quality of eggs and ensure they are suitable for use as a functional food. Carotenoids help improve the orange color in yolks, an essential consideration in shoppers’ food decisions. For example, consumers in northern European nations prefer lighter-hued yolks, while those in southern Europe prefer more strongly shaded egg yolks (Marounek and Pebriansyah, 2018).
Lipid globules aid the absorption of carotenoids in the digestive tract. Carotene particles dissolve in triacylglycerol while the xanthophylls remain near the surface of the monolayer (Faruk et al., 2018). This cycle is restricted since bile acids in the duodenum will decrease their secretion in micelles formation. At this point, while the carotenoids can still move to the lipid phase, micelles inhibit their movement in the duodenal lumen. The distribution of carotenoid from feed to egg synthesis is affected by several variables, including accessibility, openness, fringe tissue interest, a carotenoid compound, carotenoid liking, and the capacity of various lipid vesicles to cross layers in the liver and eggs. According to Ekmay et al. (2015), Lokaewmanee et al. (2013), and Sun et al. (2020), the carotenoid content of feed emphatically affects the production performance of laying hens.
Meta-analysis is a factual technique or statistical method used to gather data from past studies (Sauvant et al., 2008; St-Pierre, 2001). Jackson and Bowden (2016) stated that meta-analysis is an analytical technique used to compare and combine various data from different sources to draw an appropriate and informative conclusion based on the research questions. The review studies included in the meta-analysis should be transparent, reproducible, and up-to-date (Gurevitch et al., 2018). A meta-analysis has several advantages: 1) it provides an evidence-based summary centered on the identification of a large volume of primary evidence and its examination piece by piece, 2) it overcomes the bias associated with experiments conducted using small sample sizes, where there is insufficient statistical power to overcome variation, 3) it improves generalizability by combining the results of several studies with varying populations compared to only a primer study with a small sample size and originating from a single population, and 4) it can be updated when new primary research is published and helps to identify issues that require further research. In terms of the use of carotenoids in increasing egg yolk color, they have been shown to enhance the carotenoid content of egg yolks. Therefore, valid evidence is needed to prove the effectiveness of carotenoids in their application as an additive in laying hen feed through a meta-analysis approach.
The problem of oxidative stress that often occurs in laying hens has caused various losses for the environment, breeders, and the physiological conditions of livestock. The phenomenon of oxidative stress refers to an imbalance between the production and accumulation of reactive oxygen species (ROS) in cells and tissues and the ability of biological systems to detoxify these reactive products. ROS can function physiologically (as in cell signaling) and as a byproduct of oxygen metabolism. Therefore, alternative materials are needed to solve the above problems. One of them uses natural antioxidants from plants as feed supplements. The potential of carotenoids in feed has been extensively studied by many researchers (Nabi et al., 2020). However, there was no comprehensive conclusion supporting the use of carotenoids to boost laying hen productivity, egg quality, and immunity. As a result, this study is expected to provide more systematic information about carotenoid supplementation in feed and to be used as a database for the development of research sustainability in the feed sector.
Materials and Methods
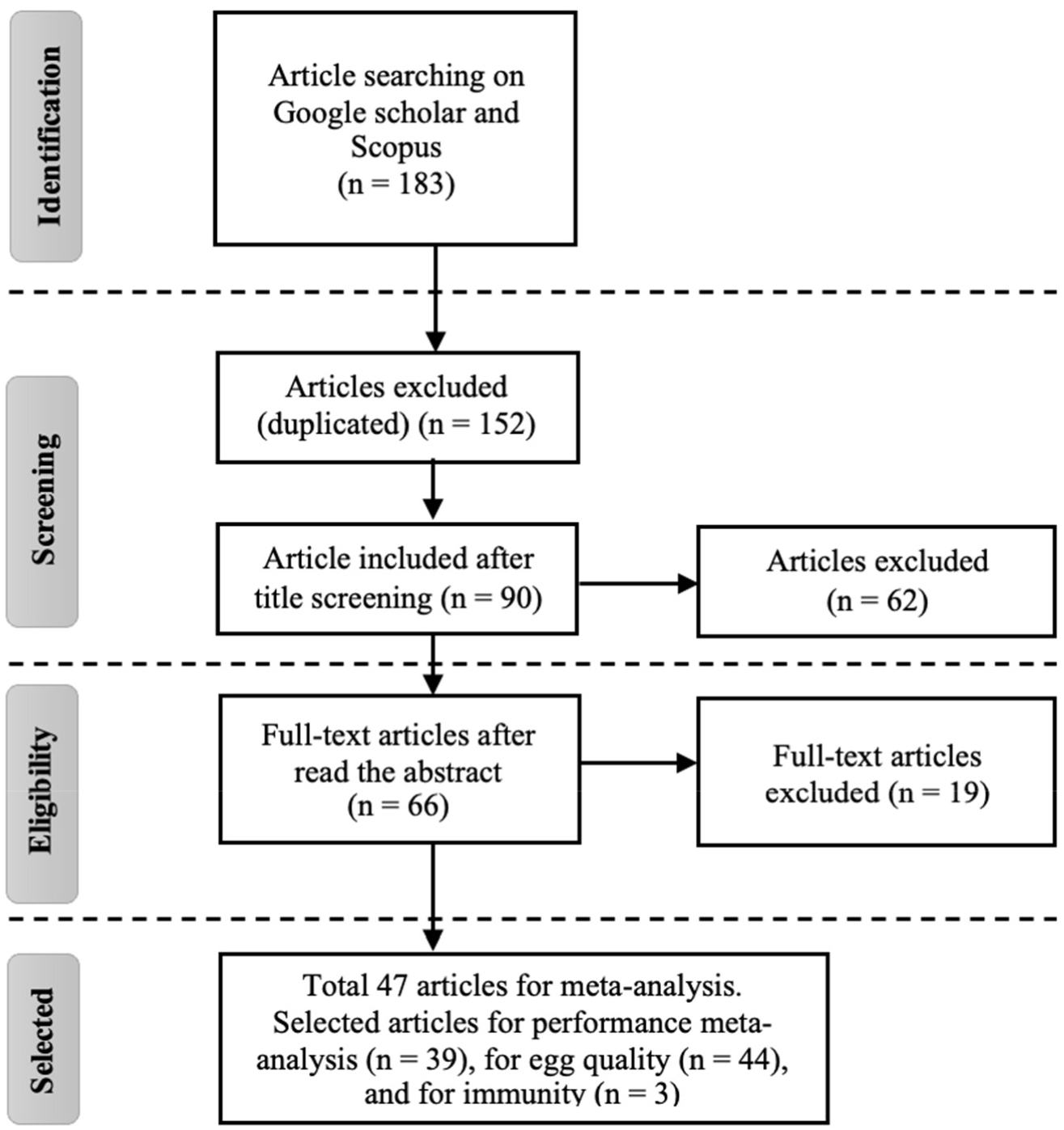
The meta-analysis was conducted in several stages, including the identification, selection, and checking of the suitability of the literature as shown in Fig. 1. The articles were obtained from Google Scholar and Scopus from 2012 to 2022. The following keywords were used in the search: ‘laying hens’, ‘maintenance’, ‘antioxidants’, ‘natural carotenoids’, ‘eggs’, ‘quality’, ‘productivity’, and ‘immunity’. An additional search was conducted using the names of the authors in the literature reference. The initial stage of identification comprised a title review. The following criteria were applied in the selection stage: (1) published in English as a full-text article; (2) contains the parameters of performance, egg quality, and immunity; (3) includes a comparison between control and experimental treatments; (4) contains the addition of natural carotenoids in feed; (5) egg products intended for consumption. The articles were selected using Mendeley reference management software to peruse the journal abstracts and eliminate duplicate journals. The appropriateness evaluation stage was completed by viewing the articles and selecting them based on their reasonableness for the point under study.
A total of 183 articles were rated as relevant to the study. This was reduced to 152 articles after duplicates had been eliminated. A total of 62 articles was excluded because the title of the study was not on the effect of natural carotenoid on performance, egg quality, and immunity of laying hens. After reading the abstract, 66 articles were indicated according to the research objectives. Finally, after considering the similarity of aims, methods, and forms of articles, 47 articles were used for meta-analysis. A total of 39 of these were selected for the performance meta-analysis, 44 full-text articles for the egg quality parameter, and 3 articles for the immunity parameter (presented in Table 1).
| Articles | Study location | Treatment | Compound | Carotenoid level (g/kg) | Result |
|---|---|---|---|---|---|
| Lokaewmanee et al. (2013) | Japan | Red pepper | Capsanthin and capsaicin | 0–5.0 | ↑ yolk color ↑ villi height |
| Nobakht (2015) | Iran | Watermelon skin | β-Carotene | 0–2.0 | ↑ performance |
| An et al. (2019) | South Korea | Tomato paste | Lycopene | 0–17.0 | ↑ egg production ↑ yolk lycopene |
| Grela et al. (2020) | Poland | Alfalfa | β-Carotene | 0–30.0 | ↑ yolk color and PUFA |
| Moeini et al. (2013) | Iran | Red pepper, marigold | Zeaxanthin and lutein | 0–3.0 | ↑ yolk color ↓ cholesterol |
| Oliveira et al. (2017) | Brazil | Red pepper, marigold | Lutein and zeaxanthin | 0–7.0 | ↑ yolk color |
| Moreno et al. (2020) | Spain | New maize gen (Ph3, BKT) | β-Carotene | 0–62.06 | ↑ yolk color |
| Tufarelli et al. (2021) | Italy | Dried tomato pomace | Lycopene | 0–15.0 | No significant effect |
| Krawczyk et al. (2015) | Poland | Lupinus luteus | β-Carotene | 0–300.0 | ↑ yolk color and final body weight |
| Kotrbáček et al. (2013) | Czech Republic | Chlorella algae | Lutein and zeaxanthin | 0–20.0 | ↑ yolk color ↑ yolk carotenoid |
| Bidura et al. (2020) | Indonesia | Moringa oleifera leaves | β-Carotene | 0–6.0 | ↑ performance, egg quality ↓ yolk cholesterol |
| Kulshreshtha et al. (2017) | Canada | Chondrus crispus and Sarcodiotheca sp. | Carotene | 0–40.0 | ↑ immunity ↓ radical effect |
| Zhu et al. (2021) | China | Phaffia rhodozyma | Astaxanthin | 0–1.6 | ↑ yolk color, immunity ↓ MDA blood |
| Ao et al. (2015) | USA | Schizochytrium limacinum | Carotene | 0–30.0 | ↑ egg yolk color and DHA |
| Ekmay et al. (2015) | USA | Desmodesmus spp., Staurosira spp. | Carotene | 0–117.0 | ↑ digestion in ileum ↑ protein metabolism |
| Magnuson et al. (2018) | USA | Haematococcus pluvialis(algae) | Astaxanthin | 0–0.08 | ↑ yolk carotenoid ↓ free radicals |
| Rutkowski et al. (2017) | Poland | L. luteus L. | Xanthophyll | 0–250.0 | ↓ performance ↑ yolk color |
| Al-Nedawi et al. (2014) | Iraq | Fruit extracts | Lycopene and carotene | 0–0.20 | ↑ egg production ↑ egg quality |
| Omri et al. (2017) | Tunis | Linseed, tomato, paprika, fenugreek | Carotene | 0–45.0 | ↑ egg weight and feed intake |
| Altuntaş and Aydin (2014) | Turkey | Marigold (Tagetes erecta L.) | Lutein and zeaxanthin | 0–20.0 | ↑ SFA ↓ MUFA |
| Sriagtula et al. (2019) | Indonesia | Indigofera leaves | Xanthophyll | 0–6.00 | No significant effect |
| Sun et al. (2020) | China | Fermented Rhodotorula mucilaginosa | β-Carotene | 0–12.5 | ↑ performance ↑ yolk color |
| Duru et al. (2017) | Turkey | Daucus carota leaves | Capsanthin and capsaicin | 0–8.00 | ↑ internal and external egg quality |
| Titcomb et al. (2019) | USA | Marigold, carrot leaves | Lutein and zeaxanthin | 0–40.0 | ↑ yolk carotenoid ↑ yolk color |
| Shevchenko et al. (2021) | Ukraine | Haematococcus algae, LycoRed | Lycopene, astaxanthin | 0–0.09 | ↑ yolk color ↑ egg quality |
| Englmaierová et al. (2013) | Czech Republic | Lutein, chlorella | Lutein and zeaxanthin | 0–12.5 | ↑ yolk color ↑ shell strength |
| Laudadio et al. (2014) | Italy | Alfalfa | β-Carotene | 0–150.0 | ↑ yolk carotenoid ↑ yolk color |
| Skřivan et al. (2015) | Czech Republic | Marigold flowers | Lutein and zeaxanthin | 0–0.35 | ↑ yolk color ↑ egg quality |
| Zheng et al. (2019) | China | Alfalfa | β-Carotene | 0–100.0 | ↓ yolk cholesterol ↑ yolk color |
| Salajegheh et al. (2012) | Iran | Tomato | Lycopene | 0–190.0 | ↑ egg yolk color |
| Panaite et al. (2021) | Romania | Kapia pepper, sea buckthorn, carrot | β-Carotene | 0–20.0 | ↑ egg yolk color |
| Honda et al. (2020) | Japan | Paracoccus carotinifaciens Panaferd-AX dan P | Astaxanthin | 0–0.008 | ↑ egg yolk color and egg quality |
| Spasevski et al. (2018) | Serbia | Dried carrot, paprika | Carotene | 0–15.0 | ↑ performance ↑ yolk carotenoid |
| Grčević et al. (2019) | Croatia | Marigold flowers | Lutein | 0–2.00 | ↑ egg yolk color |
| Shahsavari (2015) | Iran | Paprika, carrot, alfalfa, tomato | Oxycarotenoid (xanthophyll) | 0–50.0 | No significant effect |
| Siti et al. (2019) | Indonesia | M. oleifera leaves | β-Carotene | 0–60.0 | ↑ shell thickness ↑ egg yolk color |
| Skřivan et al. (2016) | Czech Republic | Marigold flower | Lutein and zeaxanthin | 0–0.95 | ↑ egg yolk color |
| Panaite et al. (2019) | Romania | Dried tomato waste | Lycopene | 0–75.0 | ↑ yolk carotenoid and color |
| King and Griffin (2015) | USA | Tomato pomace | Lycopene | 0–10.0 | No-significant effect |
| Bidura et al. (2021) | Indonesia | Fermented carrot leaves | Carotene | 0–60.0 | ↑ yolk color ↓ yolk cholesterol |
| Spasevski et al. (2017) | Serbia | Marigold flowers, paprika | Lutein and zeaxanthin | 0–15.0 | ↑ egg yolk color |
| Saleh et al. (2021) | Egypt | Paprika | Lutein and zeaxanthin | 0–4.00 | ↑ yolk color, fatty acids ↑ egg production |
| Akdemir et al. (2012) | Turkey | Tomato | Lycopene | 0–10.0 | ↑ egg quality ↓ FCR |
| Habanabashaka et al. (2014) | Rwanda | Tomato (peel and seed) waste meal | Lycopene | 0–9.00 | ↑ yolk color and carotenoid ↓ yolk cholesterol |
| Walker et al. (2012) | USA | Algae astaxanthin | Astaxanthin | 0–2.94 | ↑ egg weight |
| Omri et al. (2019) | Tunis | Arthrospira platensis | β-Carotene | 0–2.50 | ↑ egg yolk color, HU |
| Tamiru et al. (2021) | Belgium | Papaya pomace | β-Carotene | 0–7.50 | ↑ egg production, yolk color ↓ feed cost |
Several moderator variables were used to baseline information. Inclusion criteria included moderator variables [study location, breed, treatment duration, age of hens, number of hens used in the study, carotenoid (source, level, and form) added to feed], as well as mean value, SD, and SE. The parameters observed were egg quality [egg weight, shell thickness, egg yolk color, Haugh unit (HU), egg yolk cholesterol (EYC), egg yolk carotenoids], performance (feed consumption, FCR, final body weight, egg production), and immunity [immunoglobulin A (IgA) and white blood cells (WBC)].
Databases were tabulated using Microsoft Excel 2021, and converted into Comma-Separated Value files. The file was then analyzed using OpenMEE. OpenMEE is a popular open-source and user-friendly meta-analysis software (Wallace et al., 2016). The continuous variable results were reported as the standardized mean difference (SMD) with a 95% confidence interval (CI) between the control and carotenoid treatments. The effect size was calculated from the difference between the control and treatment groups divided by the SD of the population. In measuring the effect size, Hedges’d was used to determine the distance parameter between the control and the effect of dietary carotenoids. This method was chosen because of its ability to calculate effect sizes regardless of sample size heterogeneity, the unit of measurement, and statistical test results, and based on its suitability for estimating the paired treatment effect. Random effects models (REM) were appropriate for meta-analyses because heterogeneity occurs at different levels in each pooled analysis. Meanwhile, JASP 0.16.2 software was used to test for heterogeneity (I2) and validate the data against publication bias. I2 is a statistic that represents the percentage of variance in the meta-analysis that is due to study heterogeneity (Higgins et al., 2003). If the p-value was >0.05, it was considered statistically significant in this study. The SMD result was significantly affected when the diamonds in the forest plot did not touch the control line (Koricheva et al., 2013).
Results and Discussion
Forty-seven publications were selected for meta-analysis of the effectiveness of carotenoids on performance, egg quality, and immunity (Fig. 1). Detailed articles breakdown is shown in Table 1. Meanwhile, the forest plot result can be seen in Fig. 2.
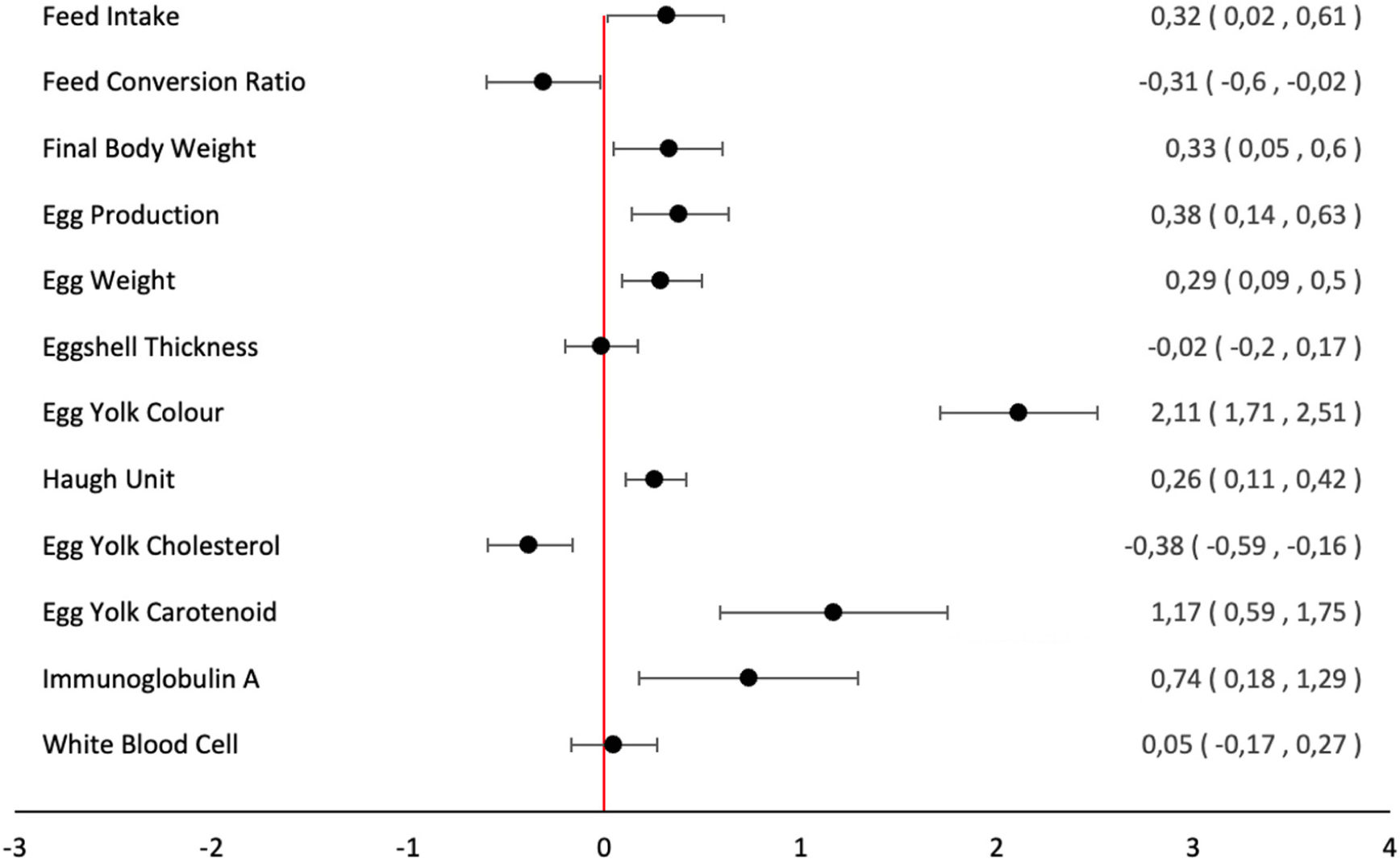
A total of 39 journals were used in the meta-analysis of the effectiveness of carotenoids on feed intake, and 93 databases were interrogated. Based on the REM calculations, the overall SMD value was 0.32 with 95% CI (0.02, 0.61; Table 2). The CI did not equal 0 (zero); therefore, the treatment in the experimental group was not quite the same as that for the control group. p SMD (<0.05; Table 2) indicated that there was a critical connection between the use of carotenoids and the consumption of laying hens. However, before concluding, it was necessary to test for heterogeneity. The results showed a p-value of <0.05 (Table 2), so the presumption of heterogeneity is accepted. This was confirmed by the high level of I2 at 99.249% (Table 2).
The validation test for publication bias using Kendall’s test (p>0.05) indicated the formation of a symmetrical funnel plot from the REM, meaning there was no publication bias. As such, the conclusions derived are valid, as indicated by the high inconsistency and absence of bias. In conclusion, carotenoids help to increase feed consumption, which aligns with the findings of Bidura et al. (2020) and Faruk et al. (2018). The provitamin A content of carotene is converted into retinol and its metabolites, such as retinoic acid, for eye function, immunity, brain function, tissue repair, and metabolism (Brossaud et al., 2017). Visually, birds given yellow feed are more palatable; as such, the addition of carotenoid pigments is vital for hens. Carotenoids work as antioxidants to counteract the effect of free radicals. Stressed chickens were reported to have significant free radical damage. This stress will disrupt physiological function and affect the behavior of chickens. One of them was eating behavior, specifically the decrease in consumption during heat stress.
A total of 27 journals were used from the literature and 78 databases were consulted. Based on calculations utilizing the REM, the overall SMD value was –0.31 (95% CI –0.60, –0.02; Table 2). The CI was not equivalent to 0 (zero), meaning that the experimental group differed from the control. The negative summary effect results indicated that the treatment could reduce the FCR value of laying hens. The heterogeneity analysis indicated that variability occurred in all studies (p<0.05); therefore, the assumption of homogeneity is rejected. This was also shown by the elevated I2 score of 99.066% (Table 2).
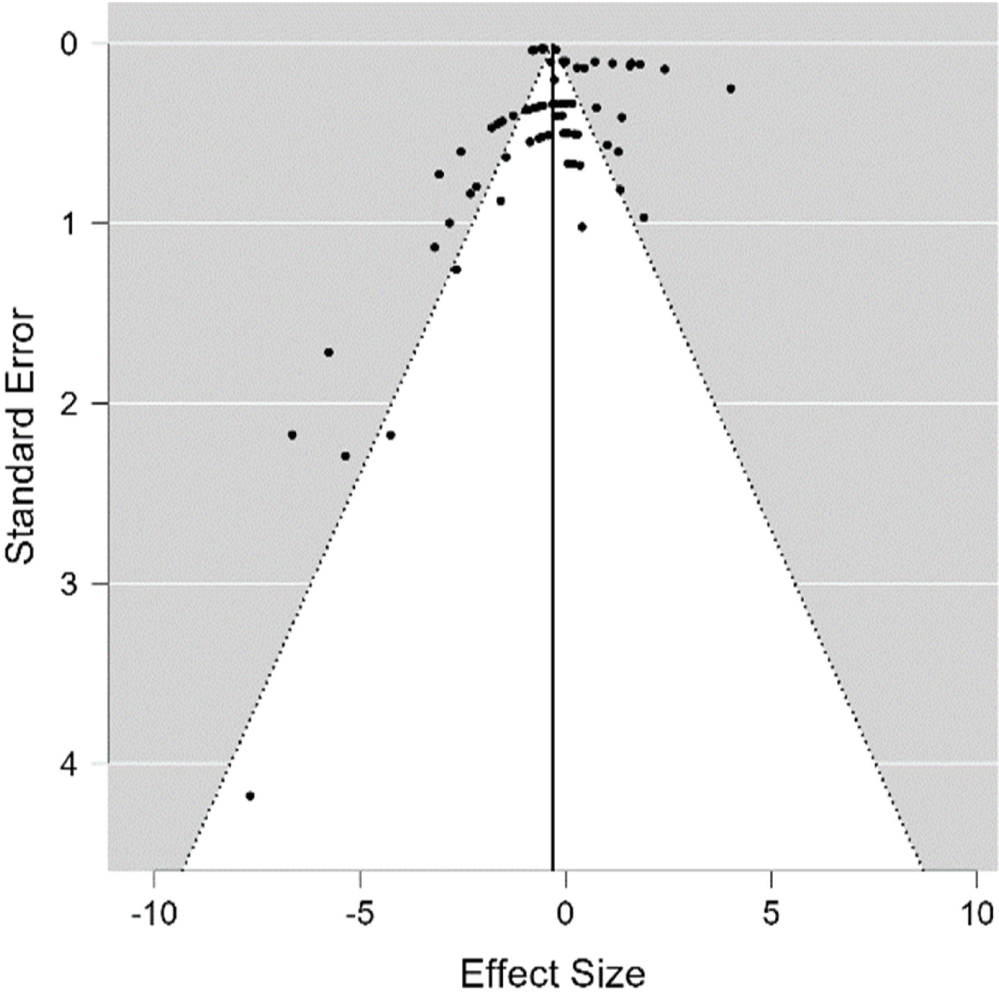
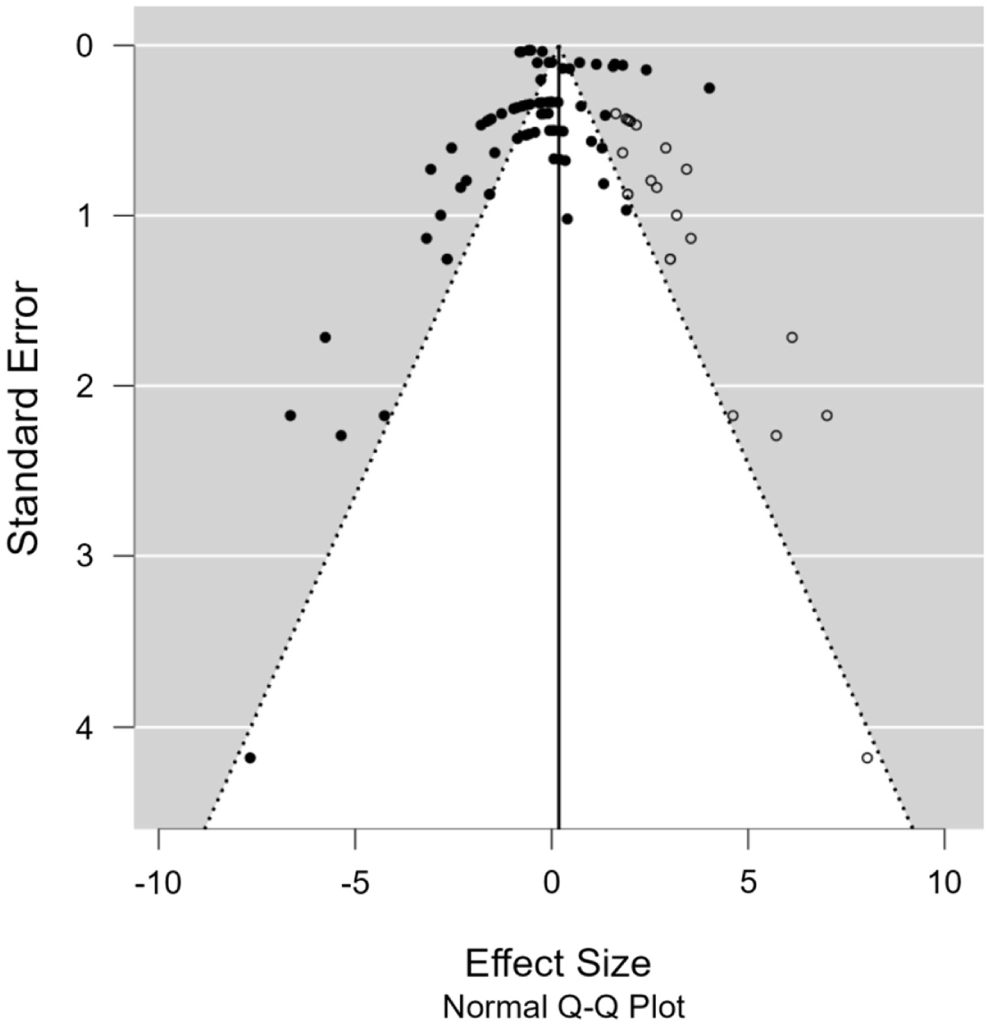
Kendall’s test was then used for validation against publication bias. The results showed that the funnel plot from the REM was asymmetric in shape; in other words, there was publication bias. The trim and fill method can help reduce publication bias. The funnel plot (Fig. 3) formed on the FCR parameter is asymmetric. The funnel plot in Fig. 4 is the result of the trim and fill analysis. Rosenthal believes that if the shift is not too far, the meta-analysis study's conclusions are acceptable and valid. The friction in the funnel plot on the FCR parameter is not very significant. So, the result can be accepted. The results indicated that carotenoids were insignificant in reducing the FCR of laying hens. This result aligns with those from an experiment by Lokaewmanee et al. (2013), who found that the use of red pepper was unable to improve the production performance of laying hens, including feed consumption and feed efficiency. On the other hand, red peppers could increase the size of the intestinal villi. The condition of the villi would have an impact on nutrient absorption. According to Zheng et al. (2019), Beijing-you chicken supplemented with 5%–10% alfalfa flour significantly reduced FCR and mortality. Alfalfa bioactive compounds improved bone strength and immune function, also lowering mortality rates (Kwiatkowska et al., 2017).
The literature reviewed for the meta-analysis of the effectiveness of carotenoids on final body weight comprised 19 journals from a total of 56 databases. Based on the REM calculations, the SMD value was 0.33 (95% CI 0.05, 0.60; Table 2). The interspace (CI) did not include 0 (zero), thus implying that the experimental group contradicted the control but that the effect was very small. The results (Table 2) indicated the existence of a significant effect between the use of carotenoids in feed and the final weight of the laying hens (<0.05). However, a heterogeneity test was required before conclusions could be drawn. The heterogeneity analysis indicated variability in all studies (<0.05; Table 2), meaning the premise of heterogeneity should be accepted. This was additionally verified by the peak percentage of I2 at 97.662%.
Furthermore, the validation test against publication bias with Kendall’s test showed that the p-value using rank correlation was p>0.05. Therefore, the funnel plot formed from the REM was symmetrical, and there was no publication bias. In conclusion, the use of the REM method in a meta-analysis of final body weight parameters was valid. The results showed that carotenoids could increase the final weight of laying chickens. This finding aligns with the investigations by Lokaewmanee et al. (2013) and Sun et al. (2020), which reported that the use of carotenoids in feed can improve the production efficiency of laying hens. The chickens used for this meta-analysis were in the egg-laying phase. Laying hens would grow quickly during the starter to pre-layer period.
The egg production parameters were calculated based on the value of hen day production (HDP). HDP is the sum of eggs produced divided by the total number of chickens and then multiplied by 100%. The literature used in the HDP meta-analysis comprised 36 journals and 101 databases. Based on the calculation utilizing the REM, the SMD value was 0.38 (CI 0.14, 0.63; Table 2). The gap (CI) did not include 0 (zero), which meant the experimental group deviated from the control but only to a very small extent. This was also demonstrated through the positive summary effect.
The heterogeneity analysis revealed the presence of variability in all studies (<0.05; evident in Table 2). Thus, the presumption of homogeneity should be declined, and the premise of heterogeneity is perceived, namely, that the variability did not occur due to a sampling failure. This was likewise confirmed by the high levels of I2(98.868%; Table 2). Furthermore, Kendall’s test was used as a validation test against publication bias. The outcomes showed that a symmetrical funnel plot was formed, or there was no publication bias. The results showed that carotenoids can increase egg production. This aligns with Al-Nedawi et al. (2014) and An et al. (2019), who found that the addition of carotenoids to feed enhances the performance of laying hens.
Chicken eggs are a nutritional food that has been widely consumed by global consumers (Lesnierowski and Stangierski, 2018). Several factors could influence egg production, including breed, age, health conditions, maintenance systems, feed, and ambient temperature. Carotenoids worked as antioxidants, reducing the free radical effects. This will affect chicken metabolism and immunity. Carotenoids are anti-inflammatory compounds that play a role in cell protection mechanisms. The addition of microalgae to feed increases the number of trophic agents in the intestinal mucosa for stimulating the mitotic process in the villi.
A total of 115 databases were used for the meta-analysis of egg weight parameters. Based on calculations using the REM, the overall SMD value was 0.29 with 95% CI (0.09, 0.50; Table 3). Since the CI did not contain 0 (zero), it can be stated that the effect of the treatment given to the experimental group was not quite the same as for the control group in terms of the egg weight parameters. The aftereffects of the analysis show that the true effect size was not equivalent to 0, thus indicating a significant correlation between carotenoid supplementation in feed and egg weight (p<0.05). However, before legitimizing this result from the REM, heterogeneity tests were required to measure the accuracy of the outcomes.
The heterogeneity analysis of the role of carotenoids on egg weight showed that variability occurred in all studies (p<0.05; Table 3). Therefore, the premise of homogeneity was dismissed while that of heterogeneity was recognized, i.e., the variability that occurred was not caused by a testing error. This was likewise confirmed by the high level of I2(inconsistency), which stood at 98.27% (Table 3). Thus, it can be concluded that dietary carotenoids effectively increase egg weight. A validation test (Kendall’s test) of the effectiveness of carotenoids in increasing egg weight was required to ascertain whether there was any publication bias. The outcomes of the analysis showed that the p-value (rank correlation test for funnel plot asymmetry) was asymmetrical, thus providing the presence of publication bias (p<0.05). The positive rating correlation (0.129) in Table 3 indicates that reviews with very large sample sizes were excluded from the meta-analysis study sample. Therefore, the result based on the REM was not valid, as indicated by the result for publication bias in Kendall’s test (p<0.05).
One of the primary criteria for egg grading was egg weight. Egg weight typically ranges between 50–70 g, depending on the age and genotype of hens (Travel et al., 2011). Carotenoids with a higher fat content in the feed will be absorbed in the small intestine. Absorption outputs are transported to the liver and tissues via portomikron and lipoproteins. Hight carotenoid and lipoprotein bonds increase yolk weight, resulting in egg weight (Fernandes et al., 2020).
The meta-analysis of natural carotenoid benefits on shell thickness was conducted using 95 databases. Based on the REM, the overall SMD value was –0.02 with 95% CI (–0.201, 0.78; Table 3). The CI contains a value of 0 (zero), meaning there was no distinction between the experimental and control groups. The results of the analysis show that the true value of the effect size was 0; therefore, there was no significant correlation between carotenoid supplementation in feed and shell thickness (p>0.05). However, before reaching any conclusions based on REM, heterogeneity tests were required to measure the accuracy of the results.
The heterogeneity analysis of the role of carotenoids on eggshell thickness showed that variability occurred in all studies (p<0.05; as shown in Table 3). Hence, heterogeneity, as opposed to homogeneity, should be perceived, i.e., the variability was not caused by a sampling inaccuracy. This was also demonstrated by the high I2(inconsistency) value of 97.964%. From this, it can be assumed that dietary carotenoids were beneficial in increasing eggshell thickness, although the effect was small.
A validation test (Kendall’s test) of the effectiveness of carotenoids in increasing shell thickness was required to determine the presence of publication bias. The outcomes of the analysis revealed that the resulting funnel plot was well-proportioned. As such, no evidence was found of publication bias (p>0.05). The positive rating correlation (0.035) indicated that studies with large samples were excluded from the meta-analysis of the study sample. It is thus possible to conclude that the result obtained from the REM was valid, as denoted by the high inconsistency and low publication bias from Kendall’s test (p>0.05).
Environmental conditions and the availability of calcium minerals in the feed are factors that influence eggshell thickness. Good-quality eggshells will protect the internals of the eggs and extend their shelf life (Kalvandi et al., 2019). When the chicken is exposed to high ambient temperatures, the quality of the eggshells suffers. High environmental temperatures cause an increase in respiratory activity (panting) in hens, thereby causing a decrease in CO compounds in the body. During the shell formation process, Ca binds with CO to form CaCO3. Alkalosis or CO deficiency causes an increase in Ca levels in the blood. Alkalosis causes a low pH in the blood, making it acidic. Carotenoids, in this case, act as antioxidants that can reduce the risk of heat stress, reduce panting, and increase eggshell quality, although the analysis reported no effect.
A total of 92 databases were interrogated for the meta-analysis of the effect of natural carotenoid feed on egg yolk colour intensity parameters. An overall SMD value of 2.110 with 95% CI (1.707, 2.51; Table 3) was obtained from calculations using the REM. Here, CI did not include 0 (zero); therefore, the treatment in the experimental group was divergent from that of the control group. The analysis results show that the true effect size was not 0, meaning there was a very large correlation between carotenoid supplementation in feed and egg yolk colour (p<0.05). However, heterogeneity tests were needed to measure the accuracy of the results based on this REM.
The heterogeneity analysis of the effect of carotenoids on egg yolk colour intensity indicated that variability occurred in all studies (p<0.05; Table 3). Therefore, the presumption of heterogeneity should be acknowledged, implying that the inconsistency was not generated by a sampling error. This was also proven by the high I2(inconsistency) value of 98.93% (Table 3). It can therefore be concluded that dietary carotenoids effectively increase egg yolk colour intensity. A validation test (Kendall’s test) of the effectiveness of carotenoids in increasing egg yolk colour intensity was then required to test for the presence of publication bias. The validation test showed that the funnel plot was balanced, meaning there was no publication bias (p>0.05). The positive correlation (0.037) indicated that studies with large samples were excluded from the meta-analysis sample. Therefore, the results of the calculations with REM were valid, as shown by the high inconsistency and no publication bias through Kendall’s test (p>0.05).
The measurement of yolk colour intensity in this meta-analysis used the Roche Yolk Color Fan method. The colour of the yolk is a necessary factor in consumer egg selection. Colour is influenced by pigment content (usually xanthophyll), type, and compounds (Altuntaş and Aydin, 2014). Several materials used in this meta-analysis provided the highest effect size for the egg yolk colour parameter, including marigold flowers (Tagetes erecta L.), algae (Haematococcus sp.), and lupine seeds (Lupinus luteus L.). β-carotene is a provitamin A compound used to improve the colour of egg yolks (Bidura et al., 2020). Through its absorption in the small intestine, β-carotene can improve the colour of egg yolks.
A total of 102 databases were used in the assessment of the HU parameter. The calculations using REM produced an overall SMD value of 0.26 with 95% CI (0.105, 0.418; Table 3). Since the CI did not touch the value of 0 (zero), it can be stated that the treatment of the experimental group was discrete from the control. The result of the true effect size was not 0, thus demonstrating an impressive relationship between carotenoid supplementation in feed and HU (p<0.05). Nonetheless, heterogeneity tests were required to measure the accuracy of the results.
The heterogeneity analysis of the role of carotenoids in the HU showed the occurrence of variability in all studies (p<0.05; as shown in Table 3). Therefore, the supposition of homogeneity is dismissed, and the presumption of heterogeneity is acknowledged, i.e., the variability was not caused by a sample distribution mistake. This was likewise shown by the high level of I2(96.82%). Thus, it can be concluded that dietary carotenoids effectively increased HU. A validation test (Kendall’s test) of the effectiveness of carotenoids in increasing HU was needed to check for the presence of publication bias. The analysis showed that the funnel was unbalanced, which implies publication bias (p<0.05). The positive correlation (0.149) in Table 3 shows that studies with enormous samples were excluded from the review. Accordingly, REM was not suitable for use in this meta-analysis, as indicated by the publication bias revealed through Kendall’s test (p<0.05).
Many factors contributed to publication bias. Those factors were writers who tend to enter only positive data, editors or reviewers who tend to select articles with positive results, and meta-analytical users who are subjective in their article selection. In addition, it can also be caused by measurement results that are indeed positive. The average HU value used in this meta-analysis was measured less than 24 hours after egg collection, so the eggs’ quality was still good. The HU value was affected by the height of the egg white (Narushin et al., 2021). A higher HU value indicated that the egg quality was getting better (Martinez et al., 2021). The HU scores in this study ranged from 60 to 99. According to the USDA (2020), HU numbers >72 are classified as “AA” quality eggs, 60–70 are “A” quality eggs, 31–60 are “B” quality eggs, and <31 are categorized as “C” quality eggs. Antioxidants in poultry feed could delay the natural oxidation process of internal egg components, but had no direct effect on albumen and HU (Fernandes et al., 2020). Measurement time could affect the value of HU. If the measurement is taken shortly after egg retrieval, the HU results will also increase.
Research information on the EYC parameters was obtained from 27 databases. Based on the REM calculations, the SMD value was –0.376 with 95% CI (–0.591, –0.160; Table 3). Given that the CI did not meet the value of 0 (zero), it can be stated that the experimental group was dissimilar to the control. The analysis results show that the true effect size was not equivalent to 0. This showed a critical negative connection between carotenoid supplementation in feed and EYC (p<0.05). Nevertheless, heterogeneity tests were still required to quantify the precision of the results of this REM.
The heterogeneity analysis of the role of carotenoids on EYC revealed that variability occurred in all studies (p<0.05; Table 3). Therefore, the premise of heterogeneity is acknowledged, i.e., the variability was not caused by a sampling omission. This was also denoted by the high level of I2, which stood at 97.85%. Hence, it can be reasoned that carotenoid content has a powerful effect in reducing EYC. A validation test (Kendall’s test) of the effectiveness of carotenoids in increasing EYC was required to test for the presence of publication bias. The investigation revealed that the funnel plot was balanced, so there was no publication bias (p>0.05). The forest plot is shown in Fig. 2 and the funnel plot is in Fig. 5. The negative rating correlation (–0.217) indicates that studies with large samples were included in the study sample. In summary, the REM outcomes were valid, as indicated by the high inconsistency and low publication bias (p>0.05).
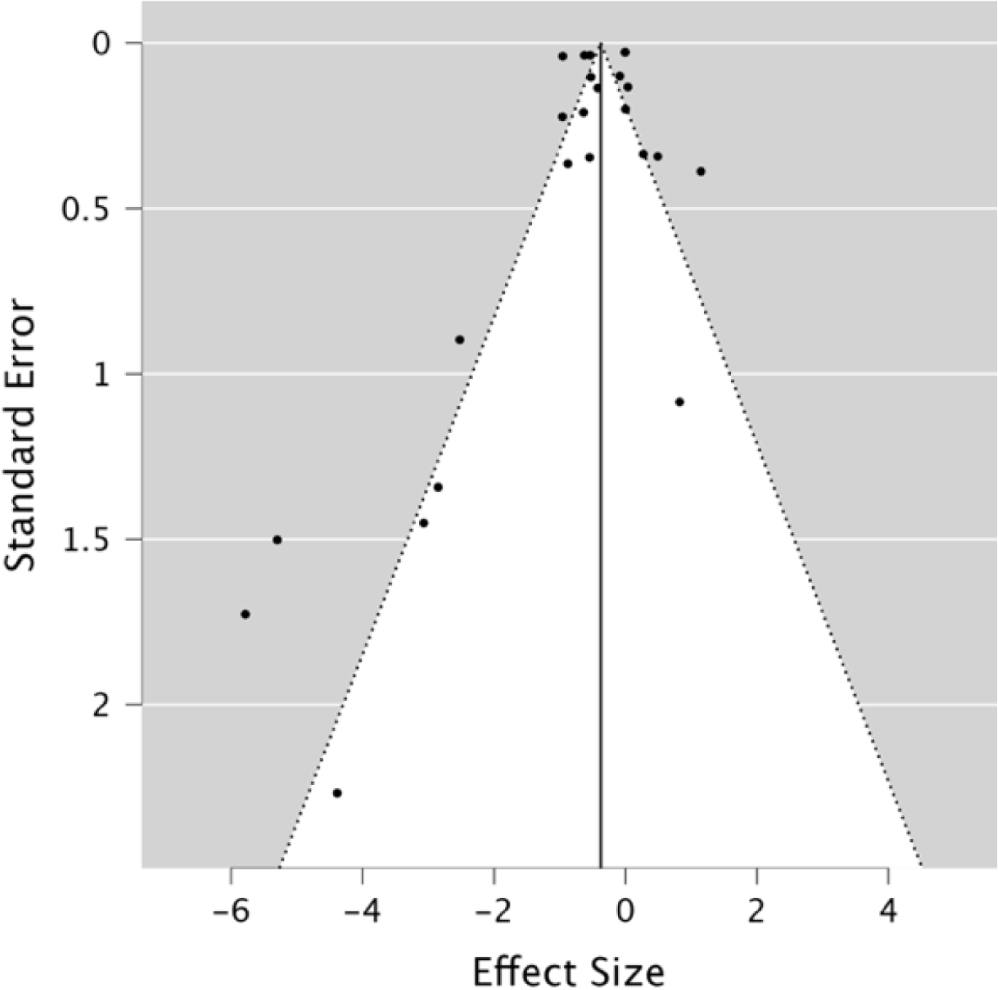
Based on subgroup analysis using OpenMEE software, the relationship between carotenoid additives and EYC levels was negatively correlated. That is, the addition of carotenoids can significantly lower EYC. Alfalfa meal, Moringa leaves, and Haematococcus pluvialis algae are more effective at lowering cholesterol than other carotenoid sources. Meanwhile, when considering the level of carotenoids, the best result for lowering EYC was obtained following the addition of 4%–6% Moringa leaves (Bidura et al., 2020).
Moringa oleifera is a plant from the Moringaceae family and is widely used in traditional medicine. Moringa plants are found widely across the continents of Asia and Africa. People use the leaves as they are thought to be rich in cancer-preventing agents, nutrients, carotenoids, alkaloids, tannins, and saponins (Leone et al., 2015). M. oleifera leaves have also been used to improve productivity and meat quality in broiler chickens (Sharmin et al., 2021a). The addition of 1%–1.5% Moringa leaves can essentially reduce serum cholesterol and EYC levels in native laying hens aged 26–42 weeks (p<0.05; Sharmin et al., 2021b). In addition to carotenoids, M. oleifera contains saponins that can bind cholesterol. The decrease in fatty substances and cholesterol content occurs mainly due to an increase in lipogenic catalyst movement and the greater discharge of bile acids in feces (Patil et al., 2010). The addition of H. pluvialis0.01%–0.08% has also been found to have a good effect on lowering EYC. Shao et al. (2019) stated that H. pluvialis(nonsaline single-celled microalgae) is one of the best sources of astaxanthin.
Many carotenoids are derived from plant parts containing crude fiber. This fiber could bind bile acids, preventing fat absorption and increasing fat excretion through feces. Carotenoids are transported from the intestinal mucosa into the blood via lymphatic vessels, then to the liver, and finally to the surrounding tissues via very low-density lipoprotein (VLDL). The high cholesterol content of egg yolks (around 200 mg) is a concern for consumers, particularly the elderly and people with hypercholesterolemia (Xia et al., 2018; Zheng et al., 2019). Excessive consumption of high-cholesterol foods may result in atherosclerosis and a fatty liver.
According to Patil et al. (2010) and Syahruddin et al. (2013), lowering lipogenic enzyme activity and increasing bile acids contributed to the decrease in cholesterol and triglyceride levels. Carotene's ability to lower cholesterol is related to the enzyme Hydroxy Methyl Glutaryl CoA (HMG-CoA). This enzyme is essential in the biosynthesis of cholesterol. β-carotene can lower blood cholesterol levels by inhibiting the function of the HMG-CoA enzyme in the formation of mevalonate in the biosynthesis of cholesterol (Bidura et al., 2021). The mevalonic pathway, which starts with acetyl CoA, is where cholesterol and beta-carotene are synthesized together. If β-carotene consumption exceeds saturated fatty acid consumption, the HMG-CoA enzyme biosynthesis process will be directed to β-carotene synthesis. Mevalonic is needed to inhibit cholesterol-forming enzymes.
A total of 26 databases were used in the analysis of egg yolk carotenoid parameters. The overall SMD value in light of the computations using REM was 1.17 with 95% CI (0.591, 1.75; Table 3). The CI was not equal to 0 (zero), which implies that the treatment in the experimental group was not the same as for the control. The true effect size was not equivalent to 0. This shows a huge correlation between dietary carotenoid and egg yolk carotenoid (p<0.05). However, heterogeneity tests were required to gauge the precision of the results from this REM.
The heterogeneity analysis of the role of carotenoids on egg yolk carotenoids indicated variability across all studies (p<0.05; as shown in Table 3). Accordingly, the premise of heterogeneity is perceived, i.e., the variability was not caused by a sampling error. Heterogeneity was likewise confirmed by the high level of I2, at 99.40%. Kendall’s test was expected to indicate the effectiveness of carotenoids in improving egg yolk carotenoids regardless of any publication bias. The result indicated an asymmetrical form for the funnel plot. As such, the result obtained from the REM was invalid due to the presence of publication bias (p<0.05). The positive rating relationship (0.43) in Table 3 shows that studies with very large samples were excluded from the meta-analysis sample.
Giving as much as 10 g of tomato flour per kg of feed can affect the concentration of carotenoids (lycopene and β-carotene) in blood serum, thus influencing egg quality (Akdemir et al., 2012). The two dominant carotenoid groups contribute to increasing egg yolk color, namely lycopene and astaxanthin (Shevchenko et al., 2021). Unlike β-carotene, the two substances are directly accumulated in the egg yolk after absorption. Meanwhile, carotene would be transformed into vitamin A, so it would not affect the color of the yolk. β-Carotene had a positive effect on the morphological parameters and egg composition.
The literature used for the meta-analysis comprised three journals with seven effect sizes. The analysis outcomes showed that the studies were heterogeneous (Q=47,327, p<0.001). Thus, the REM was suitable for estimating the mean effect size of the seven analyzed studies. The results of the analysis also indicated the potential to investigate moderator variables. The results of the analysis using the REM indicated a strong positive correlation (estimate 0.74) between the addition of natural carotenoids to the diet and IgA (z=2.610; p=0.009; 95% CI 0.18, 1.29; Table 4).
Furthermore, the validation test against publication bias using Kendall’s test yielded a p-value of <0.05. It can thus be interpreted that the funnel plot formed from the REM was asymmetric, or there was evidence of publication bias. Therefore, the findings obtained from the REM regarding the viability of carotenoids against IgA were not valid, as indicated by the high I2(94.180%). Bias was nevertheless observed through Kendall’s test (<0.05; Table 4). Several factors can contribute to biased results, including an insufficient volume of literature, databases that are too small, and unpublished literature.
IgA is one of the antibodies involved in allergic reactions. These antibodies were usually found in the mucous lining and membrane, especially in the digestive and respiratory tracts. Natural antioxidants have been extensively researched to replace antibiotic function. Giving as much as 4% of the red seaweed Chondrus crispus in laying hen feed has a positive physiological effect during Salmonella enteritidis infection. The amount of IgA in red blood cells exceeded the antibiotic treatment, which was 19.83 mg/mL (Kulshreshtha et al., 2017). However, a different response was shown by another type of seaweed, the Sarcodiotheca variant. The addition of Sarcodiotheca sp. at 4% produces lower serum IgA than 2%. This demonstrated that the use of feed ingredients required a specific dose to improve the physiological function of laying hens. In the research by Zhu et al. (2021), the use of the yeast Phaffia rhodozyma as a source of astaxanthin had a positive effect on IgG parameters but did not affect IgA parameters. Increased serum IgG is associated with T-cell proliferation.
The meta-analysis used three journals with seven databases. The analysis showed that the seven effect sizes of the analyzed studies were heterogeneous (Q=12,348) as the results were p=0.05. Thus, the REM was suitable for estimating the mean effect size. The analysis results also indicated the potential to investigate moderating variables influencing the relationship between the addition of natural carotenoids and WBC. The results of the study using the REM indicated a very strong positive correlation (estimate 0.052) between the addition of natural carotenoids in the diet and WBC (z=0.467; p=0.641; 95% CI –0.17, 0.27). Furthermore, publication bias was tested using Kendall’s test (p>0.05). The investigation revealed that the p-value of the rank correlation method was >0.05. It can thus be interpreted that the funnel plot formed from the REM was symmetrical, or there was no publication bias. In conclusion, the use of the REM in this meta-analysis was valid, as indicated by a high I2(94.180%), and no bias was found using Kendall’s test (Table 4).
Like other animal species, chickens are unable to synthesize carotenoids in their bodies and must therefore acquire them from their eating regimen. Nogareda et al. (2016) investigated differences in the general health indicators of broilers fed high levels of carotenoids and controls. The relationship between carotenoids, oxidative pressure, and the immune system in seagulls was likewise examined by Lucas et al. (2014). A diet high in carotenoids may induce more rapid follicular repopulation than a control diet, thereby lessening early irritation and improving the resistant reaction (Blount et al., 2003; Chew and Park, 2004). It has also been reported that leukocyte depletion in chickens fed a diet high in carotenoids may reflect a better response to vaccines (Nogareda et al., 2016; Oladele et al., 2005). The antioxidant function of carotenoids is important for immunomodulation. However, this results in a depletion of circulating carotenoid content during periods of immune stress and a universal reduction in product pigmentation (Hamelin and Altemueller, 2012). The liver is the main stockpiling network for carotenoids and retinol, whereas serum carotenoids represent a collection of mobile pigments transported to peripheral tissues (Jlali et al., 2012; Koutsos et al., 2003).
The term carotenoids refers to the class of naturally occurring pigments (red, yellow, and orange) that are found generally in nature and were first isolated by Berzeli in the early 19th century. More than 750 types of natural carotenoids have been distinguished to date, of which 50 have been identified as playing a significant role in the organic functions of animals and humans (Mezzomo and Ferreira, 2016; Nabi et al., 2020; Pasarin and Rovinaru, 2018). Based on their compound construction, carotenoids are grouped into two divisions: xanthophylls and carotenes (Priyadarshani and Rath, 2012; Saini et al., 2015). Xanthophylls are oxygen-containing carotenoids, including lutein, zeaxanthin, and β-cryptoxanthin, while carotenes do not contain oxygen but do feature hydrocarbon compounds. Carotenes include α-carotene, β-carotene, and lycopene. Based on the compounds found within this large group, carotenoids can be grouped into provitamin A (β-carotene, cryptoxanthin) and non-provitamin A (lycopene, lutein, and zeaxanthin).
Carotenoids have various biological functions, including antibacterial and immunomodulatory properties (Arain et al., 2018; Nabi et al., 2020) that help to boost the immune system (Simons et al., 2012; Yeum et al., 2009). The antioxidant content in carotenoids can protect the body against damage caused by ROS. Poultry is incapable of synthesizing carotenoids in vivo, which means they must be provided in feed (O’Byrne and Blaner, 2013). Lutein, zeaxanthin, and canthaxanthin are commonly used carotenoids in poultry feed. Of these, canthaxanthin is considered the most effective in increasing egg yolk color. Chicken egg yolk is a natural food source of carotenoids, especially xanthophyll, lutein, and zeaxanthin, which can help to prevent cataracts and macular degeneration (Abdel-Aal et al., 2013; Demmig-Adams et al., 2020; Kljak et al., 2021). The provitamin A in carotenoids is activated in the presence of enzymes in the intestinal mucosa. These enzymes are oxygenases that break down provitamin A molecules into retinal (vitamin A aldehyde) and retinal reductase (Goodwin, 1986). Retinal reductase converts retinal into retinol. Retinol and its metabolites, such as retinoic acid, play a role in vision, immunity, brain function, tissue repair, and metabolism (Brossaud et al., 2017). The absorption of vitamin A in the digestive tract requires fat with a combination of bile acids. It is therefore possible to absorb carotenoid-containing vitamins with the help of these two substances (Silva and Furlanetto, 2018). In the early stages of chicken growth, carotenoids are distributed in the blood, liver, adipose tissue, skin, and feathers. During sexual maturation, carotenoids are then transferred to the reproductive organs, such as the ovaries, and transported by VLDL into oocytes and stored in egg yolk (Gao et al., 2020; Hansen et al., 2015; Sun et al., 2020).
Publications bias in the meta-analysis was caused by several factors. Those factors were writers who tend to enter only positive data, editors or reviewers who tend to select articles with positive results, few data obtained, and meta-analytical users who are subjective in their article selection. Moreover, it can also be caused by measurement results that are indeed positive. Fail-Safe N is an approach suggested by Rosenthal to overcome the problem of publication bias. Rosenthal assumes that studies with statistically significant results are more likely to be published than studies with non-significant results. “File Drawer” is a label given by Rosenthal to research that has not been published. The result has not been published because of insignificant outcomes. In this condition, there will be publication bias. This method can answer how many studies need to be added to reduce the results of all tests that are significant to insignificant.
Publication bias can cause the funnel to be asymmetrical. If there are more small studies on the right, then our concern is that there may be missing studies on the left. The trim and fill method removes the enormous small experiment from the positive side of the funnel plot, recalculating the effect size at each iteration until the funnel plot is symmetrical. In theory, this would result in an unbiased effect size estimate. Besides resulting in an adjusted effect size, trimming also reduces the variance of the effect and results in a narrower CI. Therefore, it is necessary to add the original research to the analysis. Trim and Fill can create funnel plots with missing (unpublished) studies, so researchers can see how the effect size shifts when missing (unpublished) research is included in the analysis. If the friction is narrow, then other people can believe the conclusions.
Conclusion
Based on the results and discussion in this study, it can be concluded that carotenoid supplementation in laying hen feed significantly improves performance (feed intake, final body weight, egg production), egg quality (egg weight, yolk colour, HU, EYC, egg yolk carotenoid), and immunity (IgA).